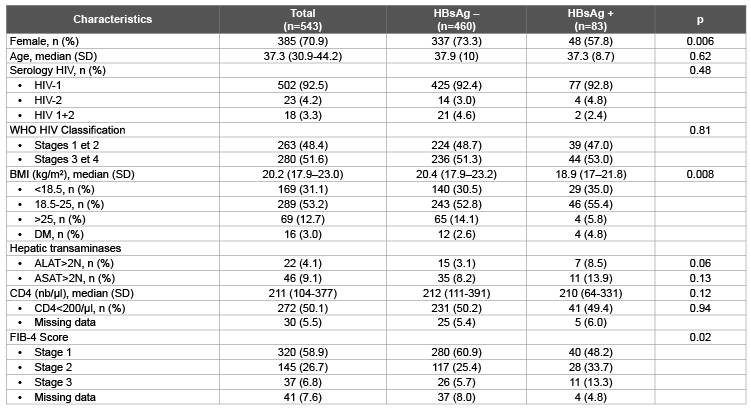
Table 1: Characteristics of Patients at Initial visits by HBsAg status

Sawadogo A1* Hema A2 Koura M1 ILBOUDO BP2 Kamboule BE1 Kabore NF2 Sore I2 Konate A2 Poda GEA2 Zoungrana J2 Sawadogo AB2
1Hepato gastro enterology service, University Hospital Souro Sanou, Bobo-Dioulasso, Burkina Faso*Corresponding author: Appolinaire SAWADOGO, Hepato-gastro-enterologie department at Sourô SANOU University Health Centre (CHUSS), 01 BP 676, Bobo-Dioulasso 01, Burkina Faso, Tel: +226251588; E-mail: drsawadogo.appolinaire@yahoo.fr
Objective: To determine the epidemiological, bioclinical and evolving characteristics of viral hepatitis B infection in HIV-infected patients.
Methods: This is a retrospective longitudinal study carried out at the Bobo-Dioulasso Day Hospital from January 1, 2008 to March 31, 2013. The study included HIV-infected patients aged at least 18 years old, naïve of Antiretroviral Treatment (ART) and who underwent HBsAg testing during the initial visit. The HBsAg was determined by the rapid test Determine (ABBOTT®). The FIB-4 score was used to assess liver fibrosis. The Cox Proportional Hazard model was used to identify factors associated with survival.
Results: The study included 543 patients, of whom 385 (70.9%) were women. The majority of patients (93%) were infected with HIV-1. At the initial visit, the mean age of patients was 37.3 years (IQR 30.9 to 44.2) and the median CD4 count was 211 cells/l (IQR 104-377). HBsAg seroprevalence was 15.3% with a male predominance (22.2% vs 12.5%, p=0.006). HbsAg positivity was associated with a high FIB-4 score. There was no statistically significant difference between the immuno-virological responses to ART between HBsAg positive and HBsAg negative patients. Body Mass Index (BMI)<18.5 and CD4<100 cells/µl at the time of introduction of HAART was associated with higher risk of death. However, HbsAg positivity was not associated with a higher risk of death. Stage 3 liver fibrosis occurrence was significantly associated with higher risk of death in the univariate analysis; however, this association disappeared in the multivariate analysis.
Conclusion: The search for hepatitis B markers and testing for liver damage should be systematic during HIV infection. In the absence of resources to test for HBV, TDF could be systematically included in first-line ART in areas were hepatitis B is highly endemic.
HBV; HIV; Infections; Bobo-Dioulasso; Burkina Faso
HBV and HIV infections share the same modes of transmission and both are treated by antiretroviral medicines. Chronic Hepatitis B infection affects 7 to 10% of HIV infected patients worldwide [1,2]. In studies carried out in developed country, the prevalence of HBV infection is higher in HIV-positive patients (around 10%) than in HIV-negative patients (less than 2%) [3]. In African studies, HBV infection rates are found to be similar in HIV-positive and HIV-negative patients [4,5].
HBV does not seem to have a negative influence on the natural history of HIV, while HIV significantly modifies the natural history of HBV [3]. First, HIV infection impacts the serological expression of HBV and its risk of chronicity, then it increase the development of liver disease and HBV infection mortality [6]. In fact, HIV infection accelerates HBV infection complications such as the progression of hepatic fibrosis, cirrhosis and hepatocellular carcinoma [7]. Thus testing for liver damage during the dual infection HIV/HBV should be systematic. Liver biopsy remains the preferred method of evaluation, but the invasive nature and the cost of this procedure are not well adapted to routine examination. Different non-invasive markers of hepatic fibrosis, such as FIB-4 score or APRI index have been developed and are better adapted to the context of countries with limited resources.
It is important for people living with HIV to be aware of their hepatitis B status in order to initiate a treatment combining medicines effective against the two viruses, so they can prevent the development of HBV resistance due to mutations. Furthermore, HBV/HIV co-infection is a factor in low response to antiretroviral treatment for chronic active hepatitis B [3]. HIV infection, hepatitis B and antiretroviral treatment (ART) could constitute hepatitic aggressive factors, leading to failure of antiretroviral treatments. However, the effect of HBV infection on the therapeutic response to HIV antiretroviral treatments is not well documented in sub-Saharan Africa.
Classified by WHO among the countries with the highest prevalence of HBV (>8%), acute hepatitis B causes 1,100 deaths annually in Burkina Faso [8]. The prevalence of HBV in HIV-positive patients is comparable to the prevalence of the general population [9].
This study describes the epidemiological, biological and evolving characteristics of adults coinfected with HIV and HBV receiving ART at the CHUSS Day Hospital in Bobo-Dioulasso (Burkina Faso).
This is a retrospective longitudinal study that was carried out at BoboDioulasso Day Hospital from January 1, 2008 to March 31, 2013. The Day Hospital specialized in treating adults infected with HIV, receives exclusively outpatients.
The source population includes patients infected with HIV who visited the clinic during the period of the study. Patients who were at least 18 years old, naïve of treatments and who underwent systematic testing for HBsAg during initial consultations were included.
The collection of clinical and biological data was carried out by physicians during routine bi-annual check-ups with HIV patients using software ESOPE®.
HBsAg were systematically searched using Determine® (ABBOTT®) rapid test each time the reactive was available. Liver damage was evaluated based on the FIB-4 score, a bio-marker for hepatic fibrosis, using the following formula:
$${{Age \times ASAT} \over {plaquette \times \sqrt {ALAT} }}$$
According to the FIB-4 score, the degree of hepatic fibrosis was classified according to the three categories: FIB-4 score ≤ 1.45: no or minimal fibrosis; FIB-4 score [1.46-3.25]: moderate fibrosis; FIB-4 score>3.25: severe fibrosis.
The statistical analyses were carried out using Stata 12 software. Patients’ socio-demographic, clinical and biological characteristics collected during initial visits were described as qualitative variables using numbers and percentages. Median and interquartile range (IQR) were used to describe quantitative variables. Fisher exact test and Student test were used to compare qualitative and quantitative variables. Occurrence of attrition was described by cause (death, transfer, loss of vision). Cox’s proportional risk method (univariate and multivariate) was used to determine factors associated with survival. The statistical analyses had a significance level of 5%.
During the period of the study, 2,901 new patients attended the HIV clinic. Among them, 543 patients (18.7%) benefited from HBsAg serological testing and were all included in the study.
Among the 543 patients who were included in the study, 385 (70.9%) were women. The median aged was 37.3 years (IQR: 30.9-44.2). The vast majority of patients (92.5%) were HIV infected and HIV-1 was the predominant serotype. Based on WHO classification for HIV infections, 150 patients (27.6%) were at stage 1; 113 (20.8%) at stage 2 and 238 (43.9%) at stage 3 and 42 (7.7%) at stage 4. The median for BMI was 20.2 kg/m2 (IQR: 17.9-23.2) and 31.1% of patients had a BMI less than 18.5 kg/ m2 . The median number of CD4 cells was 211 cells/µl (IQR; 104-377), and more than half of the patients had less than 200 CD4 cells/µl.
Based on FIB4 score, one third of patients had stage 2 hepatic fibrosis (26.7%, n=145) or stage 3 (7.6%, n=41), and among them 4% had significantly elevated alanine aminotransferase (ALT) (>2N). Table 1 provides socio-demographic and clinic-biological characteristics for patients at the time of their initial visits.
During the initial biological check-up, 83 of 543 HIV infected patients (15.3%) were positive for HBsAg. Men were more likely to be HBsAg positive (22.2% compared to 12.5%, p=0.006) than women. However, age, WHO stage of infection, HIV type and number of CD4 lymphocytes were not associated with being positive for HBsAg. Hepatic cytolysis was more frequent (ALT>2N) in HBsAg-positive patients. HBsAg positivity was also associated with elevated FIB-4 scores (Table 1).

Table 1: Characteristics of Patients at Initial visits by HBsAg status
Among the 543 patients naïve of ART included in the study, 372 patients (68.5%) secondarily received ART. Furthermore, we recorded 57 (10.5%) deaths, 7 of whom were without ART (p=20). Additionally, 14 patients (2.6%) were transferred to another treatment center for their convenience, 3 of whom were without treatment. Moreover 98 patients (18%) were lost of follow-up. The latter were mostly patients who were not receiving treatment (n=66, P<10-3). The following analyses only take into account patients under ART due to the high attrition rate (57.6%) of patients not under ART before the sixth month.
The combination of zidovudine (AZT), lamivudine (3TC) and efevirenz (EFV) was most often prescribed (50.5%, n=188), followed by the association of tenofovir (TDF), emtricitabine (FTC) (or 3TC) and EFV or nevirapine (NVP) (34.1%, n=127). The least prescribed treatment was the combination of TDF, FTC (or 3TC) and lopinavir/ritonavir (LPV/r) (4.3%, n=16). At the beginning of ART, 41.1% of HBsAg-positive patients were on ART, with a single molecule that is active on HBV (3TC) and 58.9% had two molecules in their therapeutic protocol that were active on the virus (3TC+TDF or TDF+FTC).
In the 6th month (M6), of follow-up, the median BMI in HBsAgpositive patients increased by 1.4 kg/m2 and 1.2 kg/m2 in HBsAg-negative patients (p=0.26). In the 12th month (M12), HBsAg-positive patients’ median BMIs increased by 1.4 kg/m2 , and for HBsAg-negative patients, median BMIs increased by 1.9 kg/m2 (p=0.95). At the 24th month (M24), the median BMI in HBsAg-positive patients increased by 1.8 kg/m2 and 4.5 kg/m2 in HBsAg-negative patients (p=0.02).
At M6, the median increase in CD4 cells for HBsAg-positive patients was 96 cells/µl and 106 cells/µl for HBsAg-negative patients (p=0.48). At M12, the median increase in CD4 cells for HBsAg-positive patients was 140 cells/µl and 137 cells/µl for HBsAg-negative patients (p=0.83). At M24, the median increase in CD4 cells for HBsAg-positive patients was 180 cells/µl and 186 cells/µl for HBsAg-negative patients (p=0.91).
Among 372 patients monitored, 208 received HIV-1 plasmatic viral load charge during M12 and 20 had a detectable viral load (>300 copies/ µl). The proportion of virologic failure was 6.5% (2/31) in HBsAg-positive patients compared to 10.2% (18/177) in HBsAg-negative patients. The difference between the proportions was not statistically significant (p=0.39).
At the beginning of ART, the proportion of patients with stage 3 fibrosis was higher in HBsAg-positive patients than HBsAg-negative patients (21.4% compared to 9.2%, p=0.88), and comparable in the two groups during the follow-up period (Figure 1).
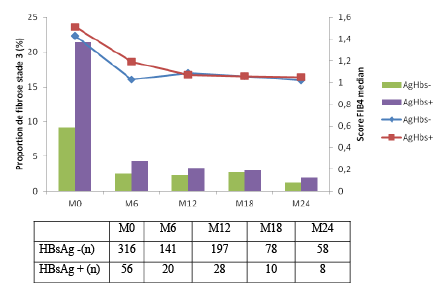
Figure 1: Evolution of FIB-4 score and proportion of stage 3 fibrosis in people living with HIV by HBsAg positivity
At the beginning of the treatment, the median FIB-4 score in HBsAgpositive and HBsAg-negative patients was comparable (1.51 compared to 1.43, p=0.27).
A constant decrease in median FIB-4 score was observed during the treatment in the two groups. At M6, the median FIB-4 score for HBsAgpositive patients decreased by 0.31, and by 0.28 for HBsAg-negative patients (p=0.54). At M12, the median FIB-4 score for HBsAg-positive patients decreased by 0.38, and by 0.21 for HBsAg-negative patients (p=0.21). At M18, the median FIB-4 score for HBsAg-positive patients decreased by 0.31, and for HBsAg-negative patients, it decreased by 0.13 (p=0.55). At M24, the median FIB-4 score for HBsAg-positive patients decreased by 0.23, and for HBsAg-negative patients, it decreased by 0.17 (p=0.83).
Atthe end of the follow-up period, 50 patients on ART were dead, 12 of them were lost to follow-up, and 16 were transferred.
The mortality rate for patients under ART was 9.6 deaths per 100 patients per year (95% CI 7.3-12.7). For HBsAg-negative patients, the mortality rate was 10.2 deaths per 100 patients per year (95% CI 7.7-13.7), and for HBsAg-negative patients, the mortality rate was 5.8 deaths per 100 patients (p=0.27).
In the univariate analysis, the mortality rate increased with age; the risk of death was 6 times higher for those over the age of 55 than those between 26 and 35 years (RR=6.19, 95% CI 2.17-17.67). The probability of death was statistically associated with low BMI (BMI<18.5) at the beginning of treatment (RR=3.38, 95% CI: 1.84-6.18). HBsAg-positivity was not associated with a higher risk of death (RR=0.54, 95% CI: 0.19-1.49). The risk of death was almost 3 times higher for patients with less than 100 TCD4 lymphocytes (RR=2.84, 95% CI 1.45-5.58), and 2.6 times higher for those with FIB-4 scores>3.25 compared to those with FIB-4 scores<1.45 (RR=2.59 95% CI: 1.31-6.56) at the beginning of ARV treatment (Table 2).
In the multivariate analysis, only older age (RR=3.77, 95% CI: 1.11- 12.74), BMI<18.5 (RR=2.37, 95% CI: 1.19-4.69) and number of CD4 ≤ 100 cells/µl (RR=2.57, 95% CI: 1.17-5.61) were significantly associated with a high risk of death (Table 2).
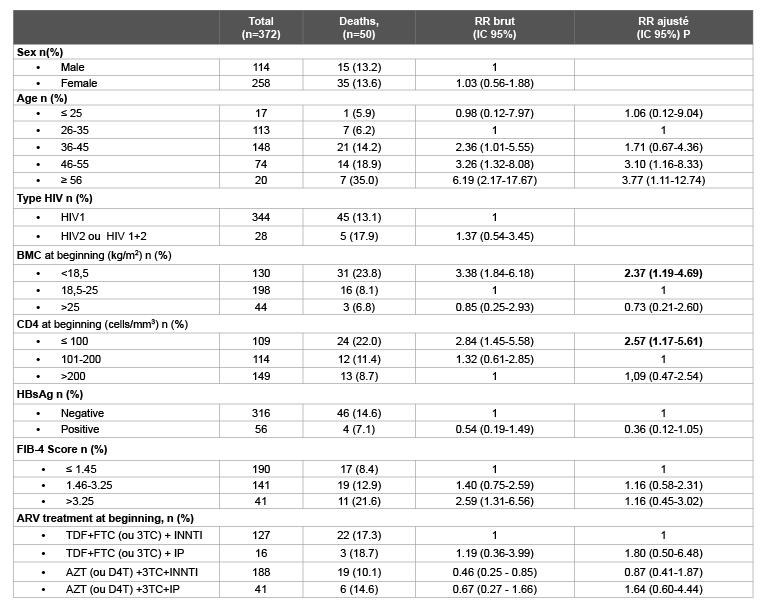
Table 2: Factors correlated with mortality
From January 1, 2008 to March 31, 2013, we carried out a longitudinal retrospective study to evaluate the prevalence and clinico-biological impact of HBsAg positivity in people living with HIV.
During the study, only 18.7% (n=543) of HIV infected patients referred to the Day Hospital in Bobo-Dioulasso for care received systematic HBsAg testing. Among 543 patients included in the study, HBsAg seroprevalence was 15.3%. HBsAg positivity was higher in men than women (22.2% vs 12.5%, p=0.006). HBsAg positivity was also associated with high FIB-4 scores (P=0.02).
In total, 372 patients were on ART. The immune-virologic responses to ART were not statistically significant between HBsAg-positive and – negative patients. BMI<18.5 and CD4<100 cells/µl were associated with higher risk of death (p<10-3). However, HBsAg positivity was not associated with higher risk of death. The occurrence of stage 3 hepatic fibrosis was significantly associated with higher risk of death in the univariate analysis. This relationship disappeared in the multivariate analysis.
In our study, HBsAg seroprevalence was comparable with those reported by some African studies [4] and to the general population in Burkina Faso [10]. In the majority of African studies, HBV infection prevalence in people infected with HIV is similar to HBV infection in the general population not infected by HIV [3,5,11]. However, in European studies, the prevalence of hepatitis B in people living with HIV is higher than the one in the general population [3]. Hepatitis B transmission in sub-Saharan Africa seems to be mainly horizontal before procreative age [12]. HIV/HBV co-infection would thus be a result of early contact during childhood with HBV and a later contact for HIV.
Male predominance of HBsAg positivity was documented in several studies on HIV/HBV co-infection [3,4,13,14]. One hypothesis suggests that the social environment constitute the origin of high hepatitis B transmission rates in boys, as they tend to be involved in higher risk behaviors than girls during their childhood.
During HIV/HBV co-infection, the HIV infection accelerates the rate of progression of fibrosis and the development of cirrhosis and hepatocellular carcinoma [7], hence the higher FIB-4 score in HBsAg-positive patients. However, hepatic fibrosis is documented in people living with HIV free of HBV or HVC infections, and in that case, the hepatic fibrosis is positively correlated with HIV viral load [15-17]. Mendeni et al. [18] demonstrated that the low HIV plasmatic viral load (<500 copies/% l) is a factor in limiting the progression of the FIB-4 score. Therefore, ART not only decreases HIV and HBV viral loads and restores immunity, but also stops the progression of hepatic fibrosis. Furthermore, some molecules, such as TDF, are known to have an impact on the histological improvement of hepatic fibrosis [19].
Weight loss during HIV infection is often related to chronic diarrhea or tuberculosis and the reduced number of TCD4 lymphocytes, which are common causes of death while under ART [17,20,21].
Several studies show that hepatitis B is a factor independently related to higher risk of death in people living with HIV [6,22,23]. High mortality rates associated with HIV/HBV infection could be due to hepatic diseases caused by HBV, whose progression is intensified by HIV. Parvez et al. [22] showed that hepathopathic mortality is 17 times higher in coinfected patients than mono-infected patients. Liver diseases induced by HIV include hepatitis, hepatic steatosis, endothelialitis, necrosis, granulomatous disease, cirrhosis and carcinoma.
However, in our study, we did not find an association between HBsAg positivity and risk of death, as documented in a number of European studies. HBsAg alone cannot always indicate the presence or absence of HBV among people living with HIV, hence their exposition to occult viral hepatitis B [24,25]. The latter is defined by HBsAg-negative, positive anti-HBc antibodies, negative anti-HBs antibodies and DNA of detectable HBV. Thus, because of the lack of HBV viral DNA, patients suffering from occult hepatitis B were not detected. This could have led to classification errors between HBV and risk of death. Moreover, our sample size was not large enough to get a good statistic test power for demonstrating the associations.
In countries where HBV is highly endemic, such as Burkina Faso, and where HBsAg tests and viral DNA dosage are not available, first-line HIV treatment should include two molecules that are active on HBV, such as TDF. This will decrease the morbidity and the mortality risk of the HIV/ HBV co-infection as well as the risk of HBV resistance caused by the monotherapy with lamivudine, which is particularly observed in HBsAgpositive HIV infected patients [26]. Thus, in areas where hepatitis B is highly endemic, absence of resources to test for hepatitis B, could justify the systematic prescription of ART containing TDF in people infected with HIV.
Hepatic fibrosis is a serious complication of infectious and toxic hepatitis. It is correlated with a high risk of death due to hepatic insufficiency [7,22]. The relationship appears in the univariate analysis in our study, but disappears in the multivariate analysis due to the low number of patients with stage 3 fibrosis.
The study has some limitations due to the context. In fact, the hepatitis B serological markers and HBV viral DNA was not determined. Additionally, the dynamic of hepatitis B viral markers under ARV treatment was not studied, and finally, no liver biopsies were realized. Only FIB-4 score was used as a non-invasive marker to test for hepatic fibrosis.
The study carried out at the Day Hospital of Bobo-Dioulasso shows that HBsAg seroprevalence is higher amongst people living with HIV than the general population. The FIB-4 score, a bio-marker for hepatic fibrosis was higher in HBsAg-positive patients compare with HBsAg-negative patients. The occurrence of death in those under ART was comparable to patients mono-infected with HIV. Testing for hepatitis B should be systematic in patients infected with HIV. In the absence of resources to test for HBV, TDF could be systematically included in first-line ART in areas were hepatitis B is highly endemic.
Prospective cohort studies with thorough biological examinations are needed to determine the precise consequences of HBsAg positivity on liver function and dynamics of viral markers of hepatitis B in people living with HIV on antiretroviral treatment.
AH collected and analyzed the data, AS drafted the manuscript; PI, DZ offered the technical or material support; JZ, AP, AS revised the manuscript.
Download Provisional PDF Here
Article Type: Research Article
Citation: Sawadogo A, Hema A, Koura M, ILBOUDO BP, Kamboule BE, et al. (2016) Epidemiological, Biological and Progressive Aspects of Hepatitis B Virus B (HBV) Infection among Patients Infected by Human Immunodeficiency Virus (HIV) in the Day Hospital of Bobo-Dioulasso, Burkina Faso. J HIV AIDS 2(3): doi: http://dx.doi.org/10.16966/2380-5536.124
Copyright: © 2016 Sawadogo A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access