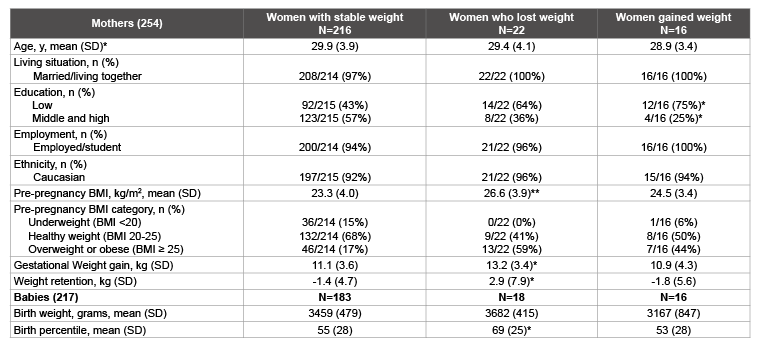
Table 1: Characteristics of the women and babies
*P<0.05; ** p<0.01 compared to women with stable weight before pregnancy, tested with student T-test for continuous variables and Chi square test for
categorical variables.

Carla L van der Wijden1,2,* Henriette A Delemarre-van der Waal3 Willem van Mechelen1 Mireille NM van Poppel1
1Department of Public and Occupational Health, EMGO Institute for Health and Care Research, VU University Medical Center, Amsterdam, The Netherlands*Corresponding author: Carla L van der Wijden, Department of Public and Occupational Health, EMGO Institute for Health and Care Research, VU University Medical Center, Van der Boechorststraat 7, 1081 BT Amsterdam, The Netherlands, Tel: 31628854239; Fax: 312304448387; E-mail: c.vanderwijden@vumc.nl
Objectives: To study the effect of weight changes before pregnancy on maternal weight development during and after pregnancy and on birth weight. Because physical activity, smoking, breastfeeding and sleeping might play a role in weight development, they were taken into account.
Methods: In a longitudinal cohort study in 254 women, anthropometric measurements were performed at 15, 25, and 35 weeks of gestation and 6, 26 and 52 weeks postpartum. In addition, women reported on weight changes in the six months preceding pregnancy, and on smoking, sleeping, and breastfeeding. Physical activity and sedentary behaviour were measured objectively using accelerometers. Of 217 babies birth weight and birth percentile were reported.
In linear regression models, the relation between weight changes before pregnancy with gestational weight gain, pregnancy outcome and weight retention was assessed, controlling for lifestyle factors.
Results: About 9% of all women lost more than 5% of body weight before pregnancy, and another 6% gained more than 5% of body weight. Women who lost weight had a significantly higher BMI throughout pregnancy and the postpartum period compared to women with a more stable weight. After adjusting for BMI at 15 weeks, age, and lifestyle factors, women who lost weight had significantly more weight retention (beta 3.64 kg, 95% CI 0.57 to 6.70 kg) and delivered heavier babies (beta 391.5 g, 95% CI 106.3 to 676.7 g).
Conclusions: Maternal body weight change preceding pregnancy influenced birth weight and maternal weight retention. These findings should be confirmed in a much larger sample.
Maternal weight; Weight retention; Pregnancy outcome; Lifestyle; Preconceptional weight change
Pregnancy is considered to be an important period for the development of subsequent maternal overweight and obesity, or worsening already existing overweight [1,2].
Guidelines on gestational weight gain exist [2], in which the importance of a normal weight before pregnancy and weight gain within certain limits during pregnancy is emphasized. A normal weight before and controlled weight gain during pregnancy are related to ‘keeping in shape’, a better pregnancy outcome and less obesity in the offspring [2,3].
However, many women do not gain weight according to these guidelines [4]. Althuizen et al. [5] showed that the overall weight retention at one year postpartum was 0.9 kg and 20% retained ≥ 5 kg. There are a growing number of trials, studying the effect of dietary and physical activity interventions on the prevention of excessive weight gain during pregnancy. According to a recent review, the effect of these interventions on weight gain is moderate to very small and maternal and neonatal morbidity were not always taken into account [6]. Unfortunately most intervention studies had a short follow up, making it difficult to assess the role of intervention effects on maternal weight development in the long term. Based on the findings of this review gestational weight gain seems to be difficult to influence in pregnancy.
Whereas weight gain in pregnancy has received much attention, studies on the effects of weight changes before pregnancy on the health of mother and offspring are scarce. It has been demonstrated that in a normal weight population, maternal weight loss before pregnancy was positively associated with a risk of a small baby and maternal risk of gestational diabetes and hypertension [7]. The authors concluded that “in humans, maternal weight dynamic before conception could play a role in the process of early-life programming”. This also applies to interpregnancy weight changes. One study showed that weight loss between pregnancies was associated with small for gestational age birth, while weight gain increased the risk of large for gestational age birth at the second pregnancy [8]. Another study showed that interpregnancy weight gain, even in women with a normal BMI, was associated with adverse pregnancy outcomes [9].
We hypothesized that considerable weight change preceding pregnancy might result in different weight development during and after pregnancy. Furthermore, we hypothesized that weight changes before pregnancy might influence pregnancy outcome, in terms of birth weight and/or birth percentile. The objective of this study was to study the relationship of weight changes before pregnancy with pregnancy outcome and gestational weight gain and postpartum weight retention. The role of lifestyle factors (smoking, physical activity, sleeping and breastfeeding) was taken into account.
In the New Life (style) study, a randomized controlled trial (ISRCTN 85313483), healthy women expecting their first child and receiving antenatal care from midwives, were randomized to standard care or to an intervention group [10]. In the intervention group, at 18, 22, 30 and 36 weeks of pregnancy and at 8 weeks postpartum, individual weight gain was discussed in the midwifery practice in relation to weight gain guidelines for pregnant women of the American Institute of Medicine [2]. The intervention did not have an effect on gestational weight gain, weight retention [5] and physical activity. Therefore, in this secondary analysis, the data were analyzed combining the intervention and control group into one cohort and focusing on the relationship between weight change before pregnancy and gestational weight gain and weight retention postpartum.
Healthy women were eligible to participate in the trial if less than 14 weeks pregnant (first ongoing pregnancy) and fluent in Dutch. Exclusion criteria were serious maternal complications during pregnancy and in the year after or if the fetus/baby died. Of the 708 women who were invited to participate, 254 women (36%) were available for the baseline measurements.
The Medical Ethics Committee of VU University Medical Center approved the study design, protocols and informed consent procedure (registration number 2004/184), and all participants signed written informed consent.
Women who were not married were asked if they lived alone or together with their partner. They were asked about the highest level of education to have achieved. Low education means lower secondary education (International Standard Classification of Education) Middle means higher lower education and high means tertiary education.
Measurements were carried out in participating midwifery practices at 15 (T1), 25 (T2), 35 (T3) weeks of pregnancy and at 6 (T4), 26 (T5), and 52 (T6) weeks after delivery. Body height was measured in bare feet by means of a wall-mounted height scale (SECA 206), with an accuracy of 0.1 cm. Calibrated electronic scales (SECA 888) were used to determine body weight of the participants in underwear and pants, with an accuracy of 0.1 kg. Both body weight and body height were measured in duplicate, and the mean values were taken to calculate individual body mass index (BMI, kg/m2 ).
Prepregnancy BMI was calculated based on self-reported prepregnancy body weight just before pregnancy and objectively measured body height. Weight gain in pregnancy was defined as the objectively measured weight at 35 week minus weight at 15 weeks. Weight retention was the measured weight at 52 weeks post partum minus the measured weight at 15 weeks of pregnancy.
At 15 weeks pregnancy, women reported on substantial body weight changes in the six months before pregnancy. The percentage of body weight change was calculated as body weight change divided by prepregnancy body weight. We considered a change (gain or loss) in body weight of 5% or more as a considerable weight change. The terms “women who gained weight” and “women who lost weight” will be used throughout this paper, for women who gained or lost more than 5% of body weight preceding pregnancy, respectively.
Daily physical activity (PA) was measured using an accelerometer (ActiGraphTM, Pensacola, FL, USA) for four days at 15 (T1) and 35 (T3) weeks of pregnancy and 26 (T5) weeks after delivery. Data from the days with at least 8 hours registration time were used. Total counts per minute were converted into light, moderate and vigorous PA (100 to 2019 counts/ min for light PA, 2020 to 5998 counts/min for moderate PA and ≥ 5999 counts/min for vigorous PA) [11]. Sedentary behaviour was defined as <100 counts/min. The time participants spent being sedentary and conducting moderate-to-vigorous PA (MVPA) was calculated.
At 15 weeks of pregnancy, women were asked if had ever smoked and if they were currently smoking. Non smokers, who did smoke in the past were asked how long ago they quit smoking and for what reason. At the five other time points they were asked if they currently smoked.
At all six time points, women were asked how many hours per day they slept. Mean sleeping hours in pregnancy (average of 15, 25 and 35 weeks of pregnancy) and mean hours sleep postpartum (average of 6, 26 and 52 weeks postpartum) were computed. In the questionnaires at 6, 26 and 52 weeks postpartum, the following items had to be filled in: “Did you breastfeed?” If so, how often did you breastfeed and until what age. The number of months with any breastfeeding was assessed based on this information.
Mothers reported the date of birth of their baby, mode of delivery, complications and birth weight of their baby. Birth percentiles (weight adjusted for gestational age and sex) were calculated according Dutch reference curves [12].
The maternal characteristics of the participants are presented as means and standard deviations for continuous variables, and as percentages for categorical variables. For the outcomes gestational weight gain, birth weight, birth percentile, and maternal weight retention, linear regression analysis was used to test the association between prepregnancy body weight changes. In the first step of the analyses, all the models were controlled for maternal BMI at 15 weeks of pregnancy, maternal age, and allocation to intervention group or control group. In addition, the model for gestational weight gain was controlled for the difference in gestational age between the measurements at 15 and at 35 weeks, since this varied between participants. The model for birth weight was additionally controlled for gestational age at birth and gestational weight gain, and the models for birth percentile and weight retention for gestational weight gain. In the second step of the analyses, lifestyle factors were added to the models, in order to assess whether the association between weights changes before pregnancy with outcomes were was independent from lifestyle. For gestational weight gain, birth weight and birth percentile, data on MVPA, sedentary behaviour and smoking from 15 weeks of pregnancy were used, and the average number of hours of sleep in pregnancy. For weight retention, data on MVPA and sedentary behaviour from 26 weeks postpartum were used, data on smoking from 52 weeks postpartum, and the average hours of sleep in the year postpartum. All analyses were performed in SPSS20.0 (IBM Corporation, Armonk, NY, USA).
The characteristics of the 254 women included in the analyses and their babies are presented in Table 1. Women were on average 27.7 (SD 3.9) years of age with a mean prepregnancy BMI of 23.7 (SD 4.0) kg/m2 . For 217 babies data were available. The mean birth weight was 3456 (SD 516) gram, and 17 (8%) babies were below the 10th percentile, and 29 (14%) above the 90th percentile.
Weight gain from 15 to 35 weeks of gestation was 11.3 kg on average (SD 3.7 kg). Weight change from 15 weeks of gestation to 52 week postpartum was -1.0 kg (SD 5.3) on average (Table 1). The mean BMI increased from to 24.9 (SD 4.2.) at 15 weeks to 28.9 (SD 4.4) at 35 weeks of gestation and went down to 24.6 (SD 4.6) at 52 weeks postpartum (Figure 1).
A total of 38 (15%) women had substantial weight change preceding pregnancy: 22 (9%) women lost body weight and 16 (6%) women gained weight in the six months preceding their pregnancy. Although all groups showed a similar pattern of weight changes over time, women with a more stable body weight before pregnancy had the lowest BMI throughout the whole pregnancy and postpartum period (Figure 1). Women who lost body weight had the highest BMI throughout all time points (p<0.001 compared to women with stable weight). Differences in BMI between women with stable weight and women who gained weight were not statistically significant.
Albeit 10% of the women smoked at 15 weeks of pregnancy, 42 women quit smoking within the year preceding pregnancy. Of those 42 women who stopped, 88% stopped because of wanting to get pregnant. At 6 months postpartum about half of the women, who quit smoking in the year before pregnancy, had started again.
Women spent on average 10 min/day (SD 11.0) in moderate to vigorous physical activity (MVPA) in early pregnancy, 3.8 (SD 5.2) min/ day MVPA in late pregnancy and 11.5 (SD 10.6) min/day MVPA at 26 weeks postpartum. The time the women spent sedentary was relatively stable, with 1322 (SD 71) min/day in early pregnancy, 1343 (SD 61) min/ day in late pregnancy and 1316 (SD 44) min/day 26 weeks postpartum.
During pregnancy, women slept on average 8.25 (SD 1.5) h/day, and postpartum this was 7.4 (SD 1.0) h/day on average. In total, 60.6% of all women started breastfeeding with a mean duration of 4.0 (SD 3.7) months.
Gestational weight gain in women who lost or gained substantial weight before pregnancy was not different from women with stable weight before pregnancy, adjusted for BMI at 15 weeks, age, intervention group, and the time between the measurement at 15 and 35 weeks (Table 2). Adjusting for lifestyle factors did not change this result. Women who lost weight before pregnancy gave birth to significantly heavier babies (391.5 gram, 95% CI 106.3 to 676.7 gram), with a higher birth percentile (24.3, 95% CI 4.0 to 44.6), after adjusting for lifestyle factors. These women also had significantly more weight retention at 52 weeks postpartum (3.64 kg, 95% CI 0.57 to 6.70 kg).
Maternal BMI in early pregnancy and most lifestyle factors (MVPA, sedentary behaviour, breastfeeding, sleeping duration) did not influence gestational weight gain, birth weight or birth percentile as shown in Table 2. Of 217 babies data on birth weight and birth percentile were available.
The only lifestyle factor associated with birth weight was smoking. Women who smoked in pregnancy gave birth to significantly lighter babies (328.3 gram, 95% CI -577.5 to -79.2 gram). Smoking and sleep duration were associated with weight retention. Women who smoked at 52 weeks postpartum had less body weight retention (-4.75 kg, 95% CI -8.17 to -1.33 kg) at 52 weeks postpartum and women who slept longer on average in the year postpartum had more weight retention (0.02 kg, 95% CI 0.004 to 0.04 kg).
Childbearing is considered to be a significant risk factor for developing overweight and obesity. Prepregnancy weight, gestational weight gain and lifestyle factors might influence weight retention and pregnancy outcome [1,2]. In this study we assessed the influence of substantial weight changes before pregnancy on gestational weight gain, pregnancy outcome and weight retention. Surprisingly we found that maternal weight loss preceding pregnancy was related with a higher birth weight, birth percentile and more weight retention.

Table 1: Characteristics of the women and babies
*P<0.05; ** p<0.01 compared to women with stable weight before pregnancy, tested with student T-test for continuous variables and Chi square test for
categorical variables.
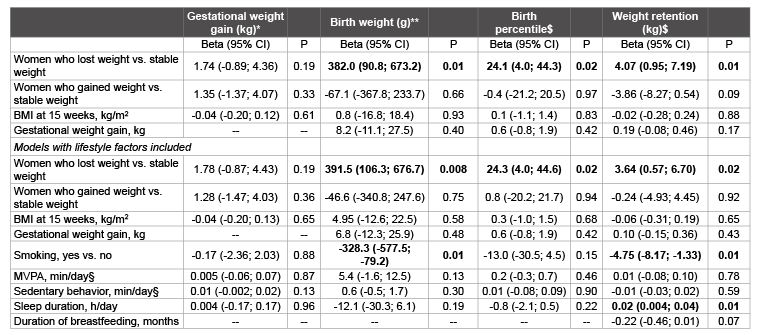
Table 2: Results from linear regression models, assessing the association between body weight changes before pregnancy with gestational weight gain,
birth weight and percentile and weight retention postpartum, with and without controlling for lifestyle factors.
BMI = body mass index, MVPA = Moderate-to-Vigorous Physical Activity.
* model additionally adjusted for time between the two measurements (15 and 35 weeks of gestation), maternal age and intervention group
** model additionally adjusted for gestational weight gain, gestational age at birth, maternal age and intervention group
$ model additionally adjusted for gestational weight gain, maternal age and intervention group
§ MVPA and sedentary behaviour measured at 15 weeks gestation was used in the models for gestational weight gain, birth weight and birth percentile,
MVPA measured at 26 weeks postpartum was used for weight retention.
With regards to weight retention, we might have observed a yo-yo effect, frequently seen in women losing weight [13]. For body weight the existence of a fixed set point for energy homeostasis was postulated recently [14,15]. Whether in humans this set point can be modulated by weight change preceding pregnancy or by gestational weight gain is not known, nor the effect of preconceptional weight change on the development of a set point in the offspring. However, according to the set point hypothesis, it is not unlikely that after giving birth, women regained the weight they had lost when preparing for pregnancy and returned to their “set point”.
With regards to the difference in birth weight, an explanation for our findings is not straightforward. To our knowledge, the EDEN motherchild cohort study is the only other study that looked at weight changes before pregnancy in relation to pregnancy outcomes. In the EDEN study, in normal weight women body weight loss within 9 years preceding pregnancy was associated with poor fetal growth and small for gestational age babies [7]. This is not in line with our results. This might be due to differences in how long before conception the weight loss took place and the difference in what assumed to be a considerable body weight change. In the EDEN study only maternal body weight change in relation to pregnancy outcomes was studied and not in relation to gestational weight gain or weight retention.
Another comparison could be made with women undergoing bariatric surgery before pregnancy, since they also loose considerable body weight. However, results from bariatric surgery are not comparable to our participants, because of differences with regards to prepregnancy BMI.
In our study BMI at 15 weeks and gestational weight gain were not related to birth weight. Although perhaps unexpected, this finding is in line with recently published data on birth weight and maternal body composition [16]. Birth weight was positively correlated with maternal fat-free mass and not with adiposity [16]. The authors suggested that interventions intended to reduce fat mass during pregnancy may not prevent large-for-gestational-age neonates. Although we could not distinguish between fat-free or fat mass, unfortunately, it may explain the lack of association of maternal BMI and gestational weight gain with birth weight in our study.
Whether prepregnancy BMI modifies the effect of weight changes before pregnancy is worth considering, because this might have implications for defining high-risk subgroups for preventive actions, and to estimate the magnitude of weight dynamics before pregnancy on weight development for the mother and her offspring. Unfortunately, in this study, the groups of women losing or gaining weight are too small to study effect modification of prepregnancy BMI. Because of the small numbers, and because our findings might be due to chance, it is important to confirm our findings in future studies, preferably in larger populations of women with a wider variety of prepregnancy BMI. This would also allow assessing the influence of prepregnancy BMI on the findings.
Lifestyle factors (physical activity, sedentary behaviour, breastfeeding and sleep duration) did not influence gestational weight gain. This is in line with the recent review on lifestyle interventions and body weight gain and results from another cohort study [6,17]. Breastfeeding and duration of breastfeeding, physical activity and sedentary behaviour did not influence weight retention in our study. These findings are also in line with other studies [17,18]. Sleeping has previously been shown to be associated with less weight retention postpartum [17,19]. In this study, however, we find more weight retention in women who sleep more on average in the year postpartum. Smokers gave birth to significantly lighter babies and had less weight retention. This is in line with literature [20,21]. A considerable part of smokers stopped smoking before getting pregnant, especially in the group of women who lost weight before pregnancy. It’s tempting to speculate that the women, who stopped smoking, adopted a healthier lifestyle in general preparing themselves to get pregnant, and therefore also lost weight in that period. However there are no other data yet to support this speculation.
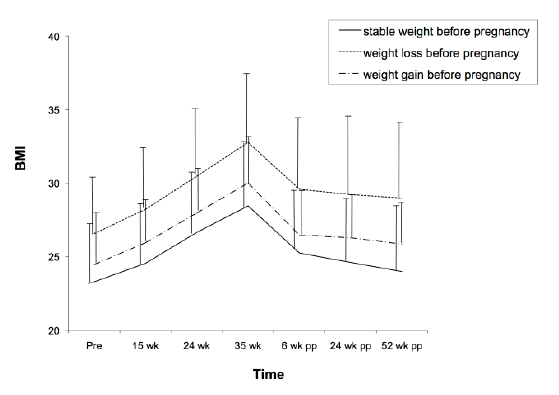
Figure 1: Development of maternal weight before, during and after pregnancy in groups of women with stable weight, weight loss or weight gain before pregnancy. Prepregnancy body mass index (BMI) is based on self reported weight and objectively measured height. At other time points, BMI is based on objectively measured height and weight. Data are presented as means and standard deviation (error bar).
Most studies reporting on lifestyle interventions for managing maternal weight and improving pregnancy outcomes restrict themselves to pregnancy. Our study shows that one needs to extend the study period beyond pregnancy in relation to pregnancy outcomes and postpartum weight changes.
Some limitations of this study need to be considered. One limitation is the participation of relatively healthy, well-educated women with an average BMI of 23.7 kg/m2 and likely to be more interested in a healthy lifestyle since they participated in a lifestyle trial. This might have led to recruitment and selection bias. Our results can therefore not be extrapolated to other, heavier groups of pregnant women. Another limitation, as mentioned earlier, was the small number of women losing or gaining weight before pregnancy. In addition, we had no objective data on dietary intake and no objective data on prepregnancy body weight (change). Though misclassification of prepregnancy BMI is quite common, particularly among women with a high BMI [22], in our study self-reporting by pregnant women seemed to be quite reliable. Comparing the difference between self-reported prepregnancy body weight and the objective body weight at 15 weeks pregnancy showed a consistent pattern and was not different between BMI groups (data not shown). Furthermore, assessing MVPA with accelerometry in late pregnancy might be less valid compared to outside and earlier pregnancy [23].
We found that maternal body weight change preceding pregnancy influenced birth weight and maternal weight retention. The percentage of maternal body weight change and the period in which the maternal body weight change takes place might be predictive for the effect on birth weight and body weight retention. These findings should be confirmed in a larger sample of women who lose or gain weight before pregnancy.
The authors declare no conflict of interest.
CvdW, MvP, HD had a role in designing and planning of the study, MvP and CvdW performed the analyses and CvdW drafted the article. HD and WvM critically appraised the study results and article. All authors were involved in writing the paper and had a final approval the submitted and published versions.
This study was financially supported by a grant from the Netherlands Organization for Health Research and Development (ZonMw, grant 4010.0017).
Download Provisional PDF Here
Article Type: Research article
Citation: van der Wijden CL, Delemarre-van der Waal HA, van Mechelen W, van Poppel MN (2015) A Longitudinal Cohort Study on Maternal Weight Change Preceding Pregnancy in Relation to Gestational Weight Gain, Pregnancy Outcome and Weight Retention. Obes Open Access 1(3): doi http://dx.doi. org/10.16966/2380-5528.110
Copyright: © 2015 van der Wijden CL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access