
Full Text
Richtier G Cruz1* Samarina GF Pereira2 Emiliane A Araújo3 Milene T Dores2
1Escola Superior de Agricultura “Luiz de Queiroz”, Universidade de São Paulo, Av. Pádua Dias 11, 13418-900- Piracicaba- São Paulo, Brazil2Universidade Federal de Viçosa, Rio Paranaíba, Highway MG-230 – Km 7, 38810-000 Rio Paranaíba – Minas Gerais, Brazil
3Universidade Federal do Triângulo Mineiro, Av. Randolfo Borges Júnior, 1250 38064-200-Uberaba- Minas Gerais, Brazil
*Corresponding author: Richtier G CRUZ, Escola Superior de Agricultura “Luiz de Queiroz”, Universidade de São Paulo, Av. Pádua Dias 11, 13418-900- Piracicaba-São Paulo-Brazil, Tel: 55 34 3331-3000; E-mail: richtier@gmail.com
Abstract
This work aimed to evaluate the viability of Lactobacillus acidophilus immobilized in calcium alginate sphere under different stress conditions. For the preparation of the sphere was dripped into a solution of 5 % (w/v) sodium alginate bacterial suspension of Lactobacillus acidophilus, previously activated. To simulate the acid stress, free and immobilized bacteria were exposed to pH values 2.5, 3.5 and 7.0, for 4 hours, incubated at 37°C. The same was done for salt stress, at concentrations of 0.85 %, 4 % and 6 %. For freezing stress, the bacteria free and immobilized was stored at -20°C for three months. In all simulations of stress conditions (pH, salts and freezing) changes in count for the immobilized bacteria were lower, with the greatest protection occurred when Lactobacillus acidophilus immobilized was subjected to high osmotic pressure in a concentration of 6 % NaCl and freezing. We can conclude calcium alginate acted as a protective agent in adverse conditions, providing protection to the microorganism. Therefore, the immobilization technique in calcium alginate has good prospects for the development of new probiotic foods.
Keywords
Community service; Attitude; Perception; Dental interns
Introduction
Several everyday aspects such as, urbanization, industrialization and globalization, may adversely affect the quality of modern life, and, with that, the intake of functional foods that improve health is increasingly sought by consumers [1]. Due to this interest, the development of healthy and attractive products that meet this demand has attracted the attention of both industry and the scientific community. This new demand for products has become a major challenge, since they must meet the sensory and technological aspects as well as ensure safety and welfare of the consumer [2].
Functional foods are those that, in addition to providing basic nutrition, bring other health benefits. They have the potential to promote health through mechanisms not provided by conventional nutrition, a role that should be emphasized that this effect is limited to prevention rather than curing disease [3]. The main ingredients responsible for the functionality of these products are constituted by dietary fiber, fish oils, plant sterols, minerals, vitamins, probiotics and prebiotics [4,5].
The internationally accepted definition is that probiotics are live microorganisms which when administered in adequate quantities confer health benefits to the host [6]. Besides these features, probiotics must also resist the pH of the gastric juice, tolerate the pancreatic and intestinal secretions without carrying antibiotic resistance genes in the genome, possess antimutagenic and anticarcinogenic properties, retain viable in a long time and be safe [7-11].
Some lactic acid bacteria besides promoting desirable technological effects in products to which they have been added are able to exert functional effects for those who ingest them, being therefore called probiotic. A requirement for probiotic microorganism is that it must resist the digestive enzymes of the gastrointestinal tract, multiply and colonize the intestine, at least temporarily, by adhering to the intestinal epithelium [12,13].
An extensively used lactic acid bacteria, Lactobacillus acidophilus, when compared to other species of the genus Lactobacillus, is one of the most recommended as probiotic, because of its high capacity for adhesion to the intestinal epithelium and the benefits to the host, as regulation of the gastrointestinal tract [14]. This is the reason it has been one of the most used and studied probiotics in recent years [15].
Several factors undertake the growth and viability of probiotic bacteria in food products. Among them stands pH, the acidity increase during storage, the storage temperature, the presence of preservatives and other microorganisms, the oxygen concentration contained in the product, its permeability through the packaging and the availability of growth factors [16]. An alternative to ensure a greater survival and viability of these organisms in adverse conditions during the shelf life of a food is its immobilization [17].
Cell immobilization decreases cell death and increase its viability, which may be affected by the storage, product processing, or during the actual food intake, since the immobilized cells have a greater resistance to the action of biological fluids related to the digestive process. Furthermore, immobilization provides a lower interaction of microorganisms and food constituents of reducing susceptibility to infection [18]. This technique consist in lodged inside or on the surface of an immobilizing agent enzymes or cells, the calcium alginate gel and K-carrageenan are the most commonly used matrix [19,20].
Alginates belong to a family of heterogeneous polymers with a wide range of chemical compositions, molecular size and functional properties. Chemically, they are unbranched polysaccharides composed of waste 1,4-linked β -D-mannuronic acid and α-L-guluronic acid monomers in varying proportions [21]. Gel properties can be summarized in its porosity and mechanical strength necessary to the process. Porosity is directly linked to the ability of the gel to protect the cells and modify the effective diffusivity of the substrate [22].
In this context, the present work aimed to study the viability of Lactobacillus acidophilus immobilized in calcium alginate beads under different stress conditions.
Material and Methods
Immobilization of Lactobacillus acidophilus
In preparing the alginate sphere was dripped, with the aid of a pipette in a solution of 5% (w / v) sodium alginate (Gastronomy lab), the bacterial suspension of Lactobacillus acidophilus, previously activated in reconstituted skimmed milk (LDR 10%) with 6.67% of skimmed milk powder to increase the viscosity (the same amount of milk powder was also added in bacteria not immobilized). The sphere remained at rest for 30 minutes so that the reaction of formation of calcium alginate occurred.
Stress condition -pH
To evaluate the pH stress condition, 1 ml of the bacterial suspension of Lactobacillus acidophilus was added to 9 ml of physiological saline (0.85% NaCl) containing lactic acid at three pH values, 2.5, 3.5 and 7.0, following the methodology described by Araújo [23]. The mixture was incubated at 37°C for 4 hours for evaluation of the stress applied. During this period, four temporal cells samples were obtained every our hour by centrifugation (Sorvall centrifuge) at 6000 g for 6 minutes for survival analysis for viable cell count. To this purpose, the cell pellet was resuspended in 9 mL of saline (0.85% NaCl) and then we performed serial dilutions in were made saline (0.85% NaCl) to countsurvivor cells on MRS agar (Merck) after incubation under anaerobic conditions at 37°C for 48 hours. Plates containing between 25-250 colonies were selected and we counted colonyforming units (CFU.mL-1) counted and the results expressed as log10. The same procedures for evaluating the stress condition of acid were made to Lactobacillus acidophilus cells immobilized in calcium alginate.
To quantify the amount of sphere which corresponds to 1 ml of bacterial suspension of Lactobacillus acidophilus, we dropped 1 ml of the cell suspension in a solution of 5% (w / v) sodium alginate spheres were at rest, and then were weighed on an analytical balance. We repeated this procedure 25 times. The average weight obtained was 1.5 g corresponding to 1 mL.
Stress Condition-NaCl
For salt stress condition, 1 ml of the bacterial suspension of Lactobacillus acidophilus was added to 9 ml of physiological saline solution with varying NaCl concentrations (0.85, 4 and 6%). The mixture was incubated at 37°C for 4 hours for evaluation of the stress applied. Each one hour the cells were collected for counting of viable cells, to this the same procedures of the stress condition with acid were made. This same procedure was done for to Lactobacillus acidophilus immobilized in calcium alginate.
Stress condition-freezing
In temperature stress condition, 1 mL of the bacterial suspension of Lactobacillus acidophilus was added to 9 mL of physiological saline (0.85% NaCl). The mixture was stored at -20°C (freezing) for 3 months. During this period, the bacterial count was determined before freezing, after freezing and 30 in 30 days during the 90 days of storage as previously performed [24]. For each time analyzed the cells was collected for counting of viable cells, making the same procedures of the stress condition with acid. This same procedure was done for to Lactobacillus acidophilus immobilized in calcium alginate.
Statistics
The experiment was conducted in triplicate with three replications. The results were analyzed using descriptive statistics.
Results and Discussion
Stress condition-pH
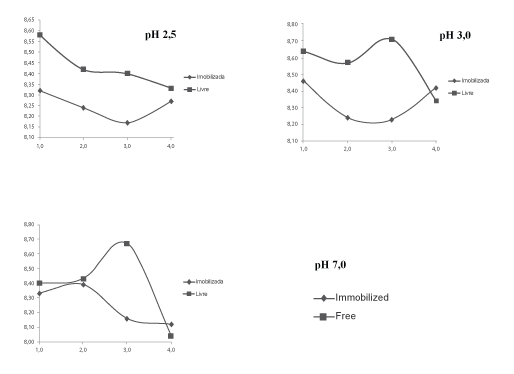
The viable cell count of Lactobacillus acidophilus free and immobilized subjected to stress condition, pH of analysis at different times (from 1 to 4 hours) is shown in Tables 1 and 2.
For free bacteria minimum and maximum cell counts ranged from 8.04 to 8.71 Log CFU.mL-1 in different pH values. While for the immobilized bacteria such variation was from 8.12 to 8.46 Log CFU.mL-1.
It is noted that for pH 2.5 the free bacteria tend to have a steeper decay than the immobilized since bacteria as the free bacteria count ranged from 8.58 to 8.33 Log-CFU.mL-1, having 0.25 of difference, and the immobilized ranged from 8.32 to 8.27 Log CFU.mL-1, the difference being 0.05.At pH 3.5 the reduction in bacterial count immobilized was markedly in initial three hours, however, the further reduction of free bacteria counts occurred after the third time analysis. The difference between the initial and final count for bacterium free is 0.30, on the other hand the difference immobilized bacteria is 0.04.At pH 7.0 both free as the immobilized bacteria exhibited similar behavior. However, the reduction in the free bacterial count was 0.36 and immobilized 0.21.
| Time (hours) | ||||
| pH | 1 | 2 | 3 | 4 |
| 7,0 | 8,40 ± 0,36 | 8,43 ± 0,25 | 8,67 ± 0,19 | 8,04 ± 0,26 |
| 3,5 | 8,64 ± 0,06 | 8,57 ± 0,17 | 8,71 ± 0,30 | 8,34 ± 0,21 |
| 2,5 | 8,58 ± 0,24 | 8,42 ± 0,23 | 8,40 ± 0,15 | 8,33 ± 0,17 |
Table 1: Number of viable cells of free Lactobacillus acidophilus (Log CFU. mL-1) subjected to stress pH.
| Time (hours) | ||||
| pH | 1 | 2 | 3 | 4 |
| 7,0 | 8,33 ± 0,29 | 8,39 ± 0,23 | 8,16 ± 0,22 | 8,12 ± 1,87 |
| 3,5 | 8,46 ± 0,22 | 8,24 ± 0,21 | 8,23 ± 0,11 | 8,42 ± 0,23 |
| 2,5 | 8,32 ± 0,18 | 8,24 ± 0,23 | 8,17 ± 0,20 | 8,27 ± 0,25 |
Table 2: Number of viable cells offree Lactobacillus acidophilus (Log CFU. mL-1) subjected to stress pH.
Lactobacillus acidophilus tolerates acid environments in the range of 0.3% to 1.9% of lactic acid and the optimum pH ranging from 5.5 to 6.0.It is resistant to the gastric acidity and bile salts, as the survival in the gastrointestinal tract is estimated between 2% and 5% to reach sufficient concentrations in the colon [25]. This resistance may explain the notable counts to acid stress found in this study. Oliveira [26] also found high counts of Lactobacillus acidophilus microencapsulated, approximately 108 CFU.mL-1, even after 3 hours of incubation at pH 1 and 3.
Yoon et al. [27] studying the behavior of lactic acid bacteria in tomato juice, found that Lactobacillus acidophilus, Lactobacillus plantarum, Lactobacillus casei and Lactobacillus delbrueckii grew rapidly in this drink, reducing the levels of sugars, an increase the concentration of lactic acid. Although the pH of the juice after 72 hours of fermentation was reduced to pH 3.5; the viability of lactic cultures remained above 106 CFU g-1 after 4 weeks of storage at 4°C. Similar results were found in this study in similar conditions, once all counts at pH 3.5 were more than 106 CFU. mL-1 at different times analyzed. This evidence may be related to a greater adaptation of Lactobacillus acidophilus lactic acid, since this is a natural metabolite of its mechanism of energy production.
The trend lines of the behavior of immobilized Lactobacillus acidophilus in free form and in different pH values are shown in Figure 1.
Through the trend lines, we may observe that the immobilization provided greater protection when these were subjected to different pH values, since in all conditions the reduction of counts was lower in bacteria immobilized bacteria, besides having more linear trends of count during the test.
In the study by Krasaekoopt et al. [28] the encapsulation with alginate and chitosan were also effective for enhancing the viability of Lactobacillus acidophilus and Lactobacillus casei in yoghurt when applied at pH 4.7 and placed in refrigerated conditions at 4°C, there was a difference of one cycle log relative to the probiotics free.
Mandal et al. [29] evaluated the tolerance of Lactobacillus casei NCDC 298 encapsulated by the technique of emulsification at different concentrations of sodium alginate and exposed to low pH, high concentrations of bile salts and heating. As this study the authors found that immobilized of probiotic improved the resistance to the conditions tested when compare with free form. Furthermore was observed that the increase in viability increased proportionally to the concentration of sodium alginate used.
Stress condition-NaCl
To the stress at different salt concentrations (0.85%, 4% and 6%) to count viable cells of Lactobacillus acidophilus free and immobilized in the different analysis times (1 to 4 hours) are shown in Tables 3 and 4. Behavior of immobilized Lactobacillus acidophilus and free form in different salt concentrations, are shown in Figure 2 through trend lines.
At a concentration of 6% salts the free bacteria count ranged from 8.60 to 7.40 Log CFU.mL-1, occurred a reduction of more than one log cycle. On the bacteria immobilized the difference was 0.19, ranging from 8.35 to 8.16 Log CFU.mL-1. This indicates that the calcium alginate provided greater protection for the Lactobacillus acidophilus when it was subjected to a stress higher of osmotic pressure. Moreover, the trend lines for the concentration of 6% salt shows that further reduction of free bacteria occurs between time 3 and 4, on the other hand with immobilized bacteria have almost a line constant during the analysis counting.

According to Gomes and Malcata [30] the species of Lactobacillus acidophilus has the particularity of being tolerant to salinity of the medium. As expected, in this paper we note that when the free bacteria were subjected to a higher concentration of NaCl (6%) occur one reduction of 1.2 log cycles, showing that in this concentration of salt the microorganism becomes less resistant to osmotic effects.
In the concentration of 4% we observed that the count of free bacteria decreased from 9.18 to 8.61 log CFU. mL-1 (difference of 0.57). Regarding the immobilized bacteria, the difference of count was 0.54, with an initial count at the end of 8.66 and 8.12 Log CFU. mL-1 respectively. However, the trend lines show that this concentration for the free bacteria the reduction time occurred between 2 and 3 hours whereas for the immobilized bacteria the reduction occurred between the first and third hour, keeping more linear trends of counts when compared with free bacteria.
The count for Lactobacillus acidophilus free at concentration of 0.85% varied from 8.54 to 8.75 Log CFU.mL-1, increasing 0.21. For the immobilized bacteria count ranged from 8.03 to 8.51 Log-1 CFU.mL-1 increasing 0.48. This behavior is expected since the bacterium is in a favorable osmotic pressure to maintaining their metabolic activity [31].
Stress condition-freezing
The behavior of the immobilized and free Lactobacillus acidophilus form during freezing for three months is shown in Figure 3 through trend lines.
The count of free bacteria ranged from 9.04 to 7.50 log UFC.mL-1 (difference of 1.54). For the immobilized bacteria the difference was 0.60, reducing from 8.53 to 7.93 Log UFC.mL-1 respectively. It is observed that the cell count of Lactobacillus acidophilus in immobilized form was higher than the free form when exposed to freezing temperatures, which suggests a better protection of the microorganisms immobilized in calcium alginate.
The same happened in the study by Lorenz [1], where the survival of microencapsulated cells was higher than that of free cells. The counting showed a decrease of 4.3 log cycles on the viability of free cells after 12 weeks of storage at -18 ± 2°C. In the same period, the viability of microencapsulated cells decreased 1.77 log and 1.75 log cycles when produced by emulsification and spray drying respectively.
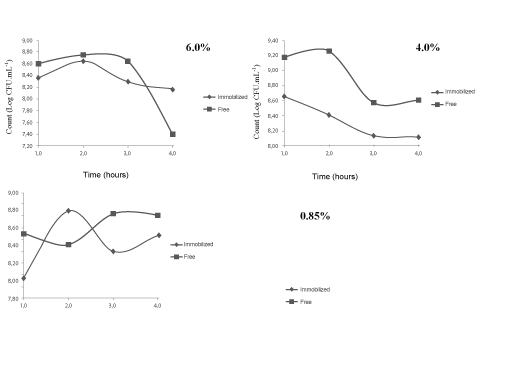
Figure 2: Trend lines of count versus time (hours) under stress condition of salt (0.85, 4.0 and 6.0%) for free and immobilized Lactobacillus acidophilus
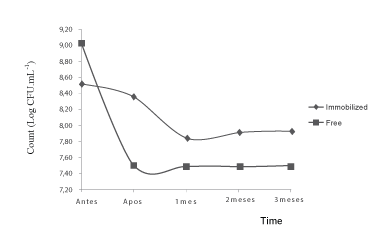
Figure 3: Trend lines of stress condition of freezing for free and immobilized Lactobacillus acidophilus at different times.
| Time (hours) | ||||
| %Salt | 1 | 2 | 3 | 4 |
| 0,85% | 8,03 ± 0,11 | 8,80 ± 0,05 | 8,33 ± 0,28 | 8,51 ± 0,30 |
| 4,0% | 8,66 ± 0,09 | 8,41 ± 0,20 | 8,14 ± 0,14 | 8,12 ± 0,07 |
| 6,0% | 8,35 ± 0,42 | 8,64 ± 0,12 | 8,29 ± 0,31 | 8,16 ± 0,24 |
Table 3: Count (Log CFU.mL-1) of viable cells of Lactobacillus acidophilus free subjected to salt stress
| Time (hours) | ||||
| %Salt | 1 | 2 | 3 | 4 |
| 0,85% | 8,54 ± 0,27 | 8,41 ± 0,58 | 8,76 ± 0,14 | 8,75 ± 0,21 |
| 4,0% | 9,18 ± 0,41 | 9,25 ± 0,05 | 8,58 ± 0,35 | 8,61 ± 0,28 |
| 6,0% | 8,60 ± 0,08 | 8,75 ± 0,56 | 8,63 ± 0,20 | 7,40 ± 0,50 |
Table 4: Count (Log CFU.mL-1) of viable cells of Lactobacillus acidophilus immobilized subjected to salt stress.
According to Desmond et al. and Tsen et al. [32,33], several studies have shown that lower temperatures may ensure a higher survive of microencapsulated cells, however, the mortality of the cells increases with storage time.
However, in this study, we note that the highest fatality occurred shortly after freezing, and over time in three months the counts tended to remain constant. This can be explained by the slow formation of ice crystals occurring almost exclusively during this phase, these crystals can cause denaturation of proteins and enzymes and may also cause lesions in the cell membrane leading to death of the microorganism [30].
Conclusions
In all simulations of stress conditions (pH, salts and freezing) the lethality to Lactobacillus acidophilus immobilized was lower than in the free form, indicating that the calcium alginate acted as a protective agent in unfavorable conditions cell viability. The greater protection occurred when the probiotic immobilized was subjected to high osmotic pressure in a concentration of 6% NaCl and the freeze condition. This shows as an alternative to the development of probiotic foods that have similar stress conditions.
The immobilization technique using calcium alginate with Lactobacillus acidophilus has proved to be an important alternative for the development of new probiotic products having adverse conditions.
Acknowledgments
The authors are grateful to Gustavo Maruyama Mori.
References
- Lorenz JG (2009) Comparação dos métodos de emulsificação e spray drying de Lactobacillusaciophilus (LA-5) e aplicação em sorvete. (Dissertação de mestrado). Universidade Federal de Santa Catarina, Florianópolis.[Ref.]
- Komatsu TR, Buriti FCA, Saad AMI (2008) Inovação, persistência e criatividade superando barreiras no desenvolvimento de alimentos probióticos. Brazilian Journal of Pharmaceutical Sciences 44: 329-337.[Ref.]
- Oliveira MN, Sivieri K, Alegro JHA, Saad SMI (2002) Aspectos tecnológicos de alimentos funcionais contendo probióticos. Revista Brasileira de Ciências Farmacêuticas 38: 1-21.[Ref.]
- Ferreira CLLF (2001) Tecnologia para produtos lácteos funcionais: probióticos. In: O Agronegócio do Leite e os Alimentos Lácteos Funcionais. [Ref.]
- Thamer KG, Penna ALB (2006) Caracterização de bebidas lácteas funcionais fermentadas por probióticos e acrescida de prebiótico. Ciência e Tecnoogia deAlimentos 26: 589-595.[Ref.]
- FAO (2001) Food and Agriculture Organization of the United Nations; World Health Organization. Evaluation of health and nutritional properties of probiotics in food including powder Milk with live lactic acid bacteria.Córdoba, 34p. Retrieved from: ftp:// ftp.fao.org/es/esn/food/ probioreport_ en.pdf.[Ref.]
- Havennar R, Huisint’t Veld MJH (1992) Probiotics: a general view. In: WOOD, B.J.B (Ed.) Lactic acid bacteria in health anddisease. (p.151- 170, 1992) Elsevier Applied Science.[Ref.]
- Salminen S, Ouwehand AC, Isolauri E (1998). Clinical applications of probiotic bacteria. International Dairy Journal 8: 563-572.[Ref.]
- Ouwehand AC, Kirjavainen PV, Shortt C, Salminen S (1999) Probiotics: mechanisms and established effects. International Dairy Journal 9: 43-52.[Ref.]
- Saarela M, Mongensen G, Fondén R, Matto J, Mattila-Sandholm T (2000) Probiotic bacteria: safety, functional and technological properties. Journal of Biotechnology 84: 197-215.[Ref.]
- Holzapfel WH, Schillinger U (2002) Introduction to pre and probiotics. Food Research International 35: 109-116.[Ref.]
- Ziemer CJ, Gibson GR (1998) An overview of probiotics, prebiotics and synbiotics in the functional food concept: perspectives and future strategies. Int Dairy J 8: 473-479.[Ref.]
- Lee YK, Nomoto K, Salminen S, Gorbach SL. Handbook of probiotics (1st edn) New York: Wiley.[Ref.]
- .Alvez CCC (2010) Comportamento da E. coli em queijo Minas Frescal elaborado com a utilização de L. acidophilus e acidificação direta com ácido lático (Dissertação de Mestrado). Universidade Federal Fluminense, Niterói. [Ref.]
- O’Sullivan DJ (2006) Primary Sources of Probiotic Cultures. In: Probiotics in food safety and human health. [Ref.]
- Silva SV (2007) Desenvolvimento de iogurte probiótico com prebiótico (Dissertação de Mestrado) - Universidade Federal de Santa Maria, Santa Maria.[Ref.]
- Canilha L, CarvalhoW, Silva JBA (2006) Biocatalizadores imobilizados uso de células imobilizadas em processos biotecnológicos. Biotecnologia Ciência e Desenvolvimento 9: 48-57.[Ref.]
- Rosa PRF (2010) Produção de probióticos com Lactobacillus imobilizados em alginato de cálcio empregando soro de queijo. (Dissertação de mestrado), Universidade Federal de Uberlândia, Uberlândia.[Ref.]
- Tampion J, Tampion MD (1987) Imobilized cells: principles and apllications. 1st edition, Cambrige University Press.[Ref.]
- Batista MA (2005) Estudo da imobilização de células de saccharomycescerevisiae em gel de alginato de cálcio no processo de fermentação alcoólica (Dissertação de Mestrado) Universidade Federal de Uberlândia, Uberlândia. [Ref.]
- Brinques GB (2009) Otimização da produção de probióticos em biorreatores e suas aplicações em sistemas alimentícios sob a forma imobilizada (Tese de Doutorado)Universidade Federal do Rio Grande do Sul, Rio Grande do Sul.[Ref.]
- Freitas FF (2007) Otimização do Processo de Imobilização de β – Galactosidase de Aspergillusoryzae em Alginato de sódio com gelatina e glutaraldeído (Tese de Doutorado) Universidade Federal de Uberlândia, Uberlândia.[Ref.]
- Araújo EA (2007) Desenvolvimento e Caracterização de Queijo tipo Cottage Adicionado deLactobacillusdelbrueckii UFV H2b20 e de Inulina. (Dissertação de Mestrado) Universidade Federal de Viçosa, Viçosa. [Ref.]
- Homayouni A, Aziz A, Ehsani MR, Yarmand MS, Razavi SH (2008) Effect of microencapsulation and resistant starch on the probiotic survival and sensory properties of symbiotic ice cream. Food Chemistry 111: 50-55.[Ref.]
- Guedes Neto LG, Penna CFAM, Fonseca LM, Cerqueira MMOP, Leite MO, et al. (2002) Lactobacillusacidophiluse a indústria de laticínios. In: Leite de derivados.[Ref.]
- OLIVEIRA AC (2006) Viabilidade de Lactobacillus acidophilus e Bifidobacterium lactis, microencapsulados por coacervação, seguida de secagem por spray drying e leito de jorro. Dissertação de Mestrado, Universidade de São Paulo.[Ref.]
- Yoon KY, Woodams EE, Hang YD (2004) Probiotication of Tomato Juice by Lactic Acid Bacteria. J Microbiol 42: 315-318.[Ref.]
- Krasaekoopt W, Bhandari B, Deeth H (2006) Survival of probiotics encapsulated in chitosan-coated alginate beads in yoghurt from UHT – and conventionally treated milk during storage. LWT - Food Science and Technology 39: 177-183.[Ref.]
- Mandal S, Puniya AK, Singh K (2006) Effect of alginate concentrations on survival of microencapsulated Lactobacillus casei NCDC-298. International Dairy Journal 16: 1190-1195.[Ref.]
- Gomes AMP, Malcata FX (2002) Agentes probióticos em alimentos: aspectos fisiológicos e terapêuticos, e aplicações tecnológicas. Biotecnologia Alimentar: Boletim de Tecnologia 101: 12-22.[Ref.]
- Jay JM (2005) Microbiologia de Alimentos. 6th edition. Porto Alegre: Artmed Editora S. A.[Ref.]
- Desmond C, Ross RP, O’callaghan E, Fitzgerald G, Stanton C (2002) Improved survival of Lactobacillus paracasei NFBC 338 in spray-dried powders containing gum acacia. J Appl Microbiol 93: 1003-1011.[Ref.]
- Tsen JH, Huang HY, King VA (2007) Enhancement of freezingresistance of Lactobacillus rhamnosus by the application of cell immobilization. J Gen Appl Microbiol 53: 215-219.[Ref.]
Download Provisional PDF Here
Article Information
Article Type: Research Article
Citation: Cruz RG, Pereira SGF, Araújo EA and Dores MT (2015) Viability of Lactobacillus acidophilus Immobilized in Calcium Alginate Spheres and Submitted to Different Conditions of Stress. Nutr Food Technol Volume1.1: http://dx.doi.org/10.16966/2470-6086.101
Copyright: © 2015 Cruz RG, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
