
Figure 1: Map of Abakaliki Lead-Zinc Mining Area showing Lead-Zinc Lodes and Sample Points


Aloh Obianuju G1 Aloh Henry E2* Obasi Nwogo Ajuka3 Chukwu Kelvin1
1Department of Meteorology/Geography, Faculty of Environmental Sciences, Enugu State University of Science and Technology, Enugu, Nigeria*Corresponding author: Aloh Henry E, Health Economics and Policy Research Unit, Department of Health Services, Federal University Ndufu-Alike Ikwo, Ebonyi State, Nigeria, E-mail: henryealoh@gmail.com; hewiedconsultnl@gmail.com
Mining industry is an economically viable enterprise in developing countries like Nigeria. This has led to increased mechanized mining and illegal artisanal mining activities. In Ameri, Abakaliki mining area of Ebonyi State, Southeast Nigeria, illegal artisanal mining activities are carried out amidst farming. This study investigated the level of heavy metals pollution from mining activities in Ameri farmland soils and the extent of inadvertent uptake by the cultivated plants. The soil and plant parts (root and stem) were obtained from farmlands within the vicinity of Ameri mining area and from control site, 30 km away where there is no mining activity. The soil and plant samples were analyzed using standard analytical protocols. The results showed that the mean concentration of heavy metals (Pb, Zn, Fe, Cd, Cu, Mn, Ni, Cr and Co) in the soil and plant parts (Discorea rotundata, Ipomea batatas, Telfeleria occidentalis and Chromolaena odorata) were significantly higher (P<0.05) in Ameri sites when compared to the control site. The results also showed that the translocation factors (TF) of the plants species varied for the various heavy metals with D. rotundata having TF less than unity (<1) and I. batatas having TF greater than unity (>1) for all the metals studied. These results revealed that the plant species studied were good phyto-extractors for the heavy metals and thus could constitute public health risk of heavy metal toxicity for populations using them for dietary and medicinal purposes.
Agricultural Soil Contamination; Heavy Metals; Artisanal Mining; Translocation Factor; Phyto-extraction
The concentration of metals in soil depends on numerous environmental factors, both natural and manmade [1]. Heavy metals usually occur in very small amounts in soils and aquatic systems [2]. However, human and other anthropogenic activities such as mining refuse dumping, agriculture; among others add these elements in large quantities exceeding their threshold limits to the environment.
In Ebonyi State, South-East Nigeria, mining and agricultural practices are of economic importance and the two often coexist in areas with mine deposits. In Ameri mining area, the principle minerals of interest are mainly lead and zinc. Mining, either artisanal or mechanized, is an economically viable enterprise. The vicinity of most mining sites in Ebonyi state is often used as arable farmlands for cultivating edible food and medicinal plants [1]. Mining activities significantly contribute to the increase in lead and other potentially harmful elements in surrounding environment and this has been reported to constitute health risks for habitual edible food and medicinal plants in these polluted areas [3,4].
The extensive use of most of the mine sites as arable lands for agriculture is on the increase in Southeast Nigeria due to population increase, economic hardship and over dependences on agriculture for livelihood. This work was designed to evaluate and provide credible data that will help decision makers in the region enunciate evidence based policy on mining, agriculture and environmental sustainability. Previous studies on the impacts of lead-zinc mining in Abakaliki lead-zinc mining area have focused mainly on water, soil and plants metal loads [4,5]. However, little or no information on the accumulation potentials of edible food and medicinal plants on farmlands within the vicinity of the mines sites are available. This work therefore is designed to fill this scientific gap by providing baseline data on the heavy metal load in agricultural soil and food and medicinal plants within the vicinity of mine sites.
Ameri is a community in Ikwo Local Government Area of Ebonyi State, southeast Nigeria, located on latitudes 06° 151 N and 06° 201 N and Longitude 008° 051 and 008° 101 N (Figure 1).

Figure 1: Map of Abakaliki Lead-Zinc Mining Area showing Lead-Zinc Lodes and Sample Points
Geologically, the Ameri lead-zinc deposits forms part of the Abakaliki lead-zinc field and this field occurs within a substantial part of the largest cretaceous sedimentary basins in Nigeria known as the Benue trough [4].
Random techniques and judgmental technique described by Cretin and Johnson [6] was used in obtaining media samples from both the study and control sites.
The coordinates for soil sampling points were obtained using a Global Positioning System (GPS)-Garmin 724 model. The study location was divided into four sampling points and then the control site, making it a total of five sampling points. All samples were collected between 100- 600 m interval to ensure even spread and in triplicate to form composite sample. Soil samples were obtained from the top soil (0-30 cm) and sub soil (30-45 cm). The soil samples were obtained from rhizosphere of the plants used for the study. The soil samples were obtained in wet and dry season to represent yearly seasonal variation in the region under study. The sampled soils were air dried, then sundried, grounded into powder and then sieved using a 2 mm mesh. They were then stored separately in an air-tight well labeled polythene bags until analysis.
The selected plant species (Discorea rotundata, Ipomea batatas, Telfeleria occidentalis and Chromolaena odorata) were obtained from marked points in the mapped out sites in the study area (farmlands in Ameri and control sites) in triplicates and mixed to form composite sample for each. They were harvested in wet and dry seasons to represent yearly seasonal variation in the region under study. The plants were sorted out and the roots separated from the shoot (stem and leaves). All collected samples were washed with de-ionized water. Three steps washing sequence were applied until the samples were free from visible soil particles. The samples were air dried and then oven dried at about 80° C for 24 hrs, to remove all moisture. Dried samples were pulverized using laboratory blender, stored in a tight screw capped container (closed system), labeled appropriately. Later, the powered samples were wet digested using aqua regia in the ratio of 1:2 (HNO3 : HCl). The digested extract was aspirated into the Atomic Absorption Spectrophotometer (spectra AA 220 FS) after inserting appropriate hollow cathode lamp (AOAC 2005).
Elemental analysis of the soil and plants were carried out using the atomic absorption spectrophotometer (AAS) model 210 VGP, Buck Scientific incorporated USA as described in AOAC (2005). Heavy metals concentration, comprising of Pb, Zn, Fe, Cu, Cd, Mn, Ni, Co and Cr, were analyzed using Atomic Absorption spectrophotometer (spectra AA 220 FS model).
The translocation factor (TF) defined as the ratio of heavy metals in plants shoot to that of root was calculated using the procedure described by Cui et al. [7]. Translocation factor (T/F)=[metals]Shoot/[metals]Root.
The experimental results were expressed as mean ± standard deviation (SD) of triplicate determinations. One way Analysis of variance (ANOVA) for all the measured variables was performed by SPSS ver. 9.2 (Inc., Chicago, USA) software and significant differences were shown at P<0.05 using Duncan Multiple Range Test according to Zamani et al. [8].
a) The results of the heavy metal content of the top soil and sub soil from the study area in wet and dry seasons are shown in tables 1 and 2 respectively. The results showed that Pb, Zn, Fe, Cu, Cd, Mn, Ni, Co and Cr were present at varying levels and that the levels were significantly higher (P<0.05) in soils obtained from Ameri farmlands when compared to values obtained from the control site for both seasons. The results also showed significant higher (P<0.05) metal concentrations in top soil when compared to the sub soil for both seasons. The results (Tables 1 and 2) showed that the concentrations of the heavy metals studied in wet season exceeded the permissible limit set by USEPA (2010) with the exception Ni and Cu while for dry season, only Cd, Mn and Cr were above the permissible limiti set by USEPA (2010) for top and sub soils respectively. The results showed that soils from control site had all their values within the permissible limit set by USEPA (2010) for both seasons.
| Heavy Metals | USEPA (2010) | AMERI | CONTROL | AMERI | CONTROL |
| mg/kg | Top soil | Top soil | Sub soil | Sub soil | |
| Pb | 300 | 1154.88b ± 0.96 | 30.03a ± 0.34 | 1154.05c ± 0.26 | 29.95a ± 0.15 |
| Zn | 200 | 1197.23c ± 0.74 | 8.88a ± 0.68 | 1193.13c ± 1.11 | 8.77a ±0.45 |
| Fe | 1000 | 77830.99c ± 5.12 | 70.17a ± 0.17 | 77545.80d ±1.58 | 70.12a ± 0.31 |
| Cu | 250 | 35.54b ± 0.18 | 18.02a ± 0.35 | 33.99c ± 0.33 | 12.65a ± 0.64 |
| Cd | 3.0 | 5.37b ± 0.55 | 0.39a ± 0.31 | 5.04c ± 0.23 | 0.38a ± 0.33 |
| Mn | 80 | 1238.11c ±1.37 | 5.96a ± 0.52 | 1238.28c ± 2.13 | 6.91a ± 0.75 |
| Ni | 150 | 72.28d ± 0.24 | 2.60a ± 0.51 | 72.17d ± 0.93 | 2.50a ± 0.20 |
| Co | NA | 33.07b ± 0.38 | 0.8.03a ± 0.29 | 32.44b ± 0.45 | 1.02a ± 0.81 |
| Cr | 750 | 1127.57c ± 0.47 | 1.40a ± 0.26 | 1121.80c ± 0.85 | 1.58a ± 0.33 |
Table 1: Heavy Metal Contents (mg/kg) of Soil Samples during Wet Season
| Heavy Metals | USEPA | AMERI | CONTROL | AMERI | CONTROL |
| (2010) mg/kg | Top soil | Top soil | Sub soil | Sub soil | |
| Pb | 300 | 309.60b ± 0.97 | 19.13a ± 0.62 | 209.58a ± 0.84 | 118.89a ± 0.55 |
| Zn | 200 | 155.05b ± 0.43 | 44.85a ± 0.28 | 155.01b ±1.13 | 44.74a ± 0.36 |
| Fe | 1000 | 790.02d ± 1.14 | 408.12a ± 0.96 | 758.96c ±1.07 | 477.5a ± 1.16 |
| Cu | 250 | 28.30b ± 0.45 | 6.90a ± 0.34 | 28.22b ± 0.32 | 7.02a ± 0.35 |
| Cd | 3.0 | 4.90c ± 0.42 | 1.54a ± 0.25 | 4.81c ± 0.62 | 1.52a ± 0.18 |
| Mn | 80 | 294.74d ± 0.44 | 3.03a ± 0.95 | 294.66d ±0.39 | 4.10a ± 0.55 |
| Ni | 150 | 33.02c ± 0.22 | 1.08a± 0.26 | 32.89c ± 0.36 | 1.05a ± 0.43 |
| Co | NA | 48.01d ± 0.11 | 1.80a ± 0.66 | 48.00d ± 0.72 | 1.71a ± 0.38 |
| Cr | 750 | 1112.70c ± 0.37 | 1.34a± 0.82 | 1112.60c ±0.43 | 1.13a ± 0.42 |
Table 2: Heavy Metal Contents (mg/kg) of Soil Samples during Dry Season
Values are mean of three (n=3) replicates ± Standard Deviation. Values with different alphabets for the same metal are statistical different at P<0.05 using
Duncan Multiple Range Test
b) The results of the heavy metal load in the shoots and roots of Discorea rotundata, Ipomea batatas, Telfeleria occidentalis and Chromolaena odorata are shown in tables 3 and 4 for Ameri site and control site respectively. The results showed that the different parts of the plant species accumulated heavy metals at varying levels and that the level of accumulation of these heavy metals were generally higher in plants species at Ameri than those at control sites. There were higher metal content in the roots of Discorea rotundata, Telfeleria occidentalis and Chromolaen odorata than in their shoots for all the metals except for Fe content in Discorea rotundata. The results also showed that although the plants both at Ameri and control site accumulated varying levels of these heavy metals, the values were all below the USEPA (2010) maximum permissible set limits for edible food and medicinal plants.
| Heavy Metals | Discorearotundata | Ipomea batatas | Telfeleriaoccidentalis | Chromolaenaodorata | |||||
| USEPA (1986) mg/kg-1 |
Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | |
| Pb | 5.0 | 0.92 ± 0.44 | 0.98 ± 0.97 | 0.84 ± 0.05 | 1.21 ± 0.12 | 0.08 ± 0.08 | 1.10 ± 0.02 | 0.19 ± 0.04 | 0.19 ± 0.02 |
| Zn | 100.0 | 0.64 ± 0.11 | 0.86 ± 0.22 | 0.86 ± 0.22 | 1.42 ± 0.02 | 0.14 ± 0.02 | 1.58 ± 0.04 | 2.05 ± 0.05 | 0.145 ± 0.03 |
| Fe | 250 | 23.980 ± 0.84 | 14.125 ± 1.50 | 31.04 ± 0.06 | 44.76 ± 0.17 | 6.92 ± 0.12 | 118.75 ± 0.11 | 58.32 ± 0.17 | 48.44 ± 0.12 |
| Cu | 40.0 | 0.42 ± 0.43 | 0.74 ± 0.11 | 0.88 ± 0.02 | 1.31 ± 0.11 | 0.99 ± 0.11 | 1.10 ± 0.02 | 0.92 ± 0.05 | 0.76 ± 0.04 |
| Cd | 0.10 | 0.041 ± 0.52 | 0.132 ± 0.34 | 0.98 ± 0.02 | 0.31 ± 0.03 | 0.98 ± 0.02 | 0.99 ± 0.05 | 0.52 ± 0.04 | 0.95 ± 0.05 |
| Mn | 1.0 | 0.125 ± 0.38 | 0.200 ± 0.52 | 0.24 ± 0.03 | 0.31 ± 0.01 | 0.66 ± 0.04 | 0.50 ± 0.04 | 0.26 ± 0.07 | 0.28 ± 0.02 |
| Ni | 10-100 | 0.085 ± 0.21 | 0.078 ± 0.35 | 0.08 ± 0.01 | 0.11 ± 0.01 | 0.06 ± 0.01 | 0.09 ± 0.01 | 0.10 ± 0.02 | 0.07 ± 0.01 |
| Cr | 2.0 | 0.031 ± 0.33 | 0.102 ± 0.17 | 0.57 ± 0.02 | 0.82 ± 0.02 | 0.45 ± 0.02 | 0.64 ± 0.02 | 0.18 ± 0.03 | 0.34 ± 0.03 |
Table 3: Heavy Metal Contents (mg/kg) of Plant Parts in the Ameri Site
Values are mean of three (n=3) replicates ± Standard Deviation. Source: Author̕ s Results
| Heavy Metals | Discorearotundata | Ipomea batatas | Telfeleriaoccidentalis | Chromolaenaodoranta | |||||
| USEPA (1986) mg/kg- |
Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | |
| Pb | 5.0 | 0.01 ± 0.03 | 0.03 ± 0.02 | 0.03 ± 0.02 | 0.09 ± 0.03 | 0.10 ± 0.01 | 0.09 ± 0.01 | 0.02 ± 0.01 | 0.01 ± 0.01 |
| Zn | 100.0 | 0.44 ± 0.02 | 0.92 ± 0.05 | 0.80 ± 0.05 | 0.89 ± 0.09 | 0.18 ± 0.09 | 0.13 ± 0.01 | 0.32 ± 0.01 | 0.42 ± 0.02 |
| Fe | 250 | 3.51 ± 0.03 | 3.66 ± 0.05 | 2.18 ± 0.03 | 2.99 ± 0.09 | 12.18 ± 0.03 | 13.17 ± 0.04 | 2.08 ± 0.06 | 10.20 ± 0.10 |
| Cu | 40.0 | 0.03 ± 0.05 | 0.06 ± 0.02 | 0.26 ± 0.02 | 0.02 ± 0.01 | 0.09 ± 0.05 | 0.05 ± 0.02 | 0.13 ± 0.03 | 0.12 ± 0.03 |
| Cd | 0.10 | 0.28 ± 0.02 | 0.62 ± 0.02 | 0.15 ± 0.01 | 0.26 ± 0.02 | 0.70 ± 0.02 | 0.97 ± 0.07 | 0.99 ± 0 .03 | 0.81 ± 0.03 |
| Mn | 1.0 | 0.21 ± 0.05 | 0.45 ± 0.03 | 0.23 ± 0.03 | 0.44 ± 0.02 | 0.42 ± 0.02 | 0.27 ± 0.02 | 0.47 ± 0.05 | 0.41 ± 0.05 |
| Ni | 10-100 | 0.05 ± 0.03 | 0.07 ± 0.02 | 0.04 ± 0.02 | 0.05 ± 0.01 | 0.01 ± 0.01 | 0.02 ± 0.01 | 0.06 ± 0.01 | 0.08 ± 0.02 |
| Cr | 2.0 | 0.08 ± 0.02 | 0.09 ± 0.01 | 0.01 ± 0.01 | 0.05 ± 0.01 | 0.06 ± 0.01 | 0.11 ± 0.01 | 0.04 ± 0.01 | 0.07 ± 0.01 |
Table 4: Heavy Metals Contents (mg/kg) of Plants Parts in the Control Sites
c) Translocation Factor (TF): The results of the translocation factor (Phytoextraction quotient) of the studied plants for all the metals studied are shown in figure 2. The results showed that the translocation factor for Discorea rotundata is less than unity (<1) for all the heavy metals studied while that of Ipomea batatas is greater than unity (>1) for all the metals studied. The results (Figure 2) showed similar trends in the TF of Telfeleria occidentalis and Chromoleana odorata where they had TF values greater than unity (>1) for Zn, Fe, Cu, Ni and Cr and values less than unity (<1) for Pb, Cd and Mn.
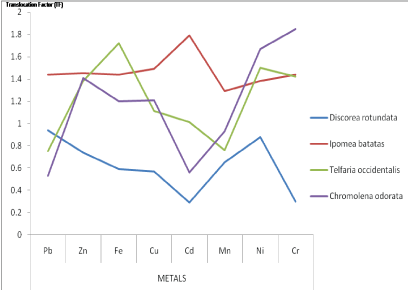
Figure 2: Translocation Factor (TF) of the plants for all the metals in the studied area
Trace amount of some heavy metals are required at optimum amount by living organisms for the maintenance of good health. However, an excess amount of these metals can be detrimental to the organisms [9]. The study (Tables 1 and 2) showed that the heavy metals concentrations in the studied sites were in most cases above the USEPA (2010) permissible limits for agricultural lands. Similar results have been reported for dumping sites and heavy metal contaminated sites in different parts of Nigeria [10,11]. High concentrations of heavy metals in the soil have been shown to influence the amounts absorbed by plants. The metals when absorbed at levels above threshold limits by plants can replace essential metals in pigments or enzymes, thus disrupting their function [12]. The study revealed that the metal concentrations in the top soil were slightly higher than the values in the sub soil (Tables 1 and 2). This might be attributed to the deposition of heavy metals from mining dusts trails during artisanal mining activities. Similar works have shown the resultant effect of dumping of tailings, packing of ores and wastes on agricultural soil surfaces [13,8].
Studies on soil-plant transfer of trace elements have shown that accumulation of metals vary greatly among different plants and is primarily dependent on the plant species, its inherent controls and the soil quality [14]. The study (Tables 3 and 4) showed that the different plants studied accumulated heavy metals in their shoots and roots in varying degrees but below the USEPA (2010) permissible set limit. The accumulation of high level of these metals may cause stunted growth, chlorosis, blackening of the roots and yellowing of leaves of the plants, thus decreasing the expected plants yield in the studied area. The ability of some of the plant to survive may be attributed to the development of tolerance mechanisms by the plants since the level of accumulation were still below the set limits for some agricultural lands [15,16,7].
The elevated range of Fe in the various parts of plant species obtained in this study could be attributed to the high availability of Fe at the sites and the general abundance of the metal in the earth’s crust [17]. Generally, the study revealed high level of most of the studied metals in the plants and implicated that dependence on the plants for food and medicinal purposes might be deleterious to health [9]. However, some of the metals studied such as Cd, Mn, Cr and Ni (Tables 3 and 4) were low and may not be enough to cause phytotoxicity according to Vecera et al. [18,19].
TF values greater than one (>1) had been used to evaluate the potential of plant species for phytoextraction and phytostabilization. The study (Figure 2) indicated that the TF values vary from one plant species to another and from one heavy metal to another. High root to shoot translocation of these metals indicated that these plants might have vital characteristics to be used in phytoextraction of the metals [15,20,21]. This study has shown that I. batatas, T. occidentalis and C. odorata whose shoot is used for edible and medicinal purposes might constitute public health risk since they have TF>1. Plant species with high TF values are considered suitable for phytoextraction because they generally translocate heavy metals to easily harvestable parts such as stems and leaves (shoot).
This study showed that the sampled plant species at Ameri with high TF values could have enormous potential for phytoextraction of heavy metals. It also indicated that the soil at Ameri and its associated environmental factors could predispose these edible staple plants to become phytotoxic and as such unsafe for human consumption. The implication is that the cultivation of food and medicinal plants in agricultural lands within the vicinity of mining sites should be avoided to avert the multiple effects of heavy metals toxicity. Also, the study showed that plants such as I.batatas in the study area could be used for phytoextraction of the metals studied. Such plants with high phytoextraction potentials could be used to remediate the polluted soils. The TF of D. rotundata is lower than unity (Figure 2) and implies that most of the adsorbed metals are stored in the root. Since the edible part of D. rotundata is the root, it also means that its consumption is unsafe as it might constitute multiple effects of heavy metal toxicity health risks.
The results of the study showed that Pb, Zn, Fe, Cd and Mn exceeded the tolerable threshold permissible USEPA limit in soil and indicated that farmlands in Ameri are highly polluted. This implies that the farmlands are not good for cultivation of edible food and medicinal plants except after remediation options. Although, the plants studied absorbed Pb, Cd, Zn, Fe, Ni and Cr lower than the critical threshold of 1000 mg/kg-1, they still posses the potential for phytoextraction. These results revealed that the plant species studied were good phyto-extractors for the heavy metals and thus could constitute public health risk of heavy metal toxicity for populations using them for dietary and medicinal purposes. The results of this study call for concern in terms of ecological, environmental and public health protection.
Download Provisional PDF Here
Article Type: Research Article
Citation: Aloh GA, Aloh HE, Obasi AN, Chukwu K (2017) Evidence of Heavy Metal Contamination of Agricultural Soil in Ameri, Abakaliki Lead-Zinc Mining Area, Ebonyi State Southeast Nigeria: An Indication for Phytoremediation. Nutr Food Technol Open Access 3(3): doi http://dx.doi.org/10.16966/2470-6086.145
Copyright: © 2017 Aloh GA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access