Introduction
Following an extensive review of the literature, the American Institute
of Medicine (IOM) concluded that “myalgic encephalomyelitis (ME)
and chronic fatigue syndrome (CFS) are serious, debilitating conditions
that affect millions of people in the United States and around the world”;
that it is a “medical - not a psychiatric or psychological - illness” without
a “known cause or effective treatment” which “can cause significant
impairment and disability” [1] rendering 25% of patients homebound
or bedridden [2] yet “the term chronic fatigue syndrome can result in
trivialization and stigmatization” of this “complex, multisystem, and often
devastating disorder” [1] .
Most doctors are unaware of the seriousness of ME or that it has been
classified as a neurological disease by the World Health Organization
(WHO) since 1969 [3]; therefore, patients often receive “hostility from
their health care provider” and are “subjected to treatment strategies that
exacerbate their symptoms” (i.e., CBT and GET) [1].
This is in stark contrast to the conclusions of the PACE trial and follow
up articles, which noted that “Chronic fatigue syndrome (CFS, also called
Myalgic encephalomyelitis/encephalopathy or ME) is a debilitating
condition with no known cause or cure” [4] but concluded that CBT and
GET are safe and cured 22% of patients [5]. The trial’s conclusions and
methodology have been criticized by patients, who have been ignored thus
far; however, a recent analysis by public health expert and investigative
reporter Tuller highlighted methodological problems in the PACE trial
[6-8]. Further issues concerning the methodology of reporting were
highlighted by Coyne [9] and Laws [10].
The PACE trial, which cost 8 million dollars [6], was a multicenter trial,
the largest CBT and GET trial for ME/CFS conducted thus far, involving
641 patients, established because systematic reviews found that CBT and
GET were promising treatments for CFS/ME [11], but “the published
trials” were “criticized for being too small, too selective, and for using
different outcome measures” [4] but also because of “an absence of data
for safety outcomes, and high dropout rates” [11]. The main aim of the
trial was “to provide high quality evidence...about the relative benefits...as
well as adverse effects, of the most widely advocated treatments for CFS/
ME” (i.e., CBT and GET) [4]. And the trial compared “pacing, defined as
adaptive pacing therapy (APT), CBT and GET, when added to specialist
medical care (SMC) with SMC alone” [11].
Objectives
Healthcare, including psychological interventions, should be evidence
based, and healthcare evaluation should incorporate the patient’s
perspective [12-14]. As a doctor who has been bedridden with severe ME
for a long period after GET caused a severe relapse from which I have not
recovered, I am in a unique position to combine the patient and doctor
perspectives, to elucidate the safety and effectiveness of CBT and GET in
ME/CFS by reviewing and analyzing the outcomes of the PACE trial and
follow-up articles, in particular the objective ones, to answer the following
questions objectively:
- Are CBT and/or GET effective treatments for ME/CFS?
- Are these treatments safe?
- Is recovery from ME/CFS with these treatments possible?
- Were there any other important findings, and if so, what were
they?
Full-text articles, including published supplementary material, were
retrieved and analyzed, using PubMed to find them. Treatment manuals,
patient newsletters, and the PACE trial protocol were also used and this
review includes extensive use of direct quotes from the PACE trial and
follow-up articles to avoid ambiguity as to whether the information
contained therein was fabricated or misinterpreted.
Background Information
On June 22, 2003, the PACE trial was registered with the International
Standard Randomised Controlled Trial Number registry (ISRCTN,
number 54285094), “a registry and curated database containing the
basic set of data items deemed essential to describe a study at inception”
[15], and placed in the “Mental and Behavioural Disorders” condition
category [16], even though ME has been classified as a neurological
disease by the WHO, which uses CFS as a synonym for ME, since
1969 [3]. Use of the WHO International Classification of Diseases is
mandatory in England [17], where the trial took place, and as stated by
Dr. L’Hours from the WHO, “according to the taxonomic principles…
it is not permitted for the same condition to be classified to more than one
rubric” [18].
The “scientific title” of the PACE trial is listed as “a randomised
controlled trial of adaptive pacing, cognitive behaviour therapy, and
graded exercise, as supplements to standardised specialist medical care...
for patients with the chronic fatigue syndrome/myalgic encephalomyelitis
or encephalopathy” [16]. “All research and therapy staff and participants
are unblinded to treatment allocation of individual participants” [4] and
“There is no masking” [16]. In addition, there was no placebo control
group; therefore it was not a randomized controlled trial but an unblinded
trial that relied on 2 subjective self-reported primary outcomes: “fatigue
(measured by Chalder fatigue questionnaire score) and physical function
(measured by short form-36 subscale score)” [11]. Yet as concluded by
Wood et al. [19], “In trials with subjectively assessed outcomes lack of
adequate allocation concealment or of blinding tend to produce overoptimistic
estimates of the effect of interventions”
The “PACE trial protocol: Final version 5.0,” was submitted to the
ISRCTN on “01.02.2006”, was “updated from protocol 3.1, 11.02.2005
and incorporates the following” 2 substantial amendments, “4.1,” dated
“05.08.2005” and “5.1,” dated “01.02.2006” [20].
The protocol’s publication history shows that “Version 1” was submitted
on “30 Oct 2006”, and “Version 2” was resubmitted “24 Jan 2007”, the
protocol was accepted on Jan 31, 2007 and published on March 8, 2007
[21]; however, the trial involved the recruitment of “641 participants”
“between March 18, 2005, and Nov 28, 2008” [11]. Therefore, the trial
began on March 18, 2005, and the final revised protocol was published 2
years later, on March 8, 2007. As Evans noted, “A fundamental principle
in the design of randomized trials involves setting out in advance the
endpoints that will be assessed in the trial, as failure to prespecify
endpoints can introduce bias into a trial and creates opportunities for
manipulation” [22]. And therefore the final version of a protocol should
be published before the trial starts.
However, Evans [22] also noted that “sometimes new information may
come to light that could merit changes to endpoints during the course of a
trial” and “Such changes can allow incorporation of up-to-date knowledge
into the trial design. However,” they “can also compromise the scientific
integrity of a trial” [22]. But “Changes in long-term trials” should be
considered “as medical knowledge evolves or when assumptions made in
design of the trial appear questionable” but only if the decision “to modify
an endpoint” is made “independent of the data obtained from the trial
to date,” and only an “external advisory committee that has not reviewed
data from the trial” should make those changes. It is “not appropriate”
that “decision makers” (i.e., “study sponsors, investigators, and DMCs”
who may have “impressions” “of the trial to date,” which “may influence
decisions regarding changes in endpoints”) make endpoint “revisions
during the trial” [22]. Yet the authors state that “The statistical analysis
plan was finalised, including changes to the original protocol, and was
approved by the trial steering committee... before outcome data were
examined” [11]. This indicates that the changes were implemented by the
authors and approved by the trial steering committee, and this procedure
differs from that described by Evans above [22]. This statement is of
particular importance, as many endpoint changes were made during the
PACE trial.
Eligibility Criteria
According to the PACE trial, “chronic fatigue syndrome (CFS) is a
condition characterised by chronic disabling fatigue and other symptoms,
which are not better explained by an alternative diagnosis. Myalgic
encephalomyelitis/encephalopathy (ME) refers to a severe debilitating
illness thought by some to be a separate illness, but by others to be
synonymous with CFS. In keeping with the MRC Research Advisory
Group report and the CMO’s working group report, we will refer to the
illness using both terms: CFS/ME” [4]. Which means that the PACE trial
regarded ME and CFS as the same illness.
The eligibility criteria used in the PACE trial included fulfillment of
the Oxford Criteria, with “a bimodal score of 6 of 11 or more on the
Chalder fatigue questionnaire and a score of 60 of 100 or less on the short
form-36 physical function subscale. 11 months after the trial began, this
requirement was changed from a score of 60 to a score of 65 to increase
recruitment” [11].
According to the first participant newsletter (June 2006), when this
change was made in February 2006, 75–80 of 641 patients had been
recruited [23], and according to the second participant newsletter
(March 2007), “Three new PACE centres have recently started recruiting
participants to the trial” and “A seventh centre in Bristol will start
recruiting in the spring” [24]. This indicates that recruitment problems
were attributed to the fact that only 3 of the 7 PACE centers were open
when the trial began in March 2005, that it took almost 2 years to open an
additional 3 centers and longer than 2 years to open all 7 centers.
The consequence of changing the eligibility criteria was that healthier
people, for whom it is easier to exercise and who experience fewer
problems doing so, were selected for participation in a trial to assess the
efficacy and safety of 4 treatments, including an exercise treatment (GET),
which could have affected related outcomes. This change also created an
overlap in the Short Form 36 (SF-36) physical function entry (a score of
≤ 65) and recovery (a score of ≥ 60) criteria. This also occurred with a
change to the Chalder Fatigue Questionnaire recovery score [5], where
by a score of 18 represented both recovery and eligibility, so that 13%
of patients were already considered to have recovered on 1 or 2 of the
primary outcomes upon entering the trial [6], the only raw data released
by the trial, other requests were deemed “vexatious” and refused [6] even
though the UK Medical Research Council policy concerning research
data sharing states that “publicly-funded research data are a public good,
produced in the public interest” which “should be openly available to the
maximum extent possible” [25]. And the Research Data Management
Policy of Queen Mary University of London, the home of the PACE
trial, states that, “publicly funded research data should be made openly
available in a timely manner” [26].
Wicherts et al. [27] explored “authors’ reluctance to share data” in
psychological studies and found that the “willingness to share research
data is related to the strength of the evidence and the quality of reporting
of statistical results”. They concluded that it was “rather disconcerting that
roughly 50% of published papers in psychology contain reporting errors
and that the unwillingness to share data was most pronounced when the
errors concerned statistical significance” [27].
The Trial’s ME/CFS Criteria
The main characteristic of ME is abnormally delayed muscle recovery
following trivial activities, as noted by infectious disease specialist Dr.
Melvin Ramsay [28], who witnessed and documented the outbreak of an
unknown disease in the Royal Free Hospital, London, in 1955 which was
initially thought to be atypical poliomyelitis and later became known as
ME [28]. However, the Oxford Criteria used by the PACE trial “require
fatigue to be the main symptom, accompanied by significant disability, in
the absence of an exclusionary medical or psychiatric diagnosis (psychosis,
bipolar disorder, substance misuse, an organic brain disorder, or an eating
disorder)” [11], even though (chronic) fatigue is not required to diagnose
ME as noted by Dr Dowsett [29] in 1992, who documented that “One of
the most striking features of ME is that the patient is not tired all the time”.
And on top of that, as noted by Ramsay, a diagnosis of ME should not be
made without the abnormally delayed muscle recovery [28].
The Oxford Criteria, which do not exclude patients with “any depressive
disorder and any anxiety disorder, including phobias, obsessivecompulsive
disorder, and post-traumatic stress disorder” [11], include
patients whose fatigue could be caused by a psychiatric disorder which
could inflate CBT and GET efficacy and safety outcomes as, contrary to
patients with a psychiatric problem, ME/CFS patients suffer from delayed
recovery and worsening of symptoms following exercise, as objective
evidence provided by Paul et al. [30] and Black et al. [31] for example,
showed. Failure to exclude psychiatric disorders resulted in the inclusion
of participants in which 33% and 47% had active depression and comorbid
psychiatric disorders, respectively [11] and could have included patients
with exercise phobia, leading to an erroneous impression of ME/CFS.
In 2003, a group of researchers led by Reeves from the Centers for
Disease Control and Prevention (CDC), which included one of the
principal investigators from the PACE trial, concluded via consensus, that
“the presence of a medical or psychiatric condition that may explain the
chronic fatigue state excludes the classification as CFS in research studies
because overlapping pathophysiology may confound findings specific to
CFS” [32]. This means that the Oxford Criteria should no longer be used.
Unfortunately the PACE trial ignored their own recommendation, which
is not surprising, as 2 of the 3 principal investigators and a PACE trial
research center leader were involved in creating these criteria in 1991 [33].
Consequently, at baseline, only 67% of participants met the “international
criteria for chronic fatigue syndrome” [11], which are the 1994 Fukuda
Criteria [34], in which the main characteristic of ME is optional, unlike
the 2011 International Consensus Criteria, in which this is a requirement
[2] and only 56% of participants fulfilled the “London Criteria for Myalgic
Encephalomyelitis” [11], yet Goudsmit, one of the authors of the London
Criteria [35], noted, “The version used in the PACE trial was not written
by any of those who were invovled “(sic!)” with the London Criteria” [36].
According to Shepherd [37], the medical advisor of the ME Association
and one of the authors of the original version of the London Criteria,
“White et al modified the Task Force version...and called it version 2”
omitting the requirement that “it is vital that the M.E. study groups we
use in research are as ‘pure’ as possible, the existence of a parallel disease
would be grounds for disqualification” [35]. However, as stated by David
[38], “British investigators have put forward an alternative, less strict,
operational definition which is essentially chronic…fatigue in the absence
of neurological signs…with…psychiatric symptoms…as common
associated features”.
The differences in percentages when using stricter criteria highlight the
heterogeneity of the population because of using the Oxford Criteria. If
the original London Criteria [35] had been used the 47% of participants
with comorbid psychiatric conditions [11] would have been excluded.
Subjective Outcomes
The chalder fatigue questionnaire
One of the two primary outcomes of the PACE trial was “fatigue
(measured by Chalder fatigue questionnaire score)... up to 52 week” [11]
yet “the original bimodal scoring” was changed “to Likert scoring to more
sensitively test our hypotheses of effectiveness” [11].
Table 1 shows the Chalder Fatigue Questionnaire [20] showing my
own situation, using the original binary or bimodal scoring on the left and
Likert scoring on the right [11]. Prior to viewing my scores, it is important
to reiterate that I have been bedridden with severe ME for a long period,
because I no longer have the muscle power to sit, stand, or walk. I am
dependent on others and I was seen by a consultant psychiatrist, who
excluded psychiatric disorders.
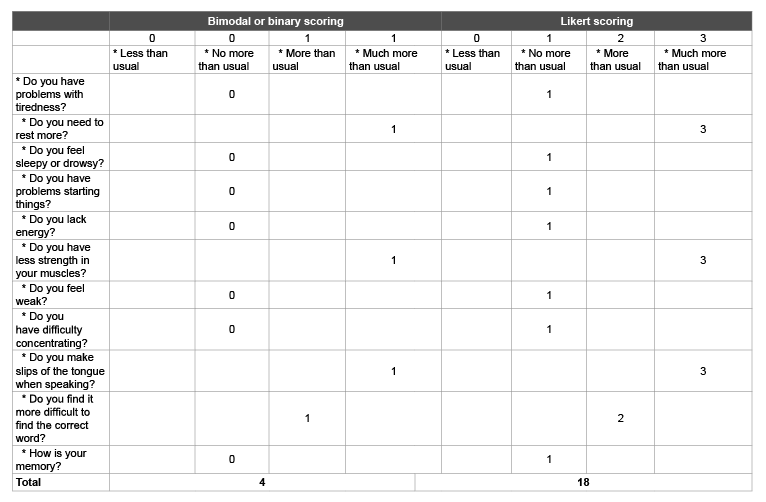
Table 1: Chalder Fatigue Questionnaire which I filled in myself assessing my own situation
The 11 questions in the Chalder Fatigue Questionnaire, as used in the PACE trial.
Bimodal scoring allocates 0, 0, 1 and 1 and Likert scoring allocates 0, 1, 2, and 3 to each of these answers [20].
Table 1 shows that I score 4 on the binary and 18 on the Likert scale.
My binary score indicates that I would have been ineligible, and therefore
not ill enough, to enter the ME/CFS PACE trial, despite having severe ME,
but the Likert score indicates that I would have been eligible for entry.
However, one cannot be ill enough and not ill enough simultaneously. A
Likert score of ≤ 18 was also one of the recovery criteria [11]; therefore,
without having received any treatment, I would also be classed as recovered
on the Chalder Fatigue Questionnaire even though my medical situation
has not changed and I remain bedridden and dependent on others.
This highlights one of several problems with this scale, showing that 2
entirely different outcomes are produced by simply changing the scoring
system. This demonstrates that these scores cannot be compared, and
changing the scoring system in the midst of a trial, makes this instrument
unreliable.
Furthermore, I did not score many points, despite having severe ME,
because too many items were depression related, which is irrelevant in
ME/CFS. Yet it is not surprising, as the scale, which does not provide a
comprehensive reflection of fatigue-related severity, symptomology, or
functional disability in ME/CFS [39], was developed by mental health
professionals [40]. This also means that improvements could simply be
improvements in comorbid psychiatric disorders, present in 47% of trial
participants [11], emphasizing the issues involved in failing to exclude
people with psychiatric disorders. The National Institute of Health
therefore concluded that “The Oxford Criteria…are flawed and include
people with other conditions, confounding the ability to interpret the
science” [41] and that “continuing to use the Oxford definition may impair
progress and cause harm” and “we recommend that the Oxford definition
be retired” [41,42].
Objective outcomes
The authors concluded that “Both self-report and objective measures
were used, and both were found to mediate treatment effects, lending
credence to the results” [43] and “Our conclusions are supported by
secondary outcomes, as both CBT and GET provided greater improvements
than did APT and SMC” [11]. In other words, the authors concluded
that both subjective and objective measures showed that CBT and GET
were effective treatments. However, following successful treatment, all 4
treatment groups’ mean scores for the 2 self-report primary outcomes
remained below the trial’s entry criteria [11,44], which meant that people
remained sufficiently ill to re-enter the trial. And lost employment and
Welfare benefit claims, 2 of the objective outcomes [45], showed that at
52-week follow up, the proportion of patients in receipt of income benefits
had increased from 10% to 13% and from 14% to 20% in the CBT and
GET group respectively [45]. The proportion of participants in receipt of
illness/disability benefits increased from 32% to 38% and from 31% to
36% in the CBT and GET group respectively [45] and there was a 100%
increase in the proportion of participants in receipt of income protection
or private pensions in both these groups [45] at 52 week follow up.
However, the authors also concluded that 22% of participants were
considered to have recovered in both treatments groups [5]. Treatment
concluded at 36 weeks, and follow up was performed at 52 weeks; therefore,
the participants, particularly the 22% who were considered recovered, had
16 weeks to find employment so that the number of participants in receipt
of benefits should have decreased dramatically, but instead it went up.
The self-paced step test
The “submaximal self-paced step test”, “strongly and reliably predicts
the maximal aerobic capacity VO2max, is sensitive to change” [46], and it
was used as an objective measure of fitness in the PACE trial. The results
of the self-paced step test [11] show the levels of fitness in response to the
4 treatments and it highlights the finding that CBT and GET produced the
least and the pacing group the greatest improvement even though patients
in the GET group had been exercising “five times a week”, in the form of
walking, or cycling or swimming [43]. The overall conclusion was “that
fitness... did not appear to mediate treatment effects” [43]. In other words,
fitness did not improve, and none of the treatments were effective.
The Six-Minute Walk Test (6MWT)
The 6MWT provides a good representation of the patient’s ability to
perform submaximal activities of daily living and may be useful in serial
evaluation of patient status and/or response to therapeutic interventions
[47]. The results of the 6MWT showed improvements with all 4 treatments.
The GET group improved most and walked 379 m within 6 min, with an
adjusted difference of 35 m beyond that of the SMC group after 52 weeks.
The CBT group walked 354 m, which was an improvement of 4 m beyond
that of the APT group but 1.5 m less than that of the SMC group [11].
None of these improvements reached statistical significance, for which an
increase of at least 86 m was required [48].
The average age of PACE trial participants was 38 years [11], and
normal distances for healthy men and women aged 38 years are 659 and
600 m, respectively [49]. Patients with severe MS manage 389 m [50];
those with Class 3 heart failure (with Class 4 considered the worst),
who experience difficulty in completing everyday activities and have a
1-year mortality rate of 10–15% [51], manage 402 m [52]; and patients
with end-stage lung or heart disease walk 335 m or less [53]. Yet ME/CFS
patients who had completed a full course of “successful” CBT provided
by qualified and experienced therapists only managed 19 m more, and
those for whom treatment was most effective (i.e., GET) only managed
379 m and were outperformed by community dwelling 80–89 year olds
with chronic health problems, who walked 417 m [54]. It is worth noting
that people in the GET group engaged in “30 min of physical exercise
five times a week,” and this was “most commonly walking” [43], so that
walking only one fifth of it during the 6MWT should be easy and they
should easily reach the age related normal levels at the 52-week follow-up,
in the absence of an underlying physical problem, which is the assumption
of the biopsychosocial model.
Patients who are able to walk 400 m or less, are placed on the waiting
list for a lung transplant [55], and within a year of transplant, their 6MWT
results return to normal [56]. However, following “effective” treatment,
ME/CFS patients would remain on the transplant list.
Yet according to the secondary mediation analysis, “Exercise tolerance
as measured by the number of metres walked in a fixed time was a strong
mediator of GET alone” [43]. However in the supplementary material
for the secondary mediation analysis, the significance of the 6MWT was
deemphasized [57], because the researchers noted an “increase in exercise
tolerance (walking distance) without an increase in exercise capacity
(fitness)” [43] which means that the patients had optimized their walking
distance without increasing their fitness, despite engaging in “physical
exercise five times a week” [43], because the therapies used did not
address the underlying metabolic problem preventing improvements in
fitness. However, rather than providing an explanation to this effect, the
following reasons (in full with cited reference numbers) were provided for
failing to use 6MWT outcomes.
“Six-Minute Walk Test Participants were asked to walk as far as possible
in six minutes, and the distance walked in metres was recorded. This is
a measure of exercise tolerance (14). Due to concerns about patients
with CFS coping with physical exertion, no encouragement was given to
participants as they performed the test, in contrast to the way this test
is usually applied (15,16). Rather than provide encouragement, we told
participants, “You should walk continuously if possible, but can slow
down or stop if you need to”.
Furthermore we had as little as 10 metres of walking corridor space
available in centres rather than the 30 to 50 metres of space used in other
studies (15-17); this meant that participants had to stop and turn around
more frequently. Due to the modifications and the associated measurement
error we considered this test as an internally referenced measure of behaviour
change or exercise tolerance, not a measure of physical fitness” [57].
It is reasonable to expect, in a trial costing 8 million dollars [6], that
the 3 principal investigators, who are mental health, and not 6MWT
professionals, should have obtained guidelines for performing the 6MWT
(one of the trial’s secondary outcomes).
Reference number 15 in the above statement refers to the American
Thoracic Society (ATS) 6MWT guidelines published in March 2002 [58],
the PACE trial began 3 years later in March 2005 [11] and the full text is
available free of charge on the ATS website [58].
There are a number of inaccuracies in the PACE trial statement above,
as the following quotes from the ATS guidelines show. “The self-paced
6MWT assesses the submaximal level of functional capacity. Most
patients do not achieve maximal exercise capacity during the 6MWT;
instead, they choose their own intensity of exercise and are allowed to
stop and rest during the test” [58]. This indicates that the 6MWT is a
measure of exercise tolerance rather than physical fitness and patients can
stop and rest whenever they need to. The guidelines stipulate that “the
walking course must be 30 m in length”. However, “some have used 20- or
50-m corridors” and there is “no significant effect of the length of straight
courses ranging from 50 to 164 ft” (i.e., 15–50 meters) [58].
Standardized “encouragement” during the walk includes “Keep up the
good work. You have only 2 minutes left” and “You are doing well. You
have only 1 minute to go”. However, the guidelines also stipulate “Do not
use other words of encouragement (or body language to speed up)” [58].
Therefore, it is standard practice to inform patients without encouraging
them to walk further than they can.
The PACE trial was based on the biopsychosocial model, whereby there
is nothing physically wrong in ME; however, this model is contradicted in
the above statement, as the researchers expressed “concerns about patients
with CFS coping with physical exertion” [57], and this refers to walking
for only 6 minutes, which constitutes an indirect acknowledgement of the
invalidity of the biopsychosocial model.
The Actometer
The Actometer, an objective and reliable measure of activity [59],
was meant to be used, at the beginning and the end of the trial, to assess
improvement objectively; the last was deemed “too great a burden” [60]
for patients, even though it weighs only 26 g [59], patients had consented
to use it, had completed “moderately effective” treatment [11], and 22% of
those in the CBT and GET groups had recovered [5]; therefore, it should
have been easier and less of a burden.
Are CBT and GET Safe?
The PACE trial protocol noted, “There is a discrepancy between patient
organisation reports of the safety of CBT and GET and the published
evidence of minimal risk from RCTs” [4]. The PACE trial concluded that
“all four treatments tested are safe” [11], which is not surprising given
that the protocol stated, “A risk assessment has been undertaken, and the
therapies are of low risk to participants” [4]. The CBT and GET treatment
manuals informed patients that these treatments were safe and effective
[61,62], even though the trial was established to determine if they were or
not, and used patient-rated subjective primary outcomes. This raises the
question as to how safety was defined and assessed.
According to the PACE trial, “safety was assessed primarily by
recording all serious adverse events, including serious adverse reactions to
trial treatments” [11] and “Adverse events were considered serious when
they involved death, hospital admission, increased severe and persistent
disability, self-harm, were life-threatening, or required an intervention
to prevent one of these” [11]. However, in patient surveys most of these
issues were not raised. Instead, they reported that CBT and GET could
exacerbate ME/CFS symptoms and cause relapses [63].
According to the PACE trial protocol, a high proportion of participants’
symptoms worsened with these treatments because of “rigidly applied
programmes that are not tailored to the patient’s disability” [4] and “PACE
treatment manuals minimize this risk by being based on mutually agreed
and flexible programmes that vary according to the patient’s response” [4].
If this were the case, GET would not have left me bedridden. However,
the basis of GET is that patients should ignore their symptoms (because
they are signs of deconditioning rather than illness) and adhere to an
established program designed by a qualified therapist, which increases
exercise incrementally without considering the patient’s symptoms during
the program. As soon as therapists tailor exercise programs to patients’
symptoms GET is not GET anymore but has become a form of pacing.
In the PACE trial “Non-serious adverse events were common” [64].
Actually they were very common, as 93% of patients reported nonserious
adverse events; 49% and 51% reported 4 and more than 4 such
adverse events, respectively, and there were no differences between the 4
groups [64]. A simple way to explain ME is to state that the M stands for
musculoskeletal and the E for neurological problems. Therefore, in the list
of non-serious adverse events, only 3 groups were considered (i.e., those
involving musculoskeletal, neurological, and ME-related non-serious
adverse events).
In the PACE trial, 46% of participants reported an increase in ME/CFS
symptoms; 31% and 19% reported musculoskeletal and neurological nonserious
adverse events, respectively [64]. It’s unclear how large the overlap
was between these three groups and therefore the proportion affected
by CBT and GET would have been between 46% and 96%. As noted by
Kindlon [65], who pooled a number of surveys, 20% and 51% of patients
reported that CBT and GET, respectively, exacerbated their symptoms;
Kindlon [65] also found that this was 82% in patients with severe ME,
and many were not severely affected or bedridden prior to GET. The real
proportion is likely to be approximately 74%, as concluded in a recent ME
Association survey involving more than 1,400 patients [63].
These figures differ entirely from the 2% and 1% of participants reporting
serious adverse events in the CBT and other 3 groups, respectively, which
the PACE trial used to declare all 4 treatments to be safe [11].
It is particularly interesting to note that these numbers are also very
high in the adaptive pacing group, in which patients were told to remain
within their limits, and the SMC group, which did not involve any exercise,
so that very low rates of adverse events would be expected in both groups.
The very high proportions of adverse events in these 2 groups suggest
that many individuals experienced difficulty completing the step test and
6MWT as expected in patients with ME/CFS, who experience abnormal
responses to exercise, as highlighted by 2-day exercise testing [66,67],
abnormal gene expression and immunity following exercise [68,69], and
a left shift in anaerobic threshold observed in a number of studies [70]
which was recently explored by Vink [71], in a former Dutch national
field hockey champion who is now bedridden with severe ME. Vink [71]
concluded that an impaired oxidative phosphorylation is the reason why
he can only perform trivial activities, and an impaired lactic acid excretion
plays an important role in the abnormally delayed muscle recovery.
The Supplement to the secondary mediation analysis shows that 90% of
patients in the APT group completed the primary outcome questionnaires
at follow up, but only 71% performed the 6MWT, and only 62% completed
the step test [57]; however, the trial reported that only “33 (5%) of 640
participants were lost to follow-up, but rates did not differ between groups”
[11] and “by 52 weeks, only 33 (5%) were missing primary outcome
data, with no significant difference between treatment groups” [5]. This
suggests that 29% and 38% of the participants, respectively, withdrew
from the study because the first step test and 6MWT exacerbated their
symptoms, and as 93% reported adverse events, it suggests that these 29%
and 38% constitute the tip of the iceberg.
Recovery
The recovery article states that “We changed our original protocol’s
threshold score for being within a normal range on this measure from
a score of ≥ 85 to a lower score as that threshold would mean that
approximately half the general working age population would fall
outside the normal range. The mean (S.D.) scores for a demographically
representative English adult population were 86.3 (22.5) for males and
81.8 (25.7) for females. We derived a mean (S.D.) score of 84 (24) for the
whole sample, giving a normal range of 60 or above for physical function” [5].
Having recovered differs from being within the normal range, and the
PACE trial did not examine the “demographically representative English
adult population.” Furthermore Bowling et al. [72] did not involve the
“general working age population,” as the participants ages ranged from
16 to 85, with 28.6% aged 65 years or older. The PACE trial cohort’s mean
age was 38 years [11] and therefore participants should be compared to
healthy adults of the same age. According to the survey by Bowling et
al. [72], which the PACE trial used, the mean physical functioning score
for adults aged 35–44 years was 93.3, with a standard deviation of 13.4
[72] providing a normal score of ≥ 79.9, rather than ≥ 60, for physical
functioning.
However, SF-36 physical functioning scores for healthy 38 year olds are
not normally distributed but skewed to the right, with nearly everyone in
the maximum range; and according to the BMJ’s statistical resources for
readers, standard deviations will then be grossly inflated, are not a good
measure of variability anymore and are therefore inappropriate for use
[73], as highlighted by the fact that, Bowling et al.’s [72] survey of 2,000
patients, 1,200 and 400 had median scores of 100 and 90, respectively.
The SF-36 physical functioning score decreases with age and ill health,
almost 600 patients (28.6%) were aged 65 years or older, and 22% and
16% had chronic and acute health problems, respectively; therefore, the
SF-36 physical functioning recovery score should have been 100 rather
than 60 or more.
According to the PACE recovery article, “Before we can determine the
proportions recovered we need an operational definition of recovery itself.
An ideal definition remains uncertain” [5]. However, this is inaccurate, as
concluded by Kennedy [74], recovery “is the elimination of...symptoms
and a return to premorbid levels of functioning”.
The recovery article also stated, “it is important to note that recovery
does not mean being free of all symptoms” [5]. However, if the symptoms
of a disease remain, than it is an overly optimistic definition of recovery,
including ill people. The following statement acknowledges this: “The
prevalence of the case-level international (CDC) definition of CFS may
have been inaccurate because we only examined for accompanying
symptoms in the previous week, not the previous 6 months” [5], as most
symptoms that have been present for less than a week are self-limiting
and symptoms that have been there for 6 months or more are not. “We
therefore use the term ‘recovery’ in this paper to mean recovery from
the current episode of the illness” [5]. According to this, instead of being
able to walk 5 or 6 yards to the toilet twice daily, I would be able to do
so 3 times per day and otherwise remain bedridden highlighting the fact
that recovery from the current episode of illness does not necessarily
constitute recovery.
The protocol stated: “‘Recovery’ will be defined by meeting all four of
the following criteria: (i) a Chalder Fatigue Questionnaire score of 3 or
less, (ii) SF-36 physical Function score of 85 or above, (iii) a CGI score
of 1, and (iv) the participant no longer meets Oxford Criteria for CFS,
CDC criteria for CFS or the London criteria for ME” [4]. Yet even though
the authors chose these themselves, during the trial they “changed three
of the thresholds for measuring recovery from our original protocol” [5],
reflected in the following 6 statements.
The Chalder Fatigue Questionnaire (CFQ)
“We changed our original protocol’s threshold score for being within a
normal range from a binary score of ≤ 3 out of 11” to “a population mean
(S.D.) Likert score of 14.2 (4.6) out of a maximum score of 33” [5]. This
indicates that both the score and the scoring system were changed.
The SF-36 physical function subscale:
“We changed our original protocol’s threshold score for being within a
normal range on this measure from a score of ≥ 85 to” “60 or above” [5].
Oxford criteria
“To satisfy the third criterion for severity of fatigue and disability,
participants had to meet trial entry thresholds for fatigue (a binary score
of ≥ 6 out of 11 on the CFQ) and abnormal levels of physical function (a
score of ≤ 65 out of 100 on the SF-36 physical function subscale)” [5].
However, the original score required for the SF-36 was “a score of 60 of
100 or less” [11]. And the bimodal Chalder Fatigue Questionnaire score of
≥ 6 out of 11 was changed to a Likert scoring of 18 or more, therefore, the
Oxford Criteria were also changed during the trial.
The CDC CFS case definition
“For the purposes of this study, the four or more symptoms needed to
be present within the previous week of the assessment date, rather than
the previous 6 months” [5]. Most symptoms that are present for less than
a week are acute and self-limiting; however, symptoms that have been
present for 6 months are chronic and not self-limiting.
The London criteria
“Specifically, these criteria included... no ‘primary’ depressive illness
and no anxiety disorder present (which we interpreted as no co-morbid
mood disorder of any kind)” [5]. However, these criteria also state that
“the existence of a parallel disease would be grounds for disqualification”
[35], which was omitted from the version used in the trial, without the
knowledge of the original authors according to Goudsmit and Shepherd
[36,37], 2 of the original authors. All psychiatric diseases should therefore
have been excluded which did not occur.
The self-rated CGI change score
“We considered scores of 1 (‘very much better’) or 2 (‘much better’)
as evidence of the process of recovery, rather than our original protocol
threshold of a score of 1 only, because we considered that participants
rating their overall health as ‘much better’ represented the process of
recovery” [5]. However, everyone who’s had a flu-like illness knows that,
while one feels much better during convalescence, relative to the day upon
which one fell ill, one is in the process of recovery but has not recovered
yet which highlights the fact that the self-rated CGI change score is a
global measure of change and clinical progress [75], rather than a measure
of recovery, and is also “unreliable and too general to measure... treatment
responses validly” [76].
Table 2 highlights the changes to the recovery criteria, made by the
authors during an unblinded trial, rather than by an independent external
trial committee with no access to the data to avoid any form of bias, as
stipulated by Evans [22].
The recovery article also stated that “Although it seemed that slightly
smaller proportions had recovered from the illness as a whole, when the
criterion ‘not meeting the London criteria for ME’ was applied, we found
that the differences were due to missing data rather than to change in
recovery status” [5]. But how did they know this if the data were missing?
One of the conclusions of the recovery article was “that CBT and GET
were both significantly more likely than APT and SMC to be associated
with recovery at 52 weeks, even when using a conservative definition
of recovery” [5]. A conservative definition of recovery ensures that the
proportion of patients who have recovered is deliberately lower than the
actual proportion. However, the endpoint changes during the trial caused
an overlap in entry and recovery criteria, whereby a score of ≥ 60 of 100 on
the SF-36 physical functioning subscale represented recovery, yet the entry
criteria required a score of ≤ 65. Therefore, even if participants’ health
deteriorated and their scores dropped from 65 to 60, they were classed as
having recovered, even though a score of 60 out of 100 is normal for 75-84
year olds [72] but represents disability in 38 year olds. As found by Tuller
[6], the consequence of this was that 13% of patients were already classed
as recovered on 1 or 2 of the primary outcomes as soon as they entered the
trial. Is that a conservative definition of recovery?
As mentioned previously and shown in Table 1, my bimodal Chalder
Fatigue Score indicated that even though I am bedridden and dependent
on others, I would not have been eligible for the trial. During the trial,
the scoring system was changed, and without changing my answers, I
suddenly scored the minimum (of 18) points required to enter the trial.
Yet with the same Likert score of 18, and without having received any
treatment or any change to my medical situation, I was also classed as
recovered, despite remaining bedridden. Is that a conservative definition
of recovery?
The authors also “changed some of the thresholds for measuring
recovery from those of the original protocols; we made the changes
before analysis and to more accurately reflect recovery” [5]. As shown
in Table 2, extensive changes were made to the recovery criteria during
the trial, which broadened the definition of recovery, rendering it less
accurate and less conservative. “Finally”, the authors said, “we cannot
be sure that recovery was sustained beyond the assessment at 52 weeks”
[5]. As the definition of recovery included people who remained ill or
disabled, they could not have sustained recovery beyond the 52-week
assessment, because they had not recovered in the first place. The authors
also noted that “Two studies of recovery in adults after CBT found similar
proportions in recovery: 23% and 24%”; “but the definition for normal
range used was the more liberal population mean ± 2 s.d. rather than the
more conservative 1 s.d. that we used” [5]. With respect to SF-36 physical
functioning scores, the standard deviation quoted in the PACE trial was
24. The minimum level of recovery defined in this manner was a score of
60 out of 100 in the PACE trial. The establishment of a second standard
deviation would have led to a minimum score of 36 out of 100 to represent
recovery which in a 38 year old indicates severe disability rather than
recovery, putting “recovery” in CBT studies for ME/CFS into perspective.
“Our finding that 22–56% of participants met various composite or single
criteria for recovery or improvement a year after starting either CBT or
GET is therefore consistent with previously published studies” [5]. Yet
even though I am bedridden and dependent on others, I fulfill the single
criterion for recovery using the Chalder Fatigue Questionnaire, the same
score also classified me as sufficiently ill to enter the trial; however, if a
patient fulfills only one of several criteria for recovery, the patient has not
recovered, regardless of the definition of recovery.
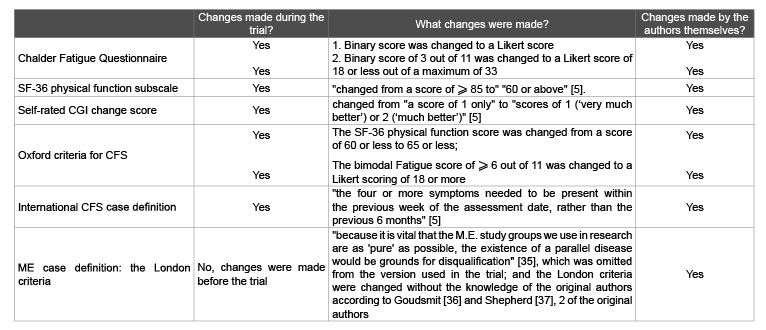
Table 2: Changes made to the recovery criteria
Source: PACE trial recovery article [5]
The authors also concluded that “The proportions recovered in
each treatment arm were similar in the subgroups meeting alternative
definitions of CFS and ME, implying that these findings generalize to
different definitions of CFS and ME” and “patients who have either CFS
or ME...should therefore be offered either CBT or GET to provide the
best chance of recovery with these treatments” [5]. Yet their definition
of recovery labeled patients as recovered, regardless of whether they
remained disabled or not.
Fear Avoidance Beliefs
In January 2015, the secondary mediation analysis was published, and
the authors stated that “Fear avoidance beliefs are characterised by fears
that activity or exercise will make symptoms worse” [43] and concluded
that “Our main finding was that fear avoidance beliefs were the strongest
mediator for both CBT and GET. Changes in both beliefs and behaviour
mediated the effects of both CBT and GET, but more so for GET. The
results support a treatment model in which both beliefs and behaviour
play a part in perpetuating fatigue and disability in chronic fatigue
syndrome” [43]. Yet in a rapid response in the BMJ two weeks later,
the authors stated, “nor did we say that fear of exercise in CFS was
“irrational”” and ME/CFS is “an illness where exercise increases
symptoms” [77], thereby acknowledging that avoiding exercise is the
appropriate course of action. And in 2005 when the PACE trial began,
one of the principal investigators coauthored an article in which the
conclusion was that “CFS patients without a comorbid psychiatric
disorder do not have an exercise phobia” [78]. The same principal
investigator also coauthored an article concluding that exercise causes
immunological damage/abnormalities in ME/CFS [79]. Why a blood
test to check these immunological parameters was not part of the
“definitive randomised trial” [80] conducted to determine whether CBT
and GET were effective and safe in ME/CFS is unclear.
The authors also stated, “the FINE trial found that fear avoidance,
embarrassment avoidance, all-or-nothing and avoidance behaviour
were cross-sectional mediators of the treatment effect” [43]. Yet the
FINE trial itself reported that the treatment effect, in more severely
affected patients, “is small and not statistically significant at one year
follow-up” [81], which was the primary outcome of the trial. Therefore,
these factors could not have been “mediators of the treatment effect,”
as there was no treatment effect. Yet the PACE trial authors concluded
“This mediational analysis strengthens the validity of our theoretical
model of CBT and supports the idea that a similar model is valid
for GET by confirming the role of fearful beliefs and avoidance
behaviour” [43], ignoring the study’s evidence to the contrary. And
when the authors concluded that “The increase in exercise tolerance
(walking distance) without an increase in exercise capacity (fitness)
might have been facilitated by the mediating effect of reduced fear
avoidance beliefs” [43], they should have concluded that these patients
had simply optimized walking distance without increasing fitness,
despite engaging in “physical exercise five times a week” [43], because
the therapies used did not address the underlying physical problem in
ME/CFS.
Removing the Naturally occurring Fluctuation of ME/
CFS
“The main finding of this long-term follow-up study of the PACE trial
participants is that the beneficial effects of the rehabilitative CBT and GET
therapies on fatigue and physical functioning observed at the final 1 year
outcome of the trial were maintained at long-term follow-up 2•5 years
from randomisation” [44]. However, this was not the main finding which
was that there was no difference in efficacy between treatments and none
of them were effective [44].
A review by Whiting et al. [82] showed that “The relapsing nature of
CFS suggests that follow-up should continue for at least an additional 6
to 12 months after the intervention period has ended, to confirm that any
improvement observed was due to the intervention itself and not just to a
naturally occurring fluctuation in the course of the illness”. The first follow
up was performed at 52 weeks, which was only 16 weeks after treatment
completion at 36 weeks, and long-term follow up was performed at least
2 years subsequent to randomization [44], which was at least 1.25 years
subsequent to treatment completion; therefore, the naturally occurring
fluctuation of the illness was omitted from consideration.
The authors stated that “In interpreting the follow-up data it is important
to note that many of the participants had received additional treatment for
chronic fatigue syndrome since completing the trial” [44] and “roughly
a quarter and a third of the participants originally allocated to APT and
SMC respectively had received a therapeutically adequate amount (ten
or more sessions) of CBT or GET after the trial final trial outcome” [44]
which made it “possible that this additional treatment was important in
improving the long-term outcome for these patients” [44]. However, the
Supplementary appendix long-term follow-up, which “formed part of the
original submission”, was “peer reviewed” and “supplied by the authors”
[83], shows that the majority of participants, i.e 76% and 83%, did not
have any additional CBT respectively GET, after the trial had finished.
Furthermore, researchers cannot control the environment subsequent
to completion of a trial; therefore, an effect cannot be attributed to the
receipt of any form of additional post trial treatment. Another problem
is the carryover effect, whereby the effect of treatment is carried over to
the second phase [84], which in this case was the phase following trial
completion. As noted by Larun et al. [84] this is a greater problem “when
the condition of interest is unstable,” and “both effects are very likely
in CFS/ME”. Last but not least, the Supplementary appendix long-term
follow- up [83] shows that in all 4 groups, patients who did not receive
additional treatment subsequent to trial completion exhibited lower
fatigue and higher physical functioning scores relative to those of patients
who received additional treatment. This suggests that additional CBT
and GET, provided by qualified and experienced therapists from the trial
subsequent to trial completion, could have been detrimental to patients’
health. And on top of that, in the CBT and SMC groups an adequate
number of sessions of CBT and GET were more detrimental than an
inadequate number of sessions [83].
Discussion
The PACE trial was a large multicenter trial established as a decisive
means of testing the safety and efficacy of CBT and GET in ME/CFS, based
on the conclusions of the Medical Research Council’s Research Advisory
Group and the CMO’s working group, that ME and CFS constitute the
same illness [4], and the biopsychosocial model “which supposes that
unhelpful interpretations of symptoms, fearful beliefs about engaging in
activity, and excessive focus on symptoms are central in driving disability
and symptom severity” [43] and that “both de-conditioning (loss of
muscle strength and reduced exercise capacity) and avoidance of activity...
maintain fatigue and disability” [43]. CBT and GET were designed “to
identify and challenge unhelpful cognitions” [43] and change and reverse
such cognitions by “gradually increasing physical activity to improve
fitness and get the body used to activity again” [5], and by doing so
cure ME/CFS. Yet the 2 subjective primary outcomes of the trial relied
on patients’ interpretations of their symptoms, but these 2 approaches
are at odds. If a model and treatment are based on patients’ incorrect
interpretations of their symptoms, then primary outcomes should not rely
on these. In addition to being unreliable, subjective data lack objectivity
and are prone to outside influences, particularly in trials that aim to
modify participants’ subjective beliefs [85]. There is a low correlation
between objective and subjective activity measurements [59] not only in
chronically ill but also in healthy people [86]. And the PACE trial authors
themselves noted that “objective measures of physical activity have been
found previously to correlate poorly with self-reported outcomes” in ME/
CFS [5]. In other words, in a decisive trial, the primary outcomes should
have been objective rather than subjective.
During the trial, extensive endpoint changes were made by the
authors themselves as shown by the following examples: “we made the
changes before analysis and to more accurately reflect recovery” [5] and
“We changed our original protocol’s threshold score for being within a
normal range from”... [5]. Yet during a trial, such alterations should only
be made for compelling reasons and only by an independent trial steering
committee without access to the data, rather than by the trial investigators,
as stipulated by Evans [22]. According to Goldacre: “Switching your
outcomes breaks the assumptions in your statistical tests. It allows the
“noise” or “random error” in your data to exaggerate your results (or even
yield an outright false positive, showing a treatment to be superior when in
reality it’s not)” leading to the wrong conclusions and “in medicine, that’s
not a matter of academic sophistry - it causes avoidable suffering” [87].
And according to Ioannidis [88], “Flexibility increases the potential for
transforming…“negative” results into “positive” results”, and the greater
the flexibility of the outcomes, the less likely it is that the research findings
are accurate [88]. This is highlighted by the fact that 13% of participants
were considered to have recovered, with respect to 1 or 2 of the recovery
criteria [6], as soon as they entered the trial, as the above-mentioned
endpoint changes created an overlap between entry and recovery criteria.
At 52-week follow up the objective outcomes showed the following: the
step test, a reliable objective measure of fitness [46], showed no significant
improvements in any of the 4 treatment groups; the 6MWT results
showed that ME/CFS patients would remain on the waiting list for a lung
transplant [55] following treatment deemed effective by the PACE trial;
employment rates did not differ significantly between the 4 treatment
groups, the number of patients claiming state sick pay and disability
benefits increased following CBT and GET [45], and the number of
patients in receipt of income protection or private pensions had actually
doubled in the CBT and GET groups [45] and the authors deemed it too
much of a burden [60] for patients to wear a 26 g weighing Actometer [59]
at the end of the trial even though the PACE trial concluded that CBT
and GET were moderately effective, and that 22% of patients in the CBT
and GET groups recovered following successful treatment [5]; therefore,
it should have been less of a burden to wear the Actometer at the end of
the trial then at the beginning and these figures should have decreased
dramatically and not gone up.
With respect to the definition of recovery, the PACE trial authors
stated, “it is important to note that recovery does not mean being free
of all symptoms” [5]. In other words, “recovery” was synonymous with
“no recovery”. The long-term follow-up study showed that patients
in all 4 groups still fulfilled the entry criteria for fatigue and physical
functioning, the 2 primary outcomes, following effective treatment [44].
So that, similar to the above-mentioned objective outcomes, both primary
outcomes showed that patients remained sufficiently ill to re-enter the
trial. At long-term follow up, no differences were found between the 4
treatments, which is known as a null effect, and none of the treatments
were effective [44]. And as noted by Ioannidis “investigators working in
any field are likely to resist accepting that the whole field in which they
have spent their careers is a “null field”” [88]; which might explain the
authors’ reluctance to release trial data.
Further, 46% of patients reported increases in their ME/CFS symptoms,
31% reported musculoskeletal, representing the M in ME, and 19%
reported neurological adverse events, representing the E in ME [64]. So
that the proportion of participants negatively affected by CBT and GET
is between 46% and 96%, and most likely estimated at 74%, as recently
reported by a large survey conducted by the British ME Association,
which involved 1,428 patients [63] and medication with such high rates
of adverse events should be withdrawn from the market with immediate
effect. This is in stark contrast to the PACE trial authors’ response to a
National Institutes of Health treatment review of ME/CFS [89], in which
the PACE trial authors stated, “It is important not to overemphasise
the harms associated with an effective treatment when there are so few
others available” [90]. Yet we should not ignore or underplay the harm
associated with CBT and GET for people with ME/CFS, and we should
also not ignore that the PACE trials long-term follow up showed that none
of the treatments were effective for ME/CFS [44]. This is in total contrast
to the promising results of the Norwegian Rituximab trials, which suggest
that ME/CFS is an autoimmune disease and showed that symptoms were
alleviated in 66% of patients, two thirds of whom remained in complete
remission at 36-month follow up [91].
The PACE trial’s null effect, confirms the experiences of patients,
which was also highlighted by Falk Hvidberg et al. [92] who concluded
that of patients with 21 diseases, including chronic renal failure, cancer,
and stroke, those with ME/CFS demonstrated the lowest quality of life
confirming the findings of the 1996 health status report [93], illustrating
the fact that nothing has changed within the last 20 years with respect
to ME/CFS patients’ health, providing further proof that CBT and GET,
which most ME patients have tried because they desperately want to
recover, are not effective.
The discovery that an increase in exercise tolerance did not lead to an
increase in fitness [43], as observed in healthy people, shows that there is
an underlying physical problem preventing this, which means that none
of the 4 treatments in the trial addressed this issue and validates that
ME/CFS is a physical disease. Therefore, the PACE trial, along with the
FINE Trial, which found that these treatments were ineffective in more
severely affected patients [81], disproved the biopsychosocial model as
an explanation of ME/CFS, confirming the conclusion of the American
Institute of Medicine, following an extensive review of the literature, that
ME/CFS is a serious and debilitating physical, rather than a psychological
or psychiatric, disease [1].
Conclusion
The PACE trial was the largest trial of its kind. During the trial
extensive endpoint changes were made, creating an overlap in entry and
recovery criteria so that 13% of patients were already recovered on 1 or
2 of the primary outcomes upon entering the trial [6]; the PACE trial’s
definition of recovery labeled patients as recovered, regardless of whether
they remained disabled or not. Its null effect invalidates the use of CBT
and GET in ME/CFS and invalidates the biopsychosocial model of
deconditioning and fear avoidance.
The PACE trial found that the proportion negatively affected by CBT
and GET was between 46% and 96%, most likely estimated at 74%, as
shown in a large survey recently conducted by the ME Association [63].
Medication with such high rates of adverse events would be withdrawn
immediately. The PACE trial’s discovery of an “increase in exercise
tolerance (walking distance) without an increase in exercise capacity
(fitness)” [43] means there’s an underlying physical problem preventing
this and that none of the treatments were addressing this issue but also that
ME/CFS is a physical disease, confirming the conclusion of the IOM [1].
And therefore, from now on our focus should be on biomedical (instead
of psychosocial) research to find effective treatment for this debilitating
disease.
Conflict of interest
No competing interests.
Funding
No funding was involved in supporting this work.
Acknowledgements
I would like to thank my parents for typing out my speech memos and
for proof reading the manuscript and Elsevier for the Language Editing of
the manuscript.