Recently, we showed that untreated patients with HIV infection
display high peripheral blood counts of regulatory B cells expressing
the serine protease granzyme B (GrB) in the absence of perforin (GraB
cells) [1]. Importantly, these GraB cells are able to directly regulate
proliferation and survival of T cells both in-vitro and in- vivo. The
mechanism of action involves a perforin-independent transfer of GrBto
T cells and GrB-dependent degradation of the T cell receptor ζ-chain in
T cells [1,2].
A known receptor for GrB, which acts in a perforin-independent
manner, is the mannose-6-phosphate receptor (M6PR, CD222), which has
been shown to mediate GrB uptake and regulation of M6PR-expressing
target cells [3,4]. A recent study in Listeria-infected mice demonstrated
that the differential expression of M6PR on cytotoxic T cells is directly
linked to their survival and proliferative capacity [5]. M6PR therefore
appears to represent an important check point for T cell expansion and
memory T cell formation after systemic infections.
Here we report our current findings confirming this mechanism in
human patients with untreated HIV infections. Since cellular uptake
of GrB in the absence of perforin can occur in an M6PR-dependent
manner [3,4], we tested the expression of M6PR on T cells from
untreated HIV patients and compared it to healthy controls. These
experiments revealed that M6PR expression by T cells from HIV
patients is significantly higher than by T cells from healthy control
subjects (Figure 1 and Table 1). Moreover, in-vitro transfection of
isolated T cells from healthy subjects with HIV confirmed that the
HIV directly triggers upregulation of M6PR on T cells (Figure 2). Our
data therefore suggest that defects in the memory T cell compartment
of HIV patients may at least in part be due to elevated expression of
M6PR by T cells, associated with a higher sensitivity of these cells to GrBmediated
apoptosis and growth arrest.
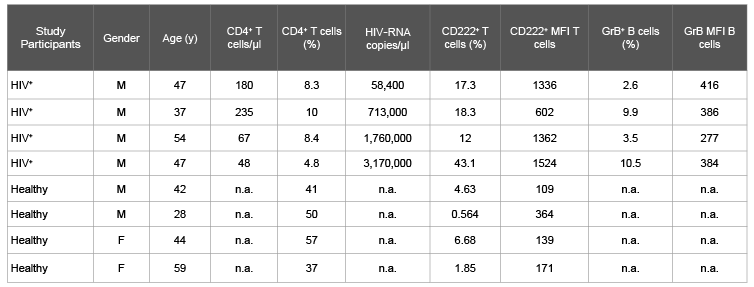
Table 1: Clinical, virological, andimmunologicalcharacteristics of HIV patients and healthy control subjects tested for GraB cells and CD222-expressing
CD4+ T cells.
Abbreviations: F: Female; M: Male; GrB: Granzyme B; MFI: Median fluorescence intensity; n.a., not available.
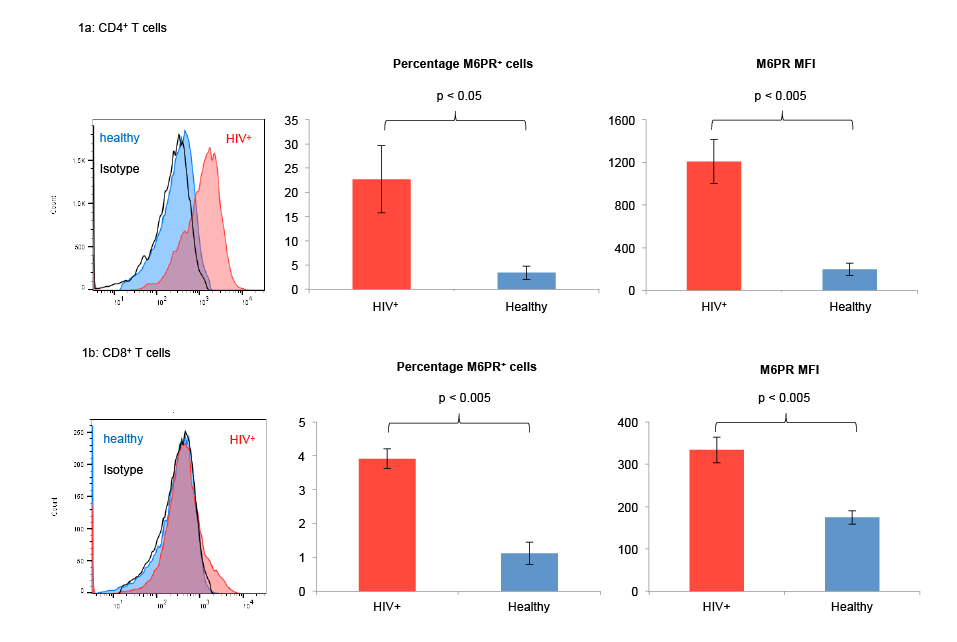
Figure 1: T cells from untreated HIV+ patients express high levels of M6PR as compared with T cells from healthy individuals. PBMC from 4
untreated HIV patients and from 4 healthy control subjects were isolated and stained with fluorescently labeled antibodies against CD3, CD4, CD8 and
M6PR (CD222). Subsequently, CD4+ (Figure 1a) and CD8+ (Figure 1b) T cells were analyzed by flow cytometry. Human peripheral granulocytes served
as positive control for CD222 expression. Histograms show M6PR surface expression from one representative experiment out of 4 with similar results
(left panels). Bar graphs show average percentages of M6PR+ T cells (middle panels) and M6PR median fluorescence intensity (MFI) values (right
panels) from 4 independent experiments. Error bars indicate SEM, *indicates p<0.05, **indicates p<0.005.
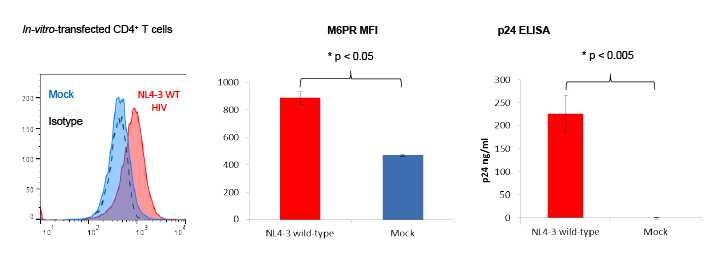
Figure 2: T cells from healthy individuals upregulate mannose-6-phosphate receptor (M6PR) following transfection with wild-type NL4-3 HIV.
CD4+ T cells from 3 healthy individuals were isolated and stimulated with CD3/CD28 dynabeads and IL-2 for 3 days. Cells were washed and transfected
with NL4-3 wild-type (WT) or mock-transfected for 6 hrs at 37°C. Three days post-transfection, cells were stained with fluorescently labeled antibodies
against M6PR (CD222) or an isotype control. Then, T cells were analyzed by flow cytometry. Culture supernatants were tested for p24 protein levels
using an in-house ELISA (Abcam). Histograms show M6PR surface expression from one representative experiment out of three with similar results
(left panel). Bar graphs show M6PR median fluorescence intensity (MFI) values (middle panel) and p24 levels (right panel). Error bars indicate SEM,
*indicates p<0.05.
In summary, our findings support the current view that after infections
with intracellular pathogens such as viruses or intracellular bacteria,
activated T cells differentially regulate M6PR on their cell surface [5].
This differential M6PR expression may not only explain how regulatory
T cells initiate the effector T cell contraction phase after an infection, but
also how other immune cell populations expressing GrB in the absence
of perforin such as plasmacytoid dendritic cells or GraB cells [2,6,7] may
directly suppress T cell expansion in an M6PR- and GrB-dependent fashion
(Figure 3). Modulation of M6PR on T cells by pharmacological means
may represent a promising novel approach to modulate T cell-mediated
immunity in different infectious diseases including HIV infection.
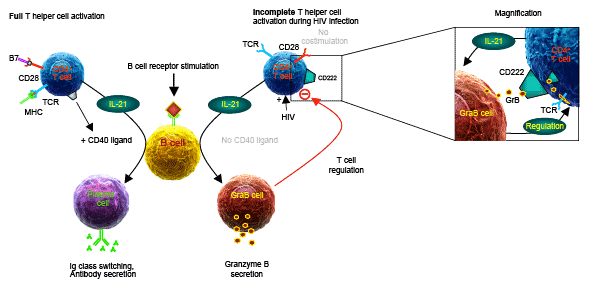
Figure 3: Mannose-6-phosphate receptor (M6PR, CD222) on T cells from HIV patients mediates their suppression by granzyme B-secreting
regulatory B cells (GraB cells). During HIV infection, the T cell receptor (TCR) of CD4+ T cells is directly stimulated via the HIV protein Nef, without
simultaneous costimulation of CD28. In contrast to fully activated T cells (left panel side), such incompletely activated T cells secrete IL-21, but barely
express CD40L, resulting in the induction of granzyme B-secreting GraB cells instead of plasmacells (right panel side, Copyright 2015. The American
Association of Immunologists, Inc.). By concomitant upregulation of CD222 on T cells in the course of an HIV infection, the cellular uptake of exogenous
granzyme B by T cells is strongly enhanced, resulting in increased cleavage of their TCR ζ-chain (magnification panel). Lower TCR ζ-chain levels are
associated with lower proliferative capacity of such T cells. Breaking of this vicious circle may be possible by exogenous addition of CD40L multimers,
which can suppress the generation of GraB cells after incomplete B cell/T cell interactions during HIV infection.
Download Provisional PDF Here
Article Information
Article Type: Short Communication
Citation: Kaltenmeier CT, Gawanbacht A, Hotter D,
Kirchhoff F, Schrezenmeier H, et al. (2016) Mannose-
6-Phosphate Receptor, a Novel Checkpoint for T cell
Expansion, is expressed at High Levels on T cells
from Untreated HIV+ Patients. J HIV AIDS 2(3): doi:
http://dx.doi.org/10.16966/2380-5536.125
Copyright: © 2016 Kaltenmeier CT, et al. This is
an open-access article distributed under the terms
of the Creative Commons Attribution License,
which permits unrestricted use, distribution, and
reproduction in any medium, provided the original
author and source are credited.
Publication history:
Received date: 05 Feb 2015
Accepted date: 27
Apr 2016
Published date: 03 May 2016





