
Figure 1: Sonochemical reactor working at 278 kHz


Dalhatou S1,2,3* Pétrier C1,2 Massaï H3 Kouotou PM4,5 Laminsi S6 Baup S1,2
1Université Grenoble Alpes, Laboratoire Rhéologie et Procédés, F-38000 Grenoble, France*Corresponding author: Dalhatou S, Department of Chemistry, Faculty of Sciences, University of Maroua, P.O.BOX. 814 Maroua, Cameroon, Tel: +237699202687; E-mail: sadou.dalhatou@fs.univ-maroua.cm
In this work, we report the application of the high ultrasound frequency (278 kHz) to degrade nonylphenol (NP) as endocrine disrupting chemical (EDC). The degradation was performed under many experimental conditions. The effects of operating parameters such as: methanol as the type of solvent, initial NP concentration (in the limit of solubility 0.5 to 24.5 µmol.L-1), ultrasonic power (in the range from 20 to 100 W) and initial solution pH (3; 5.4; 10.2) as well as the effect of the bicarbonate ions on the degradation of NP were evaluated. Results indicated that the initial degradation rate increased slowly with the increase of the substrate concentration.The kinetic studies revealed that the sonodegradation process follows a pseudo-first-order model with the high correlation coefficient under different experimental conditions. In addition, the obtained results showed that ultrasound completely destroyed NP (8.1 µmol.L-1) after 120 min of sonication. pH had not a significant effect, due to the invariable molecular form of NP during the range of pH study, which was less than pKa of NP (10.7). Finally the results clearly demonstrated the significant intensification of sonolytic destruction of NP in the presence of bicarbonate, especially at lower pollutant concentrations. This behaviour is deleterious at high pollutant concentration.
Endocrine disrupting chemical; Nonylphenol; Ultrasound; Sonodegradation; Hydroxyl radicals
Endocrine Disrupting Chemical, nonylphenol (NP), has been widely used in industries as: surfactants, detergents, wetting agents, dispersing agents, defoamers, deinking and anti-static agents [1]. It is also used in textile, pulp and paper, tannery, food processing and plastics manufacturing [2]. However, NP and its ethoxylates derivatives are discharged in high quantities to the environment either directly or indirectly through wastewater treatment plants (WWTP) during production, use, storage, transport or during spills. Nonylphenol Polyethoxylates (NPEOs) are not stable in the environment and are rapidly degraded in nonylphenol (NP), which is more biorefractory and has strong biological cumulative effect than the parent compound [3,4].
Recent studies have revealed the presence and effect of NPEOs and their metabolites in the ecosystem [5,6]. Related research has shown that NP presents not only endocrine disrupting properties, but also serious effects involving the carcinogenesis of a variety of living organisms [7-9]. A recent study involving more than 30 000 women in the USA suggests that endocrine-disrupting chemical, EDCs, are linked to earlier menopause [10]. Thus, these adverse effects on the environment and consequently on living beings, have pushed lawmakers to regulate their uses. Following their registration as priority hazardous substances, NP and NPEOs have been prohibited on usage and placed on the market for certain usages (household cleaners, industry and agriculture) by the European Union (EU) since 2003 (Directive européenne 2003/53/EC, 18/06/2003). In France, for example, stringent legislation imposed by the EU in recent years has enabled the reduction of the use of NP and NPEOs in about 50%. However, the real problem is that rejection is much dispersed in the ecosystem. Unfortunately outsourcing or relocation of industries to others countries does not induce the same respect. Most recently in 2011, the Non-Government Organism Greenpeace denounced in the countries such as China, Pakistan, Philippines, Finland, the use of NPEOs by major brands of shoemaking and clothing [11]. The problem comes again from the different application of rules across countries. In developing countries for example, no rigorous legislations exists on the use of NP.
Today, because of the low cost of NPEOs, their substitution by fatty alcohol ethoxylates in developing countries seems to be unrealistic. Therefore, the most appropriate solution is the treatment of contaminated water. For that purpose, many processes have been applied for the abatement of NP and NPEOs [12-17]. Among the advanced oxidation processes (AOP), ultrasound (US) have received an increased interest in the last years [18,19]. In fact, this technique generates a strong hydroxyl radical (OH• ) with an oxidation potential of 2.80 V [20] through the acoustic cavitations which can be defined as the cyclic formation, growth and collapse of microbubbles. Fast collapse of the bubbles compresses adiabatically gas and vapour entrapped, which conducts to short and local hot spots [21]. At the final step of the collapse, temperature inside the residual bubble is thought to be above 2000 K [22]. Under the second it ions, entrapped molecules of dissolved gases, vaporized water and solutes can be brought to an excited state and dissociate. Therefore, OH• radicals are generated from water (H2O) and oxygen (O2) dissociation [23]. In absence of any organic compound, OH• radicals recombine to form hydrogen peroxide (H2O2) that released in medium according to the following reaction:
HO•+HO•→H2O2k1= 5,5×109M-1.s-1 (1)
Ultrasound is a simple technique, requiring only electrical energy and easy to handle. But the sole application of ultrasound for treating polluted water is too time consuming and expensive, this operating cost could be partially off-set by operating at milder and optimum operation conditions, which enhance the degradation rate, reduce the reaction time [18]. However, real application or transfer to large scale requires knowledge of the basic principles and investigations of the influence of reaction parameters to achieve the optimum conditions.
To the best of our knowledge, few works in the literature report on the use of pure ultrasound to destroy nonylphenol in aqueous media [24,25]. As an example, Ince et al. [26] have worked on the effect of pH and inhibitors of hydroxyl radicals on sonochemical destruction of NP at low frequency 20 kHz. The authors found that the rate of degradation was more accelerated by alkalinisation, with the addition of hydroxide alkalinity than carbonate. The addition of low CO32- concentration and t-butyl alcohol as strong scavengers of •OH were also found to accelerate the decomposition of NP. Also, Gultekin et al. [27] studying the wastewater decontamination by ultrasound reported that the NP sonodegradation was accelerated by bubbling oxygen gas and by addition of Fenton and Fenton-like reagents (Fe2+ and Cu2+) into the solution. According to Xu et al. [28] investigation, NO3− accelerated NP sonophotolysis while HCO3− showed insignificant influence.
There have been many studies aimed at developing effective methods for degrading aromatic compounds such as phenol and related compounds, including the process of ultrasonic degradation. Ultrasonic processes could be an effective alternative way for oxidation and completely mineralizing recalcitrant organic compounds. Also, by converting the pollutants in to less harmful or lower chain compounds, more efficient biological treatment on the wastewater could be achieved.
Yim et al. [25] shown that the ultrasonic degradation of NP conducted in the first stage to quinones, organic acids and aldehydes; the next stage brings about a ring opening to form carboxylic acids and other compounds with lower carbon numbers such as CO, CH4, C2 H2, C2H4, and C2H6 then were eventually converted to carbon dioxide and water. This mineralization was confirmed by the removal of 50% of the initial Total Organic Carbon (TOC).
However, many authors used methanol as solvent of NP dissolution, yet it is an inhibitor of hydroxyl radicals. This character could be detrimental to the effectiveness of the ultrasonic technique. Herein, our purpose is to investigate the viability of high frequency ultrasound to the treatment of water contaminated with NP. To elucidate the respective role of different parameters, the effect of methanol as solvent, the initial NP concentration, the ultrasonic power, the initial pH and the bicarbonates ions have been evaluated.
4-Nonylphenol was supplied by Sigma–Aldrich, used without any purification. The physical and chemical properties and the molecularstructure of NP are shown in table 1 [29]. Potassium iodide (KI) was supplied by Chimie Plus. Ammonium heptamolybdate (NH4)6Mo7O27.4H2O, acetonitrile (CH3CN) and Sodium hydroxide were purchased from Across Organics, sulfuric acid (H2SO4) came from Merk, methanol (CH3OH) was supplied by Fluka, sodium bicarbonate (NaHCO3) was provided by Prolabo (analytical grade). The water used to prepare solutions and clean thereactor and glassware was purified with activated carboncartridges and then deionized to ion exchange resins, from Fisher Bioblock Scientific.
Compound |
4-Nonylphenol |
Structure of NP |
Formula |
C H O |
|
Molecular Mass (g/mol) |
220.60 |
|
Density |
0.937 |
|
pKa at 25°C |
10.70 |
|
Vapor Pression (Pa) |
(4.55 ± 0.35) |
|
Water solubility (mg/L) |
5.43 |
|
logK |
4.20-4.48 |
|
Constante of |
11.02 |
|
Analysis method |
HPLC |
Table 1: Physical and chemical properties of NP
The power system is a high-frequency generator (Radiocoms Systemes ULV300A), which delivers a 278 kHz frequency (Figure 1). The impedance of this generator is set at 50 Ω and regulated by an impedance adapter. At the base of thereactor, a piezoelectric disc is fixed (diameter 4 cm) on a Pyrex plate (5 cm diameter) enabling the transmission of the ultrasonic waves in the solution to be treated. The cylindrical reactor (400 mL) is double-jacketed inorder to maintain the system at a constant temperature (20 ± 1C) to avoid thermal fluctuation induced byultrasound which could influence kinetics of reactions. Temperature is monitored by a rmocouple NiCr-NiAl of 1 mm of diameter (connected to a digital display 380, PMA Prozeb Type 9404 380 62001) immersed in the solution. The water iscooled by a cryostat (Heto CB 8-30e).

Figure 1: Sonochemical reactor working at 278 kHz
NP is very viscous and slightly soluble in water. 5,4 mg of the NP sample corresponding to its limit of solubility, is mixed with deionized water, then the solution was heated up to a temperature of 50°C to facilitate dissolution. This temperature is reported to do not alter the NP [30]. After cooling, serial dilutions were made according to our needs and the resulting solutions are used immediately. The solutions were kept below 15°C, to avoid deposition of the NP on the wall of the vial.
Quantitative analysis of the Endocrine disrupting compounds NP was performedby the mean of HPLC using a Waters 515 instrument equipped with a SupelcosilLC-18 column (ID=4.6 mm, length=250 mm, dp=5 µm). The samples were injected using a Rheodyne injection system with a 200 µL sample loop for NP concentrations. The detection of the absorbed organic compounds was realized with a UV detector (Waters 486) set at 190 nm. The mobile phase, water/acetonitrile (v/v: 20/80) was degassed before analysis in an ultrasound vessel (Branson 1200) and filtered with Millipore filter in an isocraticmode. Using this methodology, an acceptable detection limit of NP was reached. The experimental error was calculated from the variance of data, and the reported result was the reproducible average value, within an error of 10%.
H2O2 concentration is analytically determined by the spectrophotometric method, using potassium iodide (1 gL-1) and ammonium heptamolybdate salt as catalyst (10-4 Mol.L-1). Aliquots (1 mL) were taken from the reactor and were immediately added in the sample quartz bowl of the Shimadzu UV-mini 1240 spectrophotometer containing the reagents. Absorbance at a 350 nm wavelength was recorded after 5 min.
In order to evaluate the effect of methanol as solvent on the effectiveness of sonochemistry, the NP solution (8.1 µM) was submitted in different concentrations of methanol namely 0, 25, 250 and 2500 ppm. Figure 2 shows the Sonodegradation of 4-NP at various concentrations of methanol. As shown on figure 2, the presence of methanol reduced the sonodegradation rate of NP. In fact, as methanol is well known to be scavengers of hydroxyl radicals, the efficiency of the process can be reduced as shown in equation 2.
CH3OH+ HO• → CH3O• + H2O (2)
Numerous studies on the NP in the literature, reported the effectiveness of methanol as dissolution solvent for NP [26]. However, methanol presents a potential negative effect on the AOP during the generation in situ of HO• radical. As an example, Neamtu and Frimmel investigated the photolysis of NP using a solar simulator in aqueous solution and determined the intermediates using HPLC [31]. However, the presence of methanolused as solvent was suggested to influence the photo-degradation process.
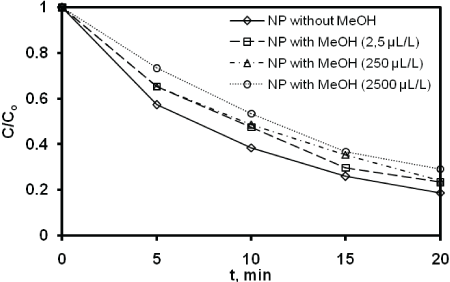
Figure 2: Sonodegradation of 4-NP at various concentrations of methanol (frequency: 278 kHz; power: 100W; volume: 400 mL; pH: natural (5.4); temperature: 20 ± 1C).
In water treatment, kinetics is one of the most studied aspects to assess a process since it gives information on the rate of removal of pollutants in water. In sonochemistry for example the degradation rate of a compound is closely related to its degradation zone namely gas phase, gas-liquid interface and liquid phase [22]. In addition, access to different areas strongly depends on the physical and chemical properties of the compound. However, as a surfactant, the relative proportions of NP in different zones may also depend on the initial concentration of NP [32].
In this work, we are inspired from the existing kinetic models [33- 36], to study the kinetics of NP sonodegradation at 278 kHz. According to Ince et al. [26] the overall results shows that the kinetic model of degradation of NP at 20 kHz was pseudo first order for concentrations more than 20 µM. To our knowledge, no kinetic work was done at NP concentrations less than 20 µM. To complete this information, we were interested in the concentration range between 0.50 and 24.32 µM (limit of solubility).
Figure 3 shows the profile of the evolutionof concentration versus ultrasonic irradiation time in the range of 0.50 to 24.32 µM. We have observed that the NP was degraded more rapidly during the first 30 minutes with a minimum of 80% of abatement rate for the different concentrations, and disappears completely after 120 min. It is observed from 60 min treatment stabilization curves. This would show the difficulty of ultrasound to mineralize very highly diluted solutions. Furthermore, it is found that the concentrations of NP (0.50 µM from 24.32 µM), remaining in solution after treatment, are almost identical and correspond to the HPLC detection limit.A description of the profiles shows that the disappearance of the NP is a pseudo-first order kinetic.
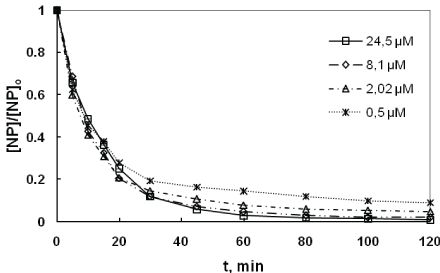
Figure 3: Sonodegradation of 4-NP at various initial substrate concentrations (frequency: 278 kHz; power: 100 W; volume: 400 mL; pH: natural (5.4); temperature: 20 ± 1C).
In fact, the NP is a hydrophobic and non-volatile compound with a bipolar structure (hydrophobic tail and hydrophilic head) [37]. It also has the surfactant properties [38,39]. These characteristics are favorable for their adsorption on the interfacial layer gas/liquid of the bubble where the density of OH• is higher [40]. Therefore, degradation mainly takes place at the interface of two essential ways that are radical attack (Equation 3) and pyrolysis [24,25].
HO• + NP → Products (3)
Figure 4 depicts the initial NP degradation rate versus initial NP concentration. It was found that the initial rate of sonolytic degradation increased with increasing the initialNP concentration. At low concentrations, a linear relationship (R2 =0.998) between the rate and concentration reminiscent of a first-order kinetics. But this linearity is not observed when the concentration exceeds a certain threshold (C>16.22 µM).
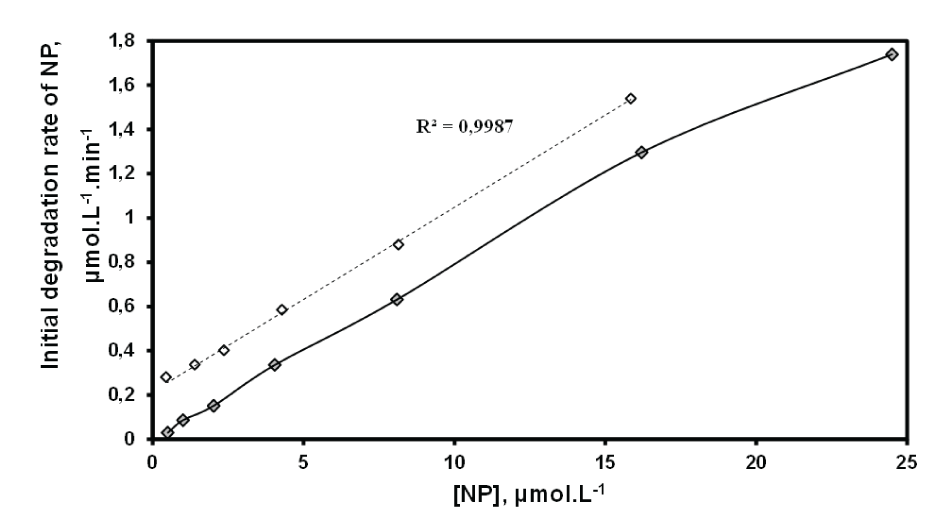
Figure 4: Initial degradation rate of NP versus initial concentration (frequency: 278 kHz; power: 100 W; volume: 400 mL; pH: natural (5.4); temperature: 20 ± 1C).
The size of the cavitation bubble is inversely proportional to the frequency (D=6.62/frequency), which necessarily implies that the bubble is smaller at 278 kHz (high frequency) compared to 20 kHz when it is relatively larger [26]. As frequency enhances, the pulsation and collapse of the bubble occur more rapidly and more radicals escape from the bubble. However, at high frequency the acoustic period is shorter and the size of cavitation bubbles decreases. As a consequence, the cavitation threshold becomes higher and cavitation intensity decreases. In addition, the interface of the cavitation bubble is a site of adsorption potential which remains constant throughout the operation [35]. This leads to a faster saturation of the bubble’s surface even at low concentration of the adsorbate. However the advantage of this system (278 kHz and 100 W) based on the presence of a large number of bubbles and the implosion more violent, resulting in a higher degradation of the pollutant. The decrease in the degradation rate at 16.22 µM could be explained by the saturation of adsorption sites and the higher competition at the interface [24]. After the examination of NP disappearance profiles in the range of the studied concentrations, it was shown that the sonodegradation of NP is performed according to a pseudo first order kinetics.
Figure 5a showed that the relative concentration of NP decreases with treatment time. A quantitative description of the profiles shows that the degradation rate increases linearly when the power increases (Figure 5b). For example, the degradation rate increases from 0.17 µM/min to 0.47 µM/min respectively at 20 W and 100 W. The increase of the reaction rate can be explained by the growth of bubbles resulting from the increase of acoustic amplitude [41]. In fact the latter causes a growth of temperature, a faster and more violent implosion of cavitation bubbles that are smaller and numerous, resulting in a high concentration of HO• radicals in the bubble interface [39]. At this level, a competition between the HO• radical recombination reaction and the degradation of NP by HO• radical takes place. On this aspect, increase in the H2O2 production is observed when the power grows, either in the presence or absence of pollutant (Figure 6). This trend shows that the involvement of the acoustic power on the production of HO• radicals is consistent with the literature [42-44].
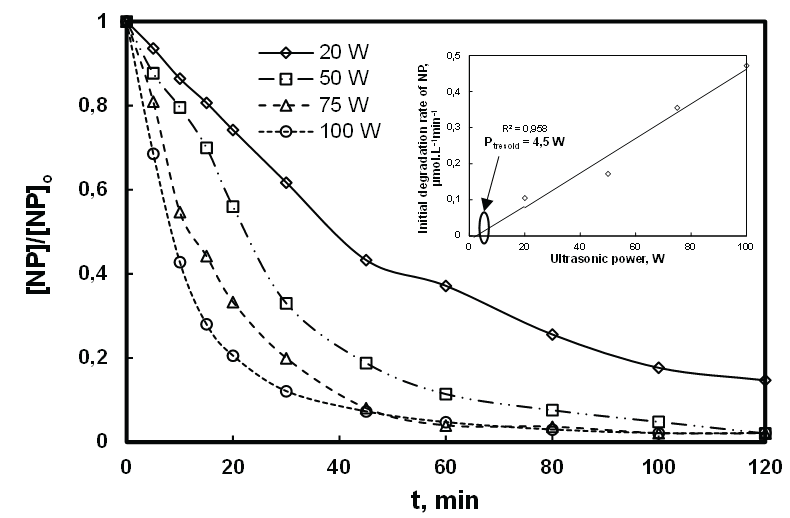
Figure 5: Effect of acoustic power on the sonodegradation of NP ([NP]0 : 8.1 µM, frequency: 278 kHz; volume: 400 mL; pH: natural (5.4); temperature: 20 ± 1C).
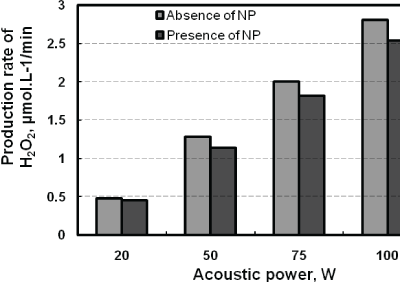
Figure 6: Effect of acoustic power on H2O2 production rate in absence and in presence of NP ([NP]0 : 8.1 µM, frequency: 278 kHz; volume: 400 mL; pH: natural (5.4); temperature: 20 ± 1C).
The pH is an important factor for determining the sonochemical reaction rate. However, previous work has shown that the pH has a negligible effect on the degradation of surfactants [45]. In this regard, some authors have observed that the pH has an effect on the degradation of organic compounds and more specifically surfactants [27]. It is worth mentioning that, the acidification of the medium increases the hydrophobicity of the molecule enriched by protonation of the phenolic moiety that can lead to accelerate degradation of the compound. Everything depends on the ionized functional group. For example, it has been demonstrated that the sonodegradation 4-nitrophenol (alcohol moiety) decreases with increasing pH, whereas the aniline (amine functional group) has an opposite behavior [46].
In this work, we irradiated at 278 kHz and 100 W the solution of NP at relatively high (8.1 µM) and low concentrations (0.5 µM) by varying the pH (3 to 5.4 and 10). As it can be observed in figures 7 and 8, the acidification of the medium (pH=3) remarkably improves the degradation of NP at both higher and low concentration respectively 8, 1 μM and 0.5 µM. With basic constant pH (pH = 10), the effect of pH is not enough significant for a concentration of 8.1 µM, while this effect is slightly positive at low concentration of NP (0.5 µM). Indeed, whatever the pH of the study, the NP is under a neutral molecular form because its pKa is 10.7 (Figure 9). However, at pH 3 HO• diffuses more easily through the bubblesolution interface that is very rich in radicals HO• where degradation of NP is slightly higher, compared to reaction at pH 10. In addition, the carbonation phenomenon of the medium with dissolved CO2 at basic pH, developed in the previous study is confirmed here [46], where one can notice a very rapid deterioration of the NP in the vicinity of the bubble/solution interface at low concentration of pollutant and pH 10. Therefore it may be necessary to question the potential effect of bicarbonate on a non-volatile hydrophobic compound such as NP, based on the fact that it’s positively affects the elimination of hydrophilic organic compounds, but not volatile at low concentration [45].
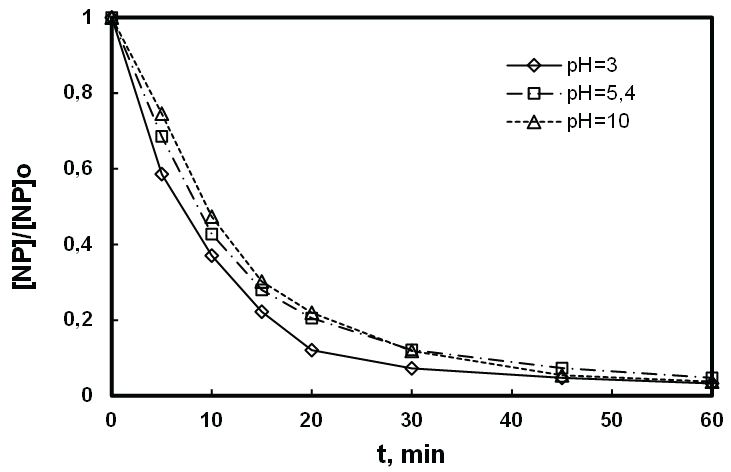
Figure 7: Effect of pH on the sonodegradation of high relative concentration of NP ([NP]0 : 8.1 µmol.L-1, f: 278 kHz, P: 100 W, V: 400 mL and T: 20 ± 1°C).
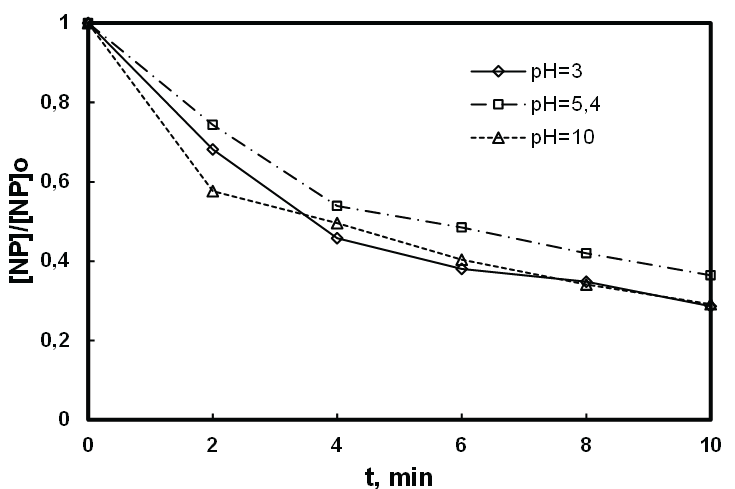
Figure 8: Effect of pH on the sonodegradation of low relative concentration of NP ([NP]0 : 0.5 µmol.L-1, f: 278 kHz, P: 100 W, V: 400 mL and T: 20 ± 1°C).
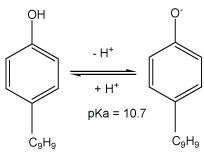
Figure 9: Molecular structure of NP at condition acidic and basic condition.
To get an idea of the effect of bicarbonates ions (HCO3-) on sonochemical degradation of a hydrophobic and non-volatile compound, solutions of NP in the limit of solubility (0.5 to 24.5 µM) were treated by ultrasonic irradiation at 278 kHz and 100 W in the absence and presence of HCO3- ions (2.97 mM). The experimental results are summarized in figure 10. It is apparent for these results that the addition of bicarbonate ions in the sonochemical degradation of NP improves processing efficiency. It is seen in this figure that the effect of bicarbonate is more pronounced at low concentrations of the pollutant where an improvement factor was 3 for 0.5 µM of NP. This effect decreases as the concentration of the pollutant increases, then becomes zero at about 19 µM and becomes negative beyond this concentration (Figure 10).
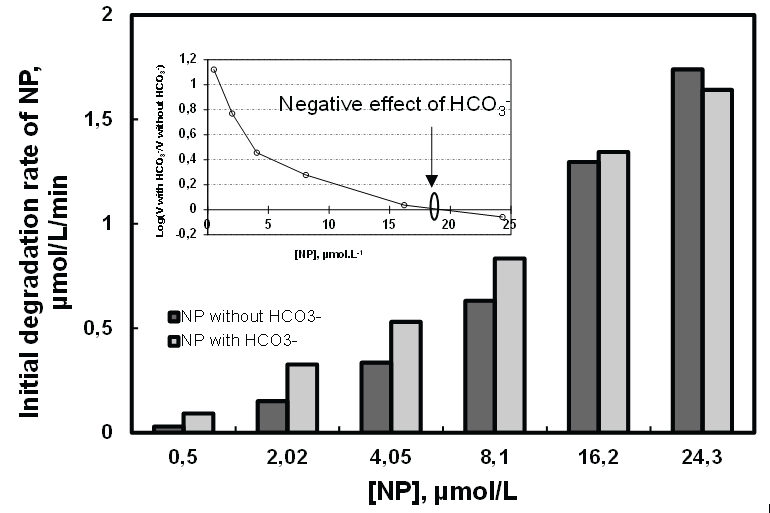
Figure 10: Effect of bicarbonates ions on the sonodegradation of NP ([HCO3-]=2.97 µmol.L-1, f=278 kHz, P=100 W, V=400 mL and T=20 ± 1°C).
Indeed, bicarbonate ions react in solution with HO• radicals during the sonolysis (K4=8.5 × 106 M-1.s-1) giving place to bicarbonates radicals (Equation 4) that are released into the solution during the implosion of the bubble. Bicarbonates radicals are characterized by their oxidizing power (E=1.59 V/SHE) [46], their stability, their high mobility and their higher selectivity than the HO• radical. This justifies the increase in performance of the art at low concentration of micro pollutant [47]. At high concentration of the micropollutant, on the contrary, the competition phenomenon takes place. HO• radicals capable of reacting with HCO3- ions are intercepted by the NP molecules in high concentration at gas/ liquid interfacial region and therefore, will form very few CO3-• radicals which are inadequate to enhance the degradation of NP but enough significant compared to the HO• radicals consumed. The results show that at high concentration of NP, the HCO3- ions have an inhibiting effect on the degradation of NP [36]. Overall, these results are consistent with the literature [48].
HCO3- + HO• → CO3-• + H2 O k26 = 8,5x106 M-1.s-1 (4)
CO3-• + NP → Products (5)
At about 19 µM, the effect of bicarbonate ions becomes harmful (Figure 11) and the logarithm of ratio of NP sonodegradationrate in thepresence and absence of bicarbonate ions becomes negative. For any concentration of NP>19 µM, there will be a speed reduction which corresponds to the negative effect of bicarbonates.
We also highlighted the effect of competition by studying the influence of NP (8.1 µM) and bicarbonates anions (2.97 µmol.L-1) on the H2O2 production at 278 kHz during a period of 20 min. Then results are displayed in figure 11. It is observed that the concentration of NP decreases exponentially, according to a pseudo-first-order kinetics (as explained in paragraph of the section Effect of NP concentration on the destruction process). Furthermore, H2O2 formation increases linearly with the ultrasonic irradiation time, in absence and presence of NP. However, in the presence of NP, the formation of H2O2 is reduced compared to the same experiment without NP in deionized water. Indeed, the presence of NP in solution, induces to HO• radicals the competition between its sonodegradation reaction and its recombination (Equations 3 and 4), favoring the decreases of the H2O2 concentration. The decrease of H2O2 formation in the presence of NP attest of the involvement of HO• radical in NP sonodegradation; Moreover, competition increases with increase the ions HCO3 - in solution as demonstrated in figure 11. Thus it results in decrease of H2O2 production due to the inhibition of HO• radicals by HCO3- ions. However, the degradation of NP increased in the presence of HCO3- ions as consequence of the electrons transfer from HO• radicals to CO3-•, which responsible of additional NP degradation.
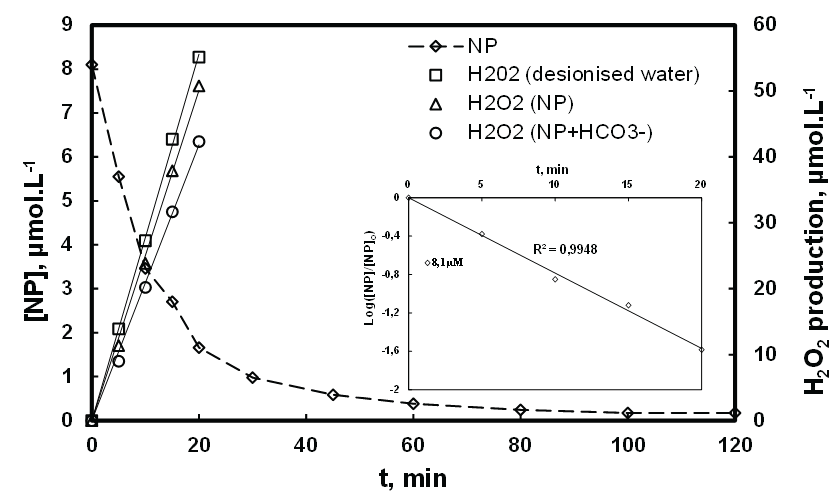
Figure 11: H2O2 production in presence and in absence of NP ([NP]0 : 8,1 µM, f: 278 kHz, P: 100 W, V: 400 mL and T: 20 ± 1°C).
The study has shown that the degradation of Endocrine disrupting compound NP can be effectively reached at 278 kHz ultrasonic irradiation. NP sonodegradation in water occurs mainly through reactions with hydroxyl radicals at gas/liquid interfaceand its kinetic respectsthe pseudo-first order model. The extent of degradation was inversely proportional to the initial concentration of substrate. The initial sonodegradation rate of NP at different concentrations shows that, the higher initial decomposition rate is achieved with the highest substrate concentration. In addition, pH presents no significant effect, the most favorable condition for the degradation of low concentration of NP was observed in basic media, whilethe acoustic power increases with the increaseof the ultrasonic power from 20 to 100 W. More importantly, the amount of hydrogen peroxide produced was decreased in presence of NP and bicarbonates.
The authors wish to express their gratitude to the “Service de Coopération et des Actions Culturelles” (SCAC) of the French Embassy to Cameroon for their financial support.
Download Provisional PDF Here
Article Type: Research Article
Citation: Dalhatou S, Pétrier C, Massaï H, Kouotou PM, Laminsi S, et al. (2016) Degradation of Endocrine Disrupting Chemical Nonylphenol in Aqueous Milieu using High Frequency Ultrasound. Int J Water Wastewater Treat 2(4): doi http://dx.doi. org/10.16966/2381-5299.129
Copyright: © 2016 Dalhatou S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access