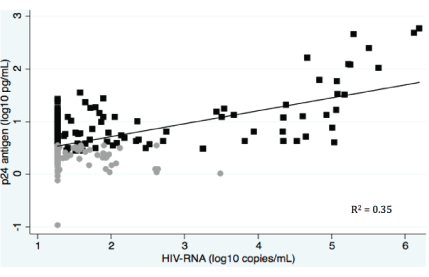
Figure 1: Scatter plot illustrating correlations between plasma p24 antigen and HIV-RNA measurements. Grey circles: undetectable p24 antigen. Black squares: detectable p24 antigen.


Mari S Melaaen1 Lasse S Vestergaard1,2 Nyagonde Nyagonde2,3 Ben Amos2,3 Filbert Francis2,3 Martha M Lemnge2,3 Ib C Bygbjerg1,2 Jan Gerstoft1 Marie Helleberg1 Terese L Katzenstein1*
1Department of Infectious Diseases, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark*Corresponding author: Terese L. Katzenstein, Department of Infectious Diseases, Copenhagen University Hospital, Rigshospitalet, Blegdamsvej 9, DK-2100 Copenhagen, Denmark, Tel: +45 35451492; E-mail: terese.katzenstein@regionh.dk
Background: In many resource-limited settings, virological monitoring of HIV-infected patients receiving combination antiretroviral therapy (cART) is not routinely available due to the high cost of HIV-RNA measurements. We aimed to examine whether a p24 antigen-based assay can be used as an alternative and cheaper method to monitor HIV treatment.
Methods: Samples collected in conjunction with a study at Muheza Designated District Hospital, Tanzania, investigating possible interactions between HIV drugs and antimalarials, were retrospectively analysed for HIV load (HIV-RNA and p24 antigen). HIV-infected patients, who had received cART>180 days were included in this cross sectional study. Plasma p24 antigen levels were measured by PerkinElmer p24 ultrasensitive assay and HIV-RNA by COBAS® AmpliPrep/COBAS® TaqMan® HIV-1 test. We analysed the use of p24 antigen results as a predictor of cART response, where cART treatment failure was defined as HIV-RNA ≥ 1,000 copies/mL.
Results: Two-hundred-and-sixty-six patients, who had been on cART for a median of 2.7 years (IQR: 1.7-3.8), were included. Eighty-eight percent had HIV-RNA<1,000 copies/mL and 40% had undetectable p24 antigen. There was only a weak correlation between HIV-RNA and p24 antigen (R2 =0.35), but undetectable p24 antigen had a high negative predictive value for treatment failure (99%). If only positive p24 antigen tests were re-analysed by PCR technique, costs for treatment monitoring could be substantially reduced.
Conclusion: The p24 antigen assay might be useful to improve access to virological monitoring of HIV-infected patients in resource-limited countries.
p24 antigen; HIV-RNA; Virological failure; Treatment monitoring
Human immunodeficiency virus (HIV) infection remains a worldwide problem [1]. Increased availability of combination antiretroviral therapy (cART), even in resource-limited settings, accelerates the need for affordable tools to regularly monitor treatment of HIV-infected patients. Monitoring helps to ensure successful treatment outcomes and reduce development of cART resistance [2,3]. In resource-replete countries HIVRNA is usually measured routinely at intervals between 3 to 12 months to monitor virological response to treatment. In many resource-limited countries, such as Tanzania, viral load monitoring is only available in a few places, and many sites therefore have to rely on clinical and CD4+ T-cell count monitoring [4,5]. Low sensitivity and low positive predictive value for identifying patients with virological failure are the results [6- 8]. This can lead to both unnecessary cART switches and increased levels of drug resistance, which could be avoided if cART monitoring by viral load measurements were practiced [9,10]. A retrospective cohort study from Botswana showed that between 10 and 39% of the population with detectable viremia would go unnoticed if only CD4+ T-cell count was used to monitor for treatment failure [6]. A very recent publication from Ifakara, Tanzania found the sensitivity of WHO’s criteria for immunological failure to be “alarmingly low”; among children and adolescents only five and 14% of those having treatment failure (HIV-RNA>1,000 copies/mL) were correctly identified [5]. Measuring p24 antigen levels might serve as a cheaper and more feasible alternative to HIV-RNA for the routine monitoring of HIV-infected patients receiving cART in resource-limited countries [11,12].
We aimed to examine whether p24 antigen might serve as a sensitive alternative tool to improve virological monitoring of HIV-infected patients on cART in a resource-limited setting
This is a cross-sectional study, based on patient data and samples collected as part of the InterACT clinical trial, which examined interactions between antimalarial and antiretroviral treatment for adult patients coinfected with malaria and HIV-1 [13]. The InterACT study was conducted between July 2009 and October 2012 at the HIV Care and Treatment Clinic (CTC) at the Muheza Designated District Hospital, Northern Tanzania. It examined clinical and pharmacokinetic interactions between the nonnucleotide reverse transcriptase inhibitors (NNRTI) nevirapine (NVP) and efavirenz (EFV) and artemether-lumefantrine (Coartem® ) [13].
Muheza Designated District Hospital has a capacity of 330 beds and provides medical services to a catchment population of approximately 300.000 people. Antiretroviral treatment is provided free of charge through the National HIV Programme of the Ministry of Health, funded through a grant from the United States President’s Plan for AIDS Relief (PEPFAR). One month’s supply of cART is handed out at personal attendance at the hospital’s HIV CTC.
Data on patient demographics and treatment history were collected by patient interviews and by extraction from the medical records.
Only patients enrolled in the InterACT study until November 2010, and who had received cART for at least 180 days, were included in the current analysis. All patients were malaria microscopy negative at the time of blood sampling. Patients with malaria at the day of enrolment had analyses performed on blood samples collected on day 42 after initiation of malaria treatment, while the patients enrolled without malaria had blood collected and analysed on day 0.
All participants gave written informed consent after oral and written information about the InterACT study had been provided. Furthermore, the participants agreed that the samples collected for the main study could be used for supplementary research projects. The study received national ethical approval from the Medical Research Coordinating Committe (MRCC), Minsitry of Health, Dar es Salaam (reference NIMRlHQ/ R.8alVol. IX/1150). The InterACT trial is registered with ClinicalTrials. gov, registration number NCT00885287.
Blood samples were processed within 15 minutes of collection and plasma samples were stored at -80°C [14].
The p24 antigen was measured by the PerkinElmer p24 ultrasensitive assay protocol (Perkin Elmer, Boston, MA) at the AIDS laboratory, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark. The analyses were performed according to the manufacturer’s instructions, though modified by the addition of a virus disruption buffer and nine standard concentrations (between 0.17 and 1111 pg/mL) developed at the Swiss National Centre for Retroviruses [15].
The assay result was determined by an end-point absorbance reading of p24 antigen on an Anthos 2010 reader (Anthos Labtec Instruments, Wals, Austria). A lower limit of detection (LLD) was calculated for each of the four runs i.e. the LLD varied for the different runs [14].
Plasma levels of HIV-RNA were measured by real time polymerase chain reaction (PCR), COBAS® AmpliPrep/COBAS® TaqMan® HIV-1 test, v2.0, (Branchburg, New Jersey, US) according to the manufacturer’s instruction at the AIDS Laboratory, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark.
The CD4+ T-cell counts were analysed by a FACS-counter (Becton Dickinson, San Jose, Ca. USA) at the study site in Tanzania immediately after sampling.
We made a direct cost analysis based on Danish market prices for reagents for measuring HIV-RNA (year 2014) and p24 antigen (year 2012). In order to obtain the lowest prices, 22 samples were analysed simultaneously by PCR and 83 samples were analysed at each p24 antigen run. Importantly, salaries for laboratory personnel and costs of the machines were not taken into account. On average laboratory personnel used 7.7 hours on one run of p24 antigen, while 6.3 hours were used on HIV-RNA analyses. Cost analyses are calculated as USD per analysed sample.
All statistical calculations were done with STATA version 11.0 (Stata corporation, Texas, USA). Groups were compared using chi-squared test for categorical variables and Mann-Whitney test for continuous variables. Multivariate analyses were conducted by logistic regression and all variables were included. A p-value<0.05 was considered statistically significant. Concentrations of HIV-RNA and p24 antigen were log10-transformed to approximate normal distribution [14]. The fitted line in the scatterplot was made by linear regression model based on all measurements [14]. We used WHO’s threshold for treatment failure (HIV-RNA ≥ 1,000 copies/ mL) [16-18], but for the purpose of this study, we, however, considered only one HIV-RNA measurement ≥ 1,000 copies/mL as treatment failure (as opposed to the recommended confirmed HIV-RNA>1,000 copies/mL).
Two hundred sixty-six HIV-infected patients on cART were enrolled. Characteristics of the study population are summarized in table 1. Two hundred thirty-four out of 266 (88%) had HIV-RNA<1,000 copies/mL and 106/262 (40%) had undetectable p24 antigen. The median duration of cART was 2.7 years (IQR: 1.7-3.8). More than four out of five of the study population lived in rural areas. p24 antigen was measured on 262/266 HIV-infected patients, four patients did not have plasma available for analyses, while CD4+ T-cell count was available for 260 patients.
| All (n=266) | |
| Gender | |
| Female | 206 (77%) |
| Age, years (median (IQR)) | 40.5 (35-46) |
| <35 | 51 (19%) |
| 35-50 | 189 (71%) |
| >50 | 26 (10%) |
| BMI (median (IQR))* | 21 (20-24) |
| <19 | 51 (19%) |
| 19-25 | 164 (62%) |
| >25 | 48 (18%) |
| Education | |
| No | 62 (23%) |
| Yes1 | 204 (77%) |
| Employment | |
| Peasant | 179 (67%) |
| Business | 81 (30%) |
| Unemployed or housewife | 6 (2%) |
| Marital Status | |
| Married or cohabit | 120 (45%) |
| Divorced, widowed or single | 146 (55%) |
| Residence | |
| Rural | 218 (82%) |
| Periurban or urban | 48 (18%) |
| Duration of cART2 (median (IQR)) | 2.7 (1.7-3.8) |
| ½ - 1 year | 25 (9%) |
| 1-4 years | 180 (68%) |
| >4 years | 61 (23%) |
| cART2 regimen | |
| d4t, 3TC and NVP | 112 (42%) |
| ZDV, 3TC and EFV | 77 (29%) |
| ZDV, 3TC and NVP | 66 (25%) |
| TDF, FTC and EFV | 8 (3%) |
| d4T, 3TC and EFV | 3 (1%) |
| CD4 (median (IQR))** | 414 (279-572) |
| <200 cells/microliter | 33 (13%) |
| 200-349 cells/microliter | 64 (25%) |
| 350-500 cells/microliter | 75 (29%) |
| >500 cells/microliter | 88 (34%) |
| HIV-RNA | |
| <50 copies/mL | 195 (73%) |
| 50-1,000 copies/mL | 39 (15%) |
| 1,000-100,000 copies/mL | 19 (7%) |
| >100,000 copies/mL | 13 (5%) |
| p24 antigen (median (IQR))*** | 3.88 (2.63-6.6) |
| p24 antigen < LLD3 | 106 (40%) |
| p24 antigen >= LLD3 | 156 (60%) |
Table 1: Demographical characteristics, body mass index and measures of response to antiretroviral therapy. n(%) unless otherwise specified.
*Missing data on three patients (1.1%)
**Missing data on six patients (2.2%). In nine cases (3.3%) the CD4+ T-cell count was unavailable at the day of sampling for viral load measurement, why the nearest CD4+ T-cell count was used (+/- 3 months).
***Missing data on four patients (1.5%)
1 Education=98% finished primary school, 1.5% finished secondary school and 0.5% had a higher education.
2 cART=combination antiretroviral therapy
3 LLD=lower limit of detection
d4T=Stavudine; 3TC=Lamivudine; NVP=Nevirapine; ZDV=Zidovudine; EFV=Efavirenz; TDF=Tenofovir; FTC=Emtricitabine
The LLD values of p24 antigen from the four runs were: 3.01, 3.27, 3.64 and 2.76 pg/mL, respectively. There was a weak correlation between HIV-RNA and p24 antigen levels (R2 =0.35, Figure 1). All except one of the 106 patients with p24 antigen <1,000 copies/ mL. The p24 antigen was detectable among 125/230 (54%) and 106/192 (55%) of patients with HIV-RNA<1,000 copies/mL and <50 copies/mL, respectively. The p24 antigen level was higher among patients with HIVRNA ≥ 1,000 copies/mL versus <1,000 copies/mL (median 14.5 pg/mL (IQR: 5.89 - 83.7) versus 3.5 pg/mL (IQR: 2.43 - 5.32)), p <0.0001.
The proportion of patients with HIV-RNA ≥ 1,000 copies/mL was higher among HIV-infected patients with CD4+ T-cell count <200 cells/ mm3 compared to individuals with CD4+ T-cell counts ≥ 200 cells/mm3 (39.4% versus 7.5%), p<0.001, but there was no correlation between HIVRNA and CD4+ T-cell count (R2 =0.12, n=260).
The p24 antigen level was slightly higher among patients with CD4+ T-cell count <200 versus ≥ 200 cells/mm3 (median 4.3 pg/mL (IQR: 2.85- 12.6) versus 3.88 pg/mL (IQR: 2.54-6.11)), but the difference was not statistically significant (p=0.12). There was no correlation between p24 antigen and CD4+ T-cell count (R2 =0.06, n=256).
In two separate analyses we examined risk factors for HIV-RNA ≥ 1,000 copies/mL (n=266) and detectable p24 antigen (n=262), respectively (Table 2). The risk of having detectable p24 antigen was associated with age <35 years, rural residence and longer duration of cART, while age <35 years was associated with HIV-RNA ≥ 1,000 copies/mL. There were no associations with gender, education or marital status.
| Risk factors | HIV-RNA | p24 antigen | ||||||
| ≥ 1,000 copies/mL N=32 (%) |
<1,000 copies/mL N=234 (%) | Univariate p-values | Multivariate OR (95% CI) | ≥ LLD N=156 (%) |
<LLD N=106 (%) | Univariate p-values | Multivariate OR (95% CI) | |
| Gender | ||||||||
| Female | 24 (75) | 182 (78) | 0.7 | 1.00 | 120 (77) | 82 (77) | 0.9 | 1.00 |
| Male | 8 (25) | 52 (22) | 1.33 (0.52-3.40) | 36 (23) | 24 (23) | 0.98 (0.51-1.85) | ||
| Age, years (median (IQR) | 35.5 (32-40.5) | 41 (36-47) | 0.003 | 40 (35-45) | 42 (37-47) | 0.04 | ||
| <35 | 13 (41) | 38 (16) | 1.00 | 38 (24) | 13 (12) | 1.00 | ||
| 35-50 | 18 (56) | 171 (73) | 0.33 (0.14-0.77) | 105 (67) | 80 (75) | 0.44 (0.21-0.90) | ||
| >50 | 1 (3) | 25 (11) | 0.14 (0.02-1.15) | 13 (8) | 13 (12) | 0.40 (0.14-1.14) | ||
| Education | ||||||||
| Yes 1 | 24 (75) | 180 (77) | 0.8 | 1.00 | 122 (78) | 78 (74) | 0.4 | 1.00 |
| No | 8 (25) | 54 (23) | 1.32 (0.52-3.35) | 34 (22) | 28 (26) | 0.88 (0.47-1.63) | ||
| Employment | ||||||||
| Peasant | 19 (59) | 160 (68) | 0.01 | 1.00 | 103 (66) | 72 (68) | 0.1 | 1.00 |
| Business | 10 (31) | 71 (30) | 1.52 (0.61-3.81) | 47 (30) | 34 (32) | 1.23 (0.64-2.35) | ||
| Other2 | 3 (9) | 3 (1) | 7.62 (1.24-46.96) | 6 (4) | 0 (0) | (omitted) | ||
| Marital Status | ||||||||
| Not couple | 16 (50) | 130 (56) | 0.6 | 1.00 | 85 (54) | 60 (57) | 0.7 | 1.00 |
| Couple | 16 (50) | 104 (44) | 1.22 (0.55-2.72) | 71 (46) | 46 (43) | 1.14 (0.66-1.95) | ||
| Residence | ||||||||
| Periurban or urban | 3 (9) | 45 (19) | 0.2 | 1.00 | 21 (13) | 27 (25) | 0.01 | 1.00 |
| Rural | 29 (91) | 189 (81) | 3.36 (0.82-13.82) | 135 (87) | 79 (75) | 2.38 (1.11-5.12) | ||
| Years of cART (median (IQR)) | 2.90 (2.00-4.53) | 2.70 (1.69-3.80) | 0.3 | 2.79(1.85-3.77) | 2.55(1.47-4.07) | 0.05 | ||
| ½ - 1 | 1 (3) | 24 (10) | 1.00 | 10 (6) | 15 (14) | 1.00 | ||
| 1 - 4 | 21 (66) | 159 (68) | 2.83 (0.35-23.15) | 113 (72) | 64 (60) | 2.50 (1.02-6.13) | ||
| >4 | 10 (31) | 51 (22) | 4.79 (0.55-41.92) | 33 (21) | 27 (25) | 1.72 (0.63-4.68) | ||
Table 2: Risk factors for HIV-RNA ≥ 1,000 copies/mL or detectable p24 antigen, respectively. n(%) unless otherwise specified.
Missing data on four patients (p24 antigen)
All variables were included in the multivariate analysis
OR: Odds Ratio; CI: Confidence Interval; LLD: Lower Limit of Detection
1 Education=98% finished primary school, 1.5% finished secondary school and 0.5% had a higher education
2 Other=Unemployed and housewives
cART=combination antiretroviral therapy
We estimated the sensitivity, specificity, negative and positive predictive value of p24 antigen assay for identifying patients with treatment failure in three separate analyses using different cut-offs of HIV-RNA to define treatment failure (50, 400 and 1,000 copies/mL, respectively, Table 3). The specificity of p24 antigen assay was rather low in all analyses, but the assay had a high sensitivity for identifying individuals with HIV-RNA ≥ 1,000 copies/mL (97%) and the negative predictive value of an undetectable p24 antigen test was high 99% (CI: 95-100%).
| HIV-RNA | |||
| 50 copies/mL | 400 copies/mL | 1,000 copies/mL | |
| HIV-RNA above cut off | 27% (21%-33%) | 14% (10%-19%) | 12% (9%-17%) |
| Sensitivity | 71% (59%-82%) | 86% (71%-95%) | 97% (84%-99%) |
| Specificity | 45% (38%-52%) | 45% (38%-52%) | 46% (39%-52%) |
| Positive predictive value | 32% (25%-40%) | 21% (14%-28%) | 20% (14%-27%) |
| Negative predictive value | 81% (72%-88%) | 95% (89%-98%) | 99% (95%-100%) |
Table 3: Sensitivity, specificity and predictive values (95% confidence interval) of the p24 antigen assay for detecting treatment failure in three separate analyses using different cut-off values of HIV-RNA to define “treatment failure”.
The direct costs of analysing one sample by HIV-RNA and p24 antigen were 82 USD and 11 USD, respectively. If the p24 antigen assay was used as a screening tool and patients with detectable p24 antigen were further tested for HIV-RNA, the average cost associated with identifying patients with virological failure (defined as HIV-RNA ≥ 1,000 copies/mL [16]) (using Danish prices for reagents) was 60 USD per patient with treatment failure compared to 82 UDS if all patients were tested by HIV-RNA (Figure 2). In this setting the costs of reagents for virological monitoring would decrease by 27% (2,900 USD for p24 antigen measurements + 12,800 USD for HIV-RNA measurements versus 21,500 USD for HIVRNA measurements in a population of 262 patients) and only less than one percent would be misclassified (Figure 1).
We found that p24 antigen testing can be used to monitor for virological failure among HIV-infected patients treated with cART. When we used HIV-RNA ≥ 1,000 copies/mL as virological failure marker, as stipulated by WHO guidelines, undetectable p24 antigen had a high negative predictive value (99%) [16,18]. As the WHO definition calls for two consecutive HIV-RNA >1,000 copies/mL our failure rate is most likely an overestimation. Among the 266 patients treated with cART >180 days, 88% had HIV-RNA <1,000 copies/mL, while 40% had undetectable p24 antigen. Given the relatively low costs of p24 antigen testing, this option might be applicable also in resource-limited settings, where the high costs of standard HIV-RNA testing methods may preclude this option for routine cART monitoring due to the need to split sparse resources among numerous essential health needs.
We found that an undetectable p24 antigen level had a high negative predictive value for virological failure among HIV-infected patients on long-term cART. Our results indicate that p24 antigen can be used as a screening test for virological failure, with HIV-RNA 1,000 copies/mL as the threshold. If the p24 antigen screening test is positive (>LLD), a subsequent HIV-RNA measurement should be performed. In places where dried blood spots are used to monitor HIV-infected patients, it should be easy to implement p24 antigen measurement on plasma, because there is developed a plasma separator that separate capillary blood to plasma [19].
A study by Schüpbach et al. found that p24 antigen was unreliable at detecting HIV-RNA rebound when HIV-RNA was between 400 and 1,000 copies/mL [12]. Both our sensitivity analysis (Table 3) and scatter plot (Figure 1) concur with this finding. In 2013 WHO described the rationale of monitoring HIV-infected patients with a treatment failure threshold of 1,000 HIV-RNA copies/mL: Firstly, intermittent low-level viremia (50- 1,000 HIV-RNA copies/mL), which might occur during efficient cART treatment, is not associated with an increased risk of treatment failure, unless the low-level viremia is persistent [16]. Secondly, the risk of HIV transmission and disease progression is minimal when viral load is <1,000 copies/mL [16,20]. Recent data, however, indicate that low-level viremia might be a risk factor for subsequent treatment failure and, hence, the cutoff value might in the future be adjusted down from 1,000 copies/mL [21].

Figure 1: Scatter plot illustrating correlations between plasma p24 antigen and HIV-RNA measurements. Grey circles: undetectable p24 antigen. Black squares: detectable p24 antigen.
In the Tanzanian national guidelines for monitoring of HIV and AIDS, 2012, virological treatment failure was defined as HIV-RNA persistently ≥ 5,000 copies/mL [4]. All patients in the study with ≥ 5,000 HIV-RNA copies/mL had measurable p24 antigen (Figure 1). Another study on HIVinfected patients found a considerably weaker negative predictive value at a threshold of 5,000 copies/mL [22]. The reason for this discrepancy is not clear, but might be explained by the relatively lower prevalence of treatment failure in our setting. Further studies are warranted.
Our direct cost analysis, based on Danish prices for reagents, showed important savings (Figure 2) if all positive p24 antigen screening tests were re-measured by PCR technique. The saving would be considerably greater if the costs of the machines/hardware were included. Some manufacturers offer cheaper viral load assay prices for resource-limited settings; Roche sells HIV-RNA tests at 20-30 USD, but corresponding p24 antigen prices are only 5-15 USD [19,23,24]. The p24 antigen method is easy to implement with basic ELISA laboratory equipment and a short training period [15]. An obvious limitation would be the need of PCR analysis for patients with a positive p24 antigen test.
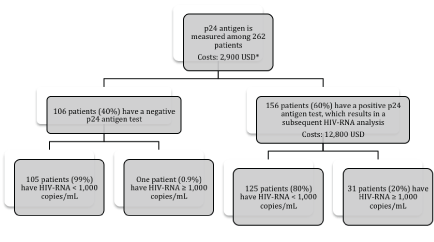
Figure 2: Flow chart illustrating a suggested algorithm for virological monitoring of HIV-infected patients on cART and the associated direct reagent costs in resource-limited settings.
*Total costs of HIV-RNA (n = 262) patients would be 21,500 USD.
We analysed treatment response among 266 HIV-infected patients who had been treated with cART for >180 days and found that 88% had suppressed HIV-RNA at a quantification limit of 1,000 copies/mL and 40% had p24 antigen
Previous studies have shown varying degrees of correlation between HIV-RNA and p24 antigen (R2 =0.11-0.69). These findings could be explained by several factors: Use of Schüpbach’s disruption buffer and possibly other factors such as duration of viral suppression [14,25,26,28,29].
There is an ongoing search for alternative methods for monitoring treatment response, and specifically for early detection for viral failure. The SAMBA point-of-care test represents another easy way to preform monitoring with technology demanding less technical skills [30]. Likewise an ELISA-based method for measuring levels of interferon-γ- inducible protein-10 (IP-10) was recently shown efficient for screening for virological failure among HIV-infected patients followed at a District Hospital in Mozambique [31]. The usage of an immunological, instead of an HIV based test, and the possible influence on the results from other infections/non-infectious health problems will have to be further studied.
We found no correlation between HIV-RNA and CD4+ T-cell count or between p24 antigen and CD4+ T-cell count, which is in accordance with previous studies [14,32]. Available data support that CD4+ T-cell count should be complemented by a marker of viral load for treatment failure monitoring [5,9].
A Tanzanian study from 2014 evaluated the national management of HIV-infected patients as stipulated by a WHO protocol. The protocol is based on four early warning indicators. The country did excellent on prescribing cART (100%), but showed a disappointing 12 months follow-up; with 26% lost to follow-up (goal: ≤ 20%), only 54% taking the appropriate first line regimen (goal: ≥ 70%) and only 38% of patients attending all clinical appointments on time (goal: ≥ 80%) [33]. Thus, Tanzanian HIV-infected patients are inadequately monitored and maybe, implementation of a p24 antigen screening test might help identify patients with treatment failure who need closer monitoring and enhanced support to achieve better adherence.
As confirmed by other studies, we found that patients younger than 35 years had significantly higher risk of having detectable viremia (both HIVRNA and p24 antigen) compared to middle-aged patients [34]. Patients living in rural areas had an increased risk of detectable p24 antigen compared to those living in periurban and urban areas. A systematic review from South Africa suggested that people living in rural areas have less access to cART [35].
In the current study, patients who had been on cART between one to four years had an increased risk of detectable p24 antigen, compared to those being on cART less than one year. They also tended to have increased risk of HIV-RNA ≥ 1,000 copies/mL, but this was not statistically significant. This finding further supports the use of virological monitoring of patients on long-term cART to identify individuals who need more intensive adherence support.
Although we found overall high rates of virological suppression as measured by HIV-RNA, our results cannot be used to estimate rates of treatment success among all HIV-infected patients who initiate cART, as we only included patients who continuously attended the HIV CTC. However, this does not hamper the aim of the study, which was to assess the utility of the p24 antigen test to monitor patients who attended followup. A limitation of this study is the relatively small number of patients participating, making it difficult to draw strong conclusions.
Poor healthcare infrastructure, human resources, and economic constraints challenge the attempt to implement virological monitoring in resource-limited countries [3]. How to implement virological monitoring is still an unanswered question. Several studies have indicated that p24 antigen testing might be an alternative or adjunct to HIV-RNA measurements in resource-limited countries to monitor treatment response [26,27,36]. In 2012, a cheap, portable and easy-touse p24 antigen test with excellent specificity and sensitivity was made for diagnosing HIV infections in infants at bedside by dried plasma spots [19,37]. If an analogous point-of-care instrument could be developed with focus on detecting treatment failure by p24 antigen, it would be a major improvement; with avoidance of advanced laboratory equipment, the dichotomous test results should be easy to understand and the patients could receive test results immediately. A screening test calibrated to have a detection limit corresponding to a HIV-RNA level at 1,000 copies/mL, would be very useful. Larger prospective studies would be needed to assess the usage of such a p24 antigen screening test.
Laboratory technician Margrethe Lüneborg-Nielsen is thanked for her excellent assistance with the virological analyses in Copenhagen. We also wish to sincerely thank the all the involved staff and participating patients in Tanzania.
Furthermore, we wish to acknowledge and thank the founders of this research, the ACT Consortium, founded through a grant from the Bill & Melinda Gates Foundation to the London School of Hygiene and Tropical Medicine.
This work was supported by the ACT Consortium at the London School of Hygiene and Tropical Medicine, through a grant from the Bill & Melinda Gates Foundation under grant number 39640.
The authors declare no competing interests.
Download Provisional PDF Here
Article Type: Research Article
Citation: Melaaen MS, Vestergaard LS, Nyagonde N, Amos B, Francis F, et al. (2017) Use of the p24 Antigen to Evaluate Virological Failure among HIV-1- Infected Patients Receiving Antiretroviral Treatment in Tanzania. J HIV AIDS 3(3): doi http://dx.doi. org/10.16966/2380-5536.144
Copyright: © 2017 Melaaen MS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access