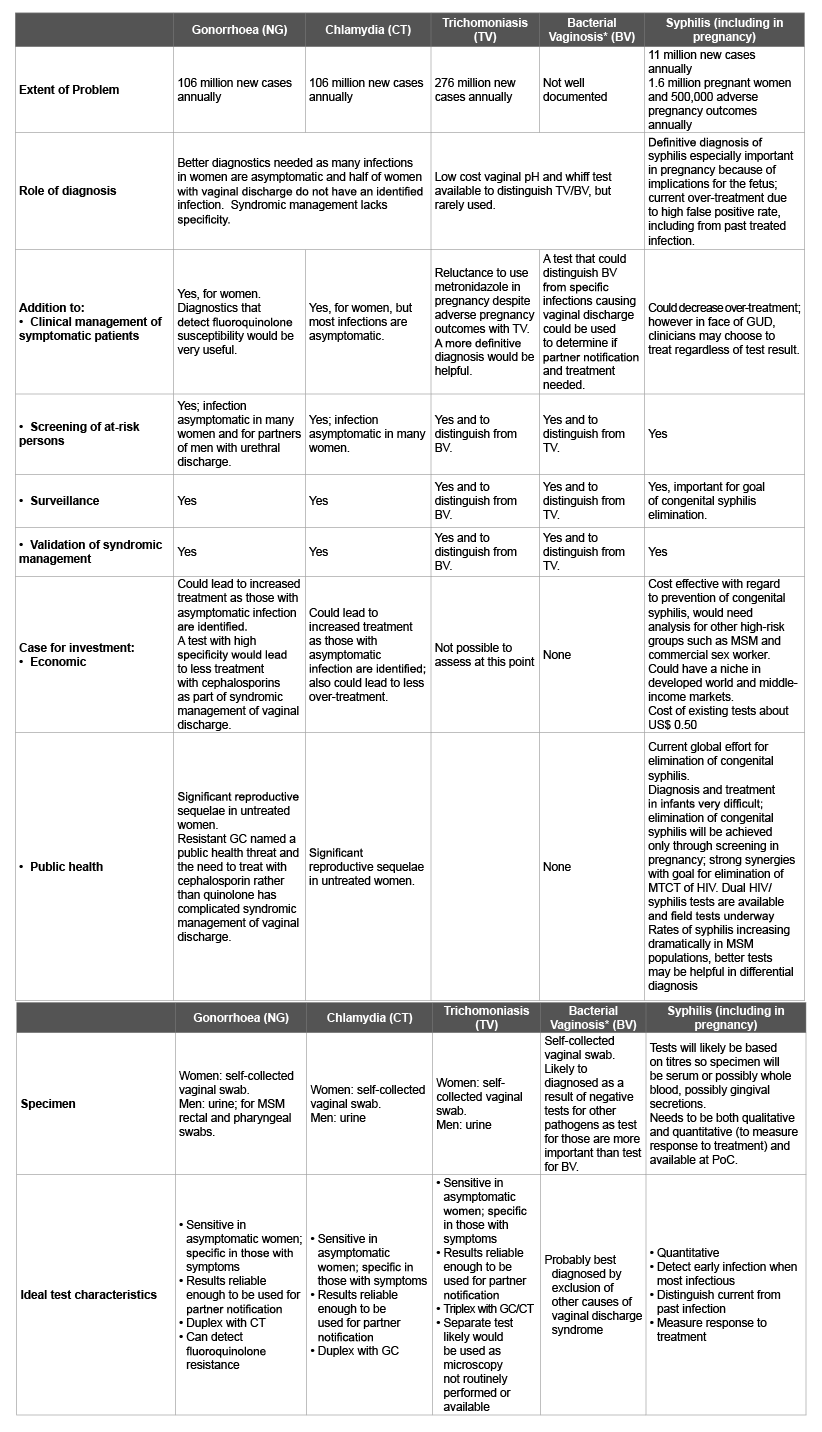
Table 1: Potential role and impact of new PoC tests by infection – High priority areas

Renee Ridzon*1 Timothy Farley2 David L Dolinger3
1AHIMSA Associates, Boston, Massachusetts, USA*Corresponding author: Renee Ridzon, AHIMSA Associates, Boston, 41 Worcester St, #3 Boston, MA 02118, USA, Tel: +12063106573; E-mail: reneeridzon@gmail.com
Introduction: Sexually transmitted infections (STIs) cause morbidity and increase rates of acquisition and transmission of HIV infection. WHO estimated that almost 500 million new cases of the four most common curable bacterial STIs (Chlamydia trachomatis, Neisseria gonorrhoeae, Treponema pallidum and Trichomonas vaginalis) occurred worldwide in 2008 among adults aged 15-49 years. Strategies for HIV and STI control include promotion of safe sexual behaviour, provision of condoms and early and efficacious management of STIs. STI diagnosis treatment in resource-limited settings relies on syndromic management leading to both under-treatment of asymptomatic infections and over-treatment of symptoms that mimic STIs. Point-of-care tests are potentially powerful contributors to STI management by allowing correct diagnosis and same day, directed treatment.
Methods: To identify unmet needs and opportunities to catalyze innovation, a review of the status and rationale for new PoC STI diagnostics was conducted through stakeholder interviews.
Results: Priorities identified for STI POC tests included ability to enable directed treatment at the time of presentation, decrease over-treatment and increase treatment of asymptomatic infections. Infections given the highest priority for development included chlamydia and gonococccal (GC) in women, detection of antimicrobial resistance in GC infections, tests for diagnosis of vaginal discharge and ability to distinguish new syphilis infection from prior, treated infection.
Conclusion: There is potential for well-performing new tests to have an impact on STI epidemiology, however to attract interest, tests must be cost effective with existing STI management strategies.
Sexually transmitted infections; HIV
Sexually transmitted infections (STIs) cause morbidity and increased risk for acquisition and transmission of HIV infection [1]. WHO estimated that almost 500 million new cases of the four most common curable bacterial sexually transmitted infections (STIs) (Chlamydia trachomatis, Neisseria gonorrhoeae, Treponema pallidum and Trichomonas vaginalis) occurred worldwide in 2008 among adults aged 15-49 years [2]. The complications of STIs include congenital syphilis, ectopic pregnancy, chronic pelvic pain, infertility and increased risk of transmission and acquisition of Human Immunodeficiency Virus (HIV) infection [3]. It has been argued that the failure of community interventions to treat STIs to reduce HIV incidence was due to inability to accurately diagnose and treat STIs [1,4].
Strategies for STI control include promotion of safe sex, provision of condoms and early and efficacious management of STIs. The backbone of accessible and affordable STI treatment in resource-limited settings is syndromic management which relies on algorithms for each STIassociated syndrome [5]. The algorithms enable presumptive diagnoses of common STIs, appropriate treatment, advice on the management of sexual partners and same-visit HIV testing. When specimens can be taken but there is delay before results are available, a similar presumptive treatment approach is followed. The diagnosis, once available is used to modify therapy as needed. Because of limited sensitivity and specificity of syndromic management, there is over-treatment of symptoms that mimic STIs and under-treatment of asymptomatic infections [6].
Reliable, simple point-of-care (PoC) tests that provide a result while the patient still is in clinic are potentially powerful contributors to STI management and HIV control by allowing screening of patients for asymptomatic infections, correct diagnosis of symptomatic infection and same day, directed treatment.
One model has estimated that in comparison to syndromic management, a 70–80% sensitive and 95% specific PoC diagnostic test for Neisseria gonorrhoeae and Chlamydia trachomatis would lead to 24–31% fewer treatments through eliminating over diagnosis and 40–60% more N. gonorrhoea, C. trachomatis and HIV infections would be averted [7]. In another model based on data from women from Zimbabwe and Uganda, incident N. gonorrhoea infection (hazard ratio (HR) 5.46, 95% CI 3.41–8.75) and bacterial vaginosis (BV) (HR 2.12, 95% CI 1.50–3.01) were independently associated with HIV acquisition [8]. Prompt initiation of appropriate treatment has considerable potential to improve STI control by reducing STI transmission and sequelae of untreated or inadequately treated infections, facilitating partner notification and treatment, reducing costs by decreasing over-treatment and impact HIV transmission and acquisition by reducing the enhancing effect of STIs on HIV [1]. We undertook a review of the rationale for new PoC STI diagnostics with the objective of identifying unmet needs and opportunities to catalyze innovation for a cost-effective contribution to sexual health in resourcelimited settings.
A total of 18 experts in international public health and STI control were interviewed for this qualitative study. All have had direct involvement with STI and reproductive health care, and public health approaches to STI control through their commitment to research and experience in international health, international agencies or in STI control programs. All interviews followed an open ended, semi-structured approach on different STIs with concentration on each interviewee’s area of expertise with regard to current practices for management of specific STIs and perceived needs for PoC diagnostic tests. Experts were asked for views on prioritization of test development by syndromes or infecting organisms. Literature obtained through data searches and the interviews was also reviewed. Information reported represents an assimilation of expert opinions and information available in the literature and quantitative analysis was not performed. Review of the current status of testing is reviewed briefly to present the context of current testing practices. As the study did not involve human subjects, clinical research or confidential information, no ethical approval was sought.
While the focus here is needs and opportunities for new PoC tests, costs of developing and deploying tests and cost effectiveness in various settings needs to be carefully assessed. If the new tests are not likely to be cost-effective, there is little point to development. If the test can be cost-effective in the long term, it is necessary to determine the recovery time for the development costs. Potential philanthropic or development agency support could offset development costs, but financial mechanisms to support long-term sustained use also need to be identified.
The ASSURED criteria (Affordable, Sensitive, Specific, User-friendly, Rapid and robust, Equipment-free and Deliverable to end-users) for PoC tests developed by the WHO Sexually Transmitted Diseases Diagnostics Initiative remain relevant in test development [9]. New tests that permit diagnosis and treatment in a single visit can be expected to increase the proportion of cases that initiate same-day treatment identifies asymptomatic infections facilitate contact tracing, and ultimately result in lower STI incidence and improved health outcomes even if the test has lower sensitivity than gold standard tests that cannot produce same-day results [10]. In one modelled scenario, if 50% of women with C. trachomatis infection return for their test results, and if half the women infect another person during the delay in treatment, a sensitivity of a PoC diagnostic test of only 29% is needed to match infections averted of a 90% sensitive gold-standard test [10].
A survey of practicing STI clinicians identified time required for test completion, test complexity and interruption of work flow as top barriers to use of PoC tests [11]. Any new PoC test would need to consider these three elements to be acceptable and feasible for use in intended settings.
Vaginal discharge in reproductive age women is frequently physiological. With abnormal discharge accompanied by vaginal erythema, vulval itching or burning, syndromic management recommends treating BV and T. vaginalis with an antimicrobial such as metronidazole. Diagnostic methods for BV include determination of vaginal fluid pH (>4.5) and/or a fishy amine odour when vaginal fluid is mixed with a 10% potassium hydroxide solution (whiff test). Microscopic examination of vaginal fluid can be performed to look for motile trichomonads (T. vaginalis), clue cells (BV) or budding yeasts and/or pseudohyphae (Candida albicans).
T. vaginalis is the most common non-viral STI worldwide with 276 million estimated cases in 2008 [2]. Whereas this infection can occur in men and women, its course is comparatively short in men and it is only suspected when urethral discharge caused by N. gonorrhoeae or C. trachomatis has been ruled out. Infection in women increases the risk of adverse pregnancy outcomes, acquiring HIV infection and HIV shedding in women with HIV infection. Recognition and treatment of T. vaginalis is a major neglected element of STI control [12].
Current and potential new diagnostic tests: Assessing vaginal pH with an indicator strip and the whiff test are very low-cost PoC tests that require minimal resources and can be based on self- or cliniciancollected vaginal swabs. Although clinical performance of these methods is not optimal, no new test is likely to compete with regard to cost. It appears that these simple methods are not routinely used in STI clinics in resource-limited settings due to a lack of supplies, the subjective nature of the whiff test and high workload. Microscopy can distinguish among BV, T. vaginalis and C. albicans but requires expertise and equipment not likely available at the first level of care. Several antigen tests for T. vaginalis infection are reported to have higher sensitivity than microscopy (51-65% vs 82%, respectively) [13,14]. NAATs have very high sensitivity for T. vaginalis infection but are not available as PoC tests.
Though the treatment of BV and T. vaginalis is identical, treatment of the sexual partner is needed in the context of T. vaginalis infection. A simple, affordable PoC test specific for T. vaginalis would allow treatment of infected sexual partner(s) to avoid reinfection of the index case. Whereas a recommendation to develop such a test was made and supported, it was not given high priority for test development because of the current availability of inexpensive, however imperfect, PoC tests. For details on the specific tests, see Table 1.
Urethral and cervical infections are the most common conditions for which patients with STI seek care. Primarily due to infection with N. gonorrhoeae (gonorrhoea) or C. trachomatis (chlamydial infection) each is estimated to have occurred in about 106 million adults in 2008 [2]. In men, infection leads to urethral discharge and/or painful urination and most seek care promptly. If microscopy is available, examination of the urethral smear can be used to aid diagnosis. As the syndromic approach to treatment of urethral discharge presumptively covers both organisms, a specific diagnosis is not required before initiating treatment. Infection of the rectum (proctitis) and pharynx with N. gonorrhoeae are commonly asymptomatic. Though these sites of infection can occur in both sexes, they are predominantly found in men who have sex with men (MSM).
In women, the situation is considerably more complex where infections with N. gonorrhoeae and C. Trachomatis may be asymptomatic in up to 85% [2]. If undetected and untreated, infection can cause pelvic pain, ectopic pregnancy and infertility. Unlike the relatively effective syndromic approach to urethritis in men, there are severe limitations to the syndromic approach to vaginal and cervical discharge because of the high proportion of asymptomatic infections. A report from South Africa of women at high risk of HIV and other STIs showed only 12% (25 of 204) with a laboratorydiagnosed, discharge-causing STI had clinically evident discharge [15].
Current and potential new diagnostic tests: While pelvic examination with a speculum is necessary to observe purulent discharge from the cervical ostium and collect specimens, this is rarely performed in resourcelimited settings and many first-line health care workers do not have the skills to perform such an exam. Self-collected vaginal swabs and urine specimens have shown good performance for diagnosis by nucleic acid amplification test (NAAT) technologies, and could therefore potentially decrease the need for pelvic examination [16,17].

Table 1: Potential role and impact of new PoC tests by infection – High priority areas
The high sensitivity and specificity of NAATs have set a high performance bar for tests for N. gonorrhoeae, (>90% sensitivity, ≥ 99% specificity), C. trachomatis (>90% sensitivity, ≥ 99% specificity), and T. vaginalis (95.3%–100% sensitivity, 95.2%–100% specificity) [13,18]. The US Food and Drug Administration has indicated that new tests considered for approval would need performance comparable to currently available NAATs, despite the potential for less expensive PoC tests to substantially improve syndromic management albeit with less sensitivity and specificity.
POC test characteristics: Microwave-accelerated metal-enhanced fluorescence (MAMEF) ultra-rapid tests have high rates of agreement with NAATs and the platform appears to be suitable for detection of N. gonorrhoeae, T. vaginalis and Haemophilus ducreyi and may warrant investigation for further development as a PoC test [19].
High priority was placed on PoC tests that can produce results in less than 20 minutes and detect symptomatic and asymptomatic N. gonorrhoeae and C. trachomatis infection, with the greatest need for detection of C. trachomatis in women, an opinion that concurs with a survey of USA-based professionals in the STI field [11,20]. This test could be combined with a test for BV and T. vaginalis, or a second dual test could be run in parallel. A triple test for N. gonorrhoeae, C. trachomatis and T. vaginalis could allow directed treatment if the result was positive for any of the three organisms; if negative to all three, the woman would be treated for BV without partner notification. Given the ease and acceptability of self-collected vaginal swabs, any new test should perform well on vaginal swabs and urine specimens (for use in men). With improved ability to detect asymptomatic infections in women, such a PoC test could have a major impact on STI management strategies, (particularly for women) and should lower STI incidence and prevalence as well as HIV incidence [7].
Antimicrobial resistance: The emergence in N. gonorrhoeae resistant to the “last-line” cephalosporins, together with high prevalence of resistance to penicillins, sulfonamides, tetracyclines and more recently, quinolones and macrolides is a major concern. Gonorrhoea has the potential to become untreatable, particularly in settings with a high burden of gonococcal infections [21]. Rates of fluoroquinolone resistance of ≥ 5% in infections in the general population or ≥ 1% in key populations has resulted in recommendations to use injected ceftriaxone as a first line treatment of gonorrhoea [22,23]. Even with rates of fluoroquinolone resistance of 5% that triggers a change in empiric first-line treatment, 95% of persons could be successfully treated with a fluoroquinolone if drug susceptibility of an individual infection could be determined. Diagnostic tests capable of detecting antimicrobial resistance (AMR), with a priority on detecting fluoroquinolone resistance could contribute to an urgent global need for more antibiotic stewardship, decrease antibiotic pressure on circulating organisms and allow easier, cheaper treatment to be used in most individuals.
Current NAAT technologies are capable of detecting AMR, but none have been developed for AMR in N. gonorrhoeae. Development of molecular tests for detecting AMR would facilitate both clinical management and monitoring of resistance trends [23,24].
Genital ulcers constitute an important syndrome for which patients seek care and may be due to infection with one of several organisms; most importantly Treponema pallidum (syphilis) and Herpes simplex virus type 2 (HSV-2) infections.
There are an estimated 10-11 million new syphilis infections worldwide annually [2]. Rising incidence, particularly among MSM, has been reported [25]. The greatest public health concern is syphilis in pregnancy, resulting in mobilization of resources for global elimination of congenital syphilis [26]. Globally in 2008, a total of 1.4 million pregnant women were estimated to have active syphilis, resulting in an estimated 520,000 adverse pregnancy outcomes (212,000 stillbirths or early foetal deaths, 92,000 neonatal deaths, 65,000 preterm or low birth weight infants, and 152,000 infected newborns) [27].
Current and potential new diagnostic tests: Diagnosing and treating neonatal syphilis is very difficult and prevention of congenital syphilis is achieved only through effective screening and treatment of pregnant women. There are severe limitations to current diagnostic methods. T. pallidum can only be recovered from active lesions that may go unnoticed and requires specialized equipment (dark field or fluorescent microscopy) and training. Whereas several highly sensitive PCR assays to detect T. pallidum-specific DNA have been developed, they are not practical for use as a PoC test if a second visit is required before initiating treatment, resulting in missed opportunities for intervention [6].
Syphilis screening relies on serological tests of blood or serum. In many instances, pregnancy included, this is performed using the specimen taken for HIV screening, either tested in a single multiplex test or in two separate tests. Non-treponemal serological tests are sensitive, but false positive results can occur in association with medical conditions other than syphilis, and all reactive non-treponemal tests should be confirmed with a more specific treponemal test. Treponemal tests cannot be used for monitoring response to therapy as they remain reactive for life, even after successful treatment. As a result of the difficulty in diagnosing recent syphilis infection, patients with GUD are presumptively treated for syphilis [6].
For speed, cost-effectiveness and efficiency, pregnant women may be screened with only a treponemal test and, if reactive, treated. Because treponemal tests remain reactive after treatment, rates of over treatment will increase with every subsequent pregnancy. Given the severity of the outcomes being prevented and the relative safety of treatment, some degree of over-treatment is tolerable, but the problem of over-treatment will worsen as progress is made towards the elimination of congenital syphilis.
A dual PoC treponemal and non-treponemal syphilis test is a potential solution, but the test currently available (DPP® Syphilis Screen & Confirm Assay, ChemBio) is less sensitive than other tests. In one report, sensitivity reported of the non-treponemal test among those with an RPR titer of 1:4 is 22.2% (vs 90.0% among those with a titer of 1:8 or greater) and sensitivity of treponemal test when compared to TPPA was 52.7% [28].
There is considerable interest and investment in the elimination of congenital syphilis and HIV. Testing in pregnancy is well supported and a large number of tests are being used annually, with many different tests on the market. New or improved dual treponemal–non-treponemal serological tests, possibly combined with HIV antibody testing, would have great potential in programmes committed to global elimination of congenital syphilis. It is important to consider price and willingness to pay for the added advantage of the new test. For example, the currently available HIV PoC tests purchased by PEPFAR cost about US$ 0.50 per test, whereas the dual test is priced at about US$ 2.50 [29]. More expensive new tests will have to demonstrate substantial utility and advantages in terms of number of cases detected and successfully treated if purchasers are to buy them. ]\
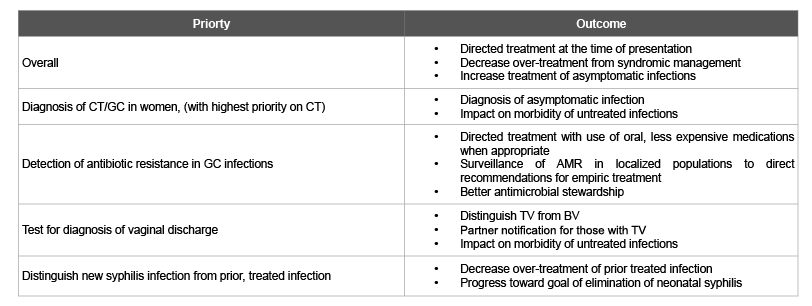
Table 2: Priorities for STI PoC Test Development
Other causes of genital ulcer disease: The most common cause of GUD other than syphilis is genital herpes, a viral infection. Unlike the bacterial STIs, herpes is incurable and requires episodic treatment or long-term suppressive therapy, neither of which is feasible in most lowincome settings. Other aetiological factors in GUD include C. trachomatis, Klebsiella granulomatis or Hemophilis ducreyi but are not prioritized for PoC diagnostic test development because of lower incidence compared with other STIs.
The global burden of STIs is enormous and current approaches to STI control are inadequate and improved approaches to control of bacterial STIs would impact these infections as well as HIV control. Rapid PoC tests with high sensitivity and specificity and a rapid turn-around time have the potential to change STI control and resources spent on STI treatment by ensuring that prompt treatment is initiated, especially for women who bear higher morbidity from these infections. This review is limited by a relatively small number of experts interviewed and the qualitative nature of the data. Despite this it did look across the field of STI control to identify areas of greatest need for test development in hopes of giving information about where to prioritize efforts. It has revealed recognition of PoC tests as an important element for global STI and HIV control with considerable enthusiasm for developing new or advancing existing promising STI PoC test technologies, particularly with priority on tests for gonorrhoea, chlamydia and trichomoniasis (Table 2). The experts emphasized that in some cases existing technologies may be utilized and improved to develop appropriate tests whereas in others, new technologies are needed.
Development of PoC STI tests however, presents challenges including likely need for several tests, even for highest priority STIs and fewer dedicated resources for test development compared to other infections such as HIV and tuberculosis. Additionally, before embarking on a development plan, a business case must be made and any new test must undergo validation and field-testing in different geographies and age strata to understand test performance and utility. There is potential for well-performing new tests to have an impact on STI epidemiology in middle and low-income as well as developed countries, but it is not known whether this impact will be cost-effective and attract donor/industry interest. Claims that new PoC tests may be cost saving when compared with existing STI management strategies, particularly in middle-income countries, must be carefully evaluated before any new initiative to develop new STI PoC tests is launched.
The authors thank the stakeholders who contributed their time and expertise to the preparation of this landscape analysis.
Download Provisional PDF Here
Article Type: Research Article
Citation: Ridzon R, Farley T, Dolinger DL (2015) Point of Care Tests for Diagnosis of Sexually Transmitted Infections: A Landscape Analysis. J HIV AIDS 2(1): doi http://dx.doi. org/10.16966/2380-5536.115
Copyright: © 2015 Ridzon R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access