Abstract
The trafficking of membrane cholesterol (Chol) in mammals represents key processes in cells function, particularly in sperm physiology since Chol removal is strictly associated with motility gain. Moreover, the involvement of oxidized Chol derivatives, known as oxysterols, has been recently summoned in the regulation of sperm function. The study of sterols dynamic in human sperm is largely hampered by the low sample availability and the inadequate sensitivity of current methods based on chromatographic techniques. In this study we aimed to develop a robust and reliable method based on liquid chromatography-mass spectrometry (LC-MS) to quantify the sperm levels of Chol and major oxysterols 7β-OH-Cholesterol (7β -OHC) and 7-keto-Cholesterol (7-KC). In particular, by finely tuning the reverse-phase chromatography parameters, the unambiguous identification of Chol, 7β -OHC and 7-KC in biological samples was allowed on the bases of their different retention times, accurate m/z determination and natural isotope abundance pattern. Finally, by applying the method on real sperm samples from 12 semen donors, we documented that normozoospermic subjects (total sperm count >39 × 106 cells/ejaculate) showed an underrepresentation of all steroids compared to subjects with oligozoospermia (total sperm count ≤ 39 × 106 cells/ejaculate; respectively P=0.048 for Chol, P=0.006 for 7β-OHC and P=0.001 for 7-KC). These preliminary data suggest further investigation about the impact of disorders of the spermatogenic process on the composition and function of the sperm membrane.
Keywords
sperm membrane, spermatogenesis, cholesterol, oxysterols, liquid chromatography, mass spectrometry
Introduction
The synthesis and trafficking of cholesterol (Chol) in mammals represent among the most tightly regulated biological processes [1]. Deregulation of this balance results in major consequences on cell homeostasis and health status of an individual. As an example, uncontrolled accumulation of Chol in inflammatory cells and smooth muscle cells within intima and media layers of the vessel represents a known feature of atherosclerosis that progressively leads to the thickening of vascular wall and occlusion of the vessel lumen [2]. Furthermore, the control of Chol homeostasis seems to rely to additional mechanisms, beyond the mere variations of intracellular levels of Chol. In fact, recent studies support the active involvement of other mono-oxygenated derivatives of Chol, globally known as oxysterols, which can be derived from enzymatic or non-enzymatic pathways [reviewed in 3]. There are more than 30 different known oxidative derivatives of Chol, but only a few of them are quantitatively important, including 7β -OH cholesterol (7β-OHC) and 7-Ketocholesterol (7-KC) which are produced through non-enzymatic pathways [3]. Interestingly, the production of oxysterol has been shown to impact on Chol homeostasis, being directly related with the cell Chol content. Once produced, oxysterols would subsequently regulate Chol synthesis by either the involvement of the Liver-X-Receptor (LXR) or through LXR-independent mechanisms Figure 1A [3, 4].
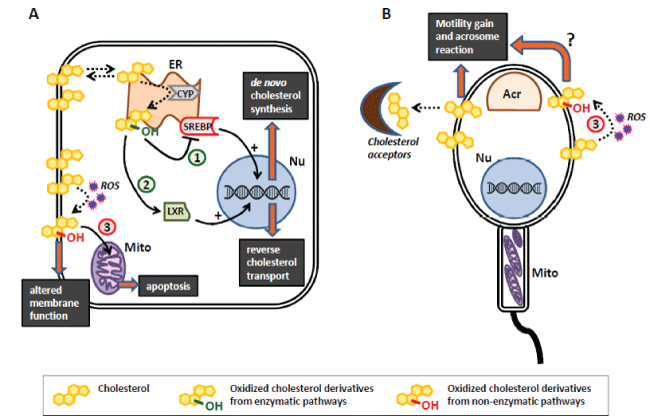
Figure 1: Recognized pathways involving cholesterol and its oxidized derivatives in somatic cells (A) and sperm cell (B).
A. Oxysterols produced by controlled enzymatic pathway (CYP) in the endoplasmic reticulum (ER) regulate cholesterol homeostasis by two different pathways: 1) through inhibition of sterol-regulatory-element-binding-protein (SREBP), which in turn translocates in the nucleus (nu) up-regulating genes involved in cholesterol biosynthesis, and 2) through the activation of liver-X receptor (LXR) that promotes reverse cholesterol transport. Oxysterols produced by non-enzymatic pathway (3), by direct reaction with reactive oxygen species (ROS), are associated with alteration of membrane fluidity and function together with mitochondria (Mito)-mediated apoptosis.
B. The removal of membrane cholesterol from sperm cells, exerted by extracellular acceptors, is a key event during the motility gain and the ability to fuse (acrosome reaction) with the oocyte. The involvement of non-enzymatic production of oxysterols (3) in this process is currently under investigation.
The dynamic trafficking of membrane Chol has also been proposed to be a key event in the regulation of sperm physiology. Indeed, early in vitro studies in mammals revealed that the gain of cell motility is obtained by the incubation of sperm with calcium ions, bicarbonate and, most importantly, Chol acceptors such as albumin, HDL and even synthetic chelators like cyclodextrins [5,6] Figure 1B. On these bases, the overall amount of membrane Chol may represent a limiting factor in the achievement of the proper fertility potential [7]. Thus, an accurate evaluation of the membrane sterol pattern may represent a useful tool to address the functional status of sperm cells.
Although the knowledge of Chol and oxysterols levels in sperm cells would greatly help to address their physiological role in the functional status of male gametes, and in particular in infertile patients, the analytical quantification of the sperm membrane content suffers of major sensitivity problems. Not as a case, most of the studies on this topic were performed on livestock featured by relatively wide availability of semen samples [7-9]. The aim of this study was to identity a robust LC-MS method for the assessment of the sterol pattern of sperm membrane. To this regard, the study focused on the quantification of Chol and 7β -OHC and 7-KC as the two main oxysterols indicated to be relevant in sperm function.
Methods
Chemicals and Instrumentation
Cholesterol, 13C isotope labelled cholesterol, 7β -OH cholesterol, 7-Ketocholesterol and Supelclean TM LC-Si SPE Tube 1mL were all purchuased from Sigma Aldrich (St.Louis, MO, USA). All other reagents, solvents and salts were obtained from Fluka AG or Merck (Darmstadt, Germany).
Semen sample preparation and processing
The study has been approved by the Ethics Committee of the UniversityHospital of Padova (protocol n. 2208P) and each participant has signed informed consent. The study has been conducted in accordance with the principles expressed in the Declaration of Helsinki. 12 male volunteers, self-reporting a good health status, were included in the study. Inclusion criteria were age between 18-25 years whilst exclusion criteria were varicocele, metabolic syndrome, malignancies, history of cryptorchidism, sexual hormone alteration. All subjects underwent semen donation by masturbation into sterile containers after 2-5 days of sexual abstinence. Samples were allowed to liquefy for 30 min and were examined according to the WHO criteria [10]. Briefly, the evaluation of total cell count was performed by Makler counting chamber (Santa Anna, CA, USA), which permits to count the concentration of sperm cells for ml and also the total sperm count (concentration per total volume of ejaculate). Furthermore, sperm motility was assessed after liquefaction of the sample, at 30 minutes, following ejaculation, to limit the deleterious effects of dehydration, pH or temperature changes that could affect motility. The overall sperm motility of each sample was graded according to WHO criteria, evaluating the progressive motility (PR), namely the percentage of spermatozoa moving actively, either linearly or in a large circle, regardless of speed; the nonprogressive motility (NP) as the percentage of sperm cells featured by all other patterns of motility with an absence of progression, e.g. swimming in small circles; immotility (IM) as the percentage of sperm cells showing no movement. For this discrimination, the above mentioned Makler counting chamber was used as well. Sperm viability was evaluated by assessing the membrane integrity of sperm cells, for samples with less than 40% of progressive motility. The percentage of viable sperm was assessed by identifying those cells with an intact cell membrane by using eosin Y (St. Louis, Missouri, USA) staining. We considered as viable the unstained cells, based on the principle that the entry of eosin Y through plasma membranes is allowed only in damaged membranes of dead cells. Clinical and semen characteristics of male healthy donors are reported in Table 1. According to the definition of normozoospermia (total sperm count >39 × 106 cells/ejaculate), 4 out of 12 (33%) normozoospermic subjects were identified in the study group. After semen evaluation, samples were washed twice in phosphate buffered saline (PBS) and the cell pellet stored at -80°C until use.
| Parameter |
Mean value (range) |
| Age (years ± SD) |
22.4 ± 3.1 |
| Sexual abstinence (days ± SD) |
3.6 ± 1.2 |
| Total sperm count (106 ± SD) |
35.2 ± 57.1 (0.01-195) |
| Progressive motility (% ± SD) |
27.8 ± 19.1 (2-60) |
| Sperm viability (% ± SD) |
76.8 ± 0.1 (56-90) |
| Normal sperm morphology (% ± SD) |
6.6 ± 5.6 (0-16) |
Table 1: Seminal characteristics of healthy semen donors (N=12)
Sperm lipids isolation and solid phase extraction (SPE)
The total lipid fraction was extracted from the pellet of sperm cells with chloroform/methanol mixture [11]. Sperm cells pellet, deriving from individual semen sample, was incubated at 37°C with 1 ml of chloroform/ methanol mixture under gentle mixing. Dilution with chloroform and water separates the extract into two layers, the chloroform layer containing all the lipids and the methanolic layer containing all the non lipids species. Thereafter, the extract was centrifuged at 10.000 g for 10 min and the chloroform layer was lyophilized in a Vacufuge β-Concentrator plus (Hamburg, Germany). After lyophilization, the extract was resuspended in 200 µl of chloroform. The sterol fraction was then isolated by solidphase extraction (SPE) on a 1-ml silica-gel column. The column was preconditioned with a methanol-chloroform (1:1 v/v) solution, while the phospholipid component was separated by elution with neat chloroform and the sterol fraction was eluted with acetone according to previous reports [12]. The lyophilized sterol fraction was finally dissolved in methanol and 20 µl of this solution underwent UPLC analysis.
Preparation of sterols-standards solutions
Stock solution containing cholesterol or oxysterols (each 1 mg/ ml in methanol) were prepared by weight, using a Mettler-Toledo (Columbus, OH, USA) XPE analytical balance, and calibration standards were obtained by diluting the stock solutions with methanol. Proper sterols mixtures were prepared and processed according to the sample preparation protocols used for real samples in order to accurately quantify of oxidized derivatives of cholesterol. In particular, the influence of excess cholesterol on the quantification of oxysterols was addressed by adding cholesterol (100 µg/ml) to an oxysterols standard mix (10 µg/ml) in methanol. Linearity of the MS signal for cholesterol and oxysterols standards was tested in a range from 10 ng to 2000 ng. The recovery of cholesterol from the sperm matrix was assessed. Before SPE, all sperm samples from healthy donors as well as calibration standards were spiked with 13C isotopically labelled cholesterol as the internal standard to account for variations during sample pretreatment.
LC-MS analysis
LC-MS analysis was performed with an Agilent (Santa Clara, CA, USA) 1290 Infinity Ultra-High Performance Liquid Chromatography (UPLC) system, equipped with a reverse phase (RP) Poroshell 120 EC-C18 column (4.6 mm × 150 mm, 2.7 µm particle size) from Phenomenex (Torrance, CA, USA), and a 920 autosampler, was downstream connected to a Waters (Milford, MA, USA) Xevo-G2S Q-TOF mass spectrometer.
Elution was performed under isocratic conditions by the use of an acetonitrile-methanol mixture, whose composition was progressively varied in order to achieve the best chromatographic separation of analytes.
Ionization and vaporization of the effluent from the column was obtained by atmospheric pressure ionization (APCI) in the positive ion mode with the capillary potential set at 1.50 kV and source temperature at 110°C
For selected applications, the mass spectrometer was operated by selecting the m/z value for each analyte and recording the total ion current (TIC) at the detector within a mass window of ± 1 a.m.u. The error on (oxy) sterol mass determination was always <2 ppm. Data were acquired by the Mass-Lynks software using a data pitch of 0.5. Integration of mass peaks was performed with the BioPharmaLynks suite (Waters). The limit of detection was calculated from methanol standard solutions after sample preparation by SPE, as described above, using a signal-to-noise (s/n) ratio of 3. The lower and upper limits of quantification were stated as the concentration levels meeting the criteria of a coefficient of variation (CV) lower than 20%, where CV was defined as the standard deviation of a set of measurements divided by the mean value of the set.
Statistical analysis
The results were expressed as means ± standard deviations (SD). Prior to data analysis, the Kolmogorov-Smirnov test was used to check for normality of distribution. Parameters not showing normal distribution were log transformed for statistical analyses. The Levene’s test was used to check the homogeneity of variance among groups. If homogeneity of variance assumption was violated, Welch test was performed and the respective p value was reported. Linear correlation between two variables was assessed with the Pearson’s correlation test. p values <0.05 were considered as statistically significant. Statistical analyses were performed using SPSS version 21.0 for Windows (Chicago, IL, USA).
Results
Method development
The mass spectra of the three analytes were firstly identified by direct infusion in the mass-spectrometer. Representative mass spectra of Chol, 7β-OHC and 7-KC are reported in Figure 2A. Consistent with data reported in the literature [13,14], Chol and 7β-OHC underwent water loss and proton addition during APCI-MS analysis and were detected as [M+H-H2 O]+ions at 369.35 m/z and 385.33 m/z, respectively, whereas 7-KC was detected as a molecular ion [M]+ at 400.33 (m/z) and [M+H]+ ions at 401.33 m/z. However, the fragmentation pattern of sterols was very complex and not specific and this evidence forced the discrimination of analytes on the bases of their different retention times during RPchromatography, accurate m/z determination and natural isotope abundance pattern, allowing unambiguous identification in the biological samples. The analysis of oxysterols resulted equally challenging due to their very low physiological concentrations, structural similarity, and the possibly interfering excess of cholesterol [15]. In Figure 2B are reported the representative LC-MS traces of commercial standards of a mixture of Chol, 7β-OHC and 7-KC 1:1:1 (250-350 pmoles), eluted under isocratic conditions in a mobile phase with a composition of acetonitrile:methanol, 90:10 v/v and recorded as total ion current. Indeed, 7β-OHC and 7-KC were eluted at nearly the same retention time, preventing any possible unambiguous identification. However, slight modification of the mobile phase (acetonitrile:methanol, 65:35 v/v) allowed a sufficient resolution of the two analytes. As reported in Figure 3A, LC-MS traces of the standards mixture recorded at the individual molecular ion, showed a sufficient chromatographic resolution of 7β-OHC and 7-KC to allow peak identification and quantification (respective retention times 5.80 minutes and 6.09 minutes).
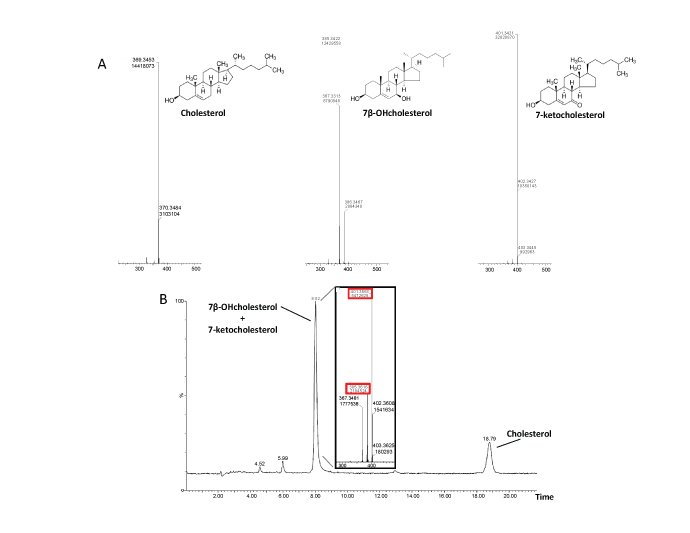
Figure 2: A. Mass spectra of the commercial standards of obtained by direct infusion of analytes into the MS detector. Mass values were determined within 2 ppm accuracy.
B. LC-MS analysis of a standards mixture of Cholesterol, 7β-OHcholesterol and 7-ketocholesterol 1:1:1 (250-350 pmoles) eluted from a reversed phase column under isocratic conditions (acetonitrile : methanol, 90:10 v/v) and recorded as total ion current. The inset reports the MS analysis of the species constituent the peak eluting a 8.02 minutes.
Quantification of cholesterol and oxysterols
The method was validated according to current FDA and EMA bioanalytical guidelines [16,17]. The intra-assay repeatability was determined on methanol standard solutions at low (2 µM) and high (100 µM) cholesterol concentration by analyzing each solution 3 times per run. For quantification purposes, calibration curves were constructed for cholesterol, 7β-OHC and 7-KC, and found to be linear over a broad dynamic range (10-2000 ng), with correlation coefficients (r2 ) between 0.99 and 1.00 (Figure 3B). The recovery of cholesterol from the sperm matrix was estimated to be 92 ± 8%. The repeatability of the LC-MS method for the determination of cholesterol, 7β-OHC and 7-KC was estimated from the coefficient of variation (CV) on intra- and inter-assay measurements, yielding values ranging from 9.5% to 15.00% respectively. The detection limits were determined as 9.2 ng for cholesterol, 11.9 ng for 7β-OHC and 6.0 ng for 7-KC.
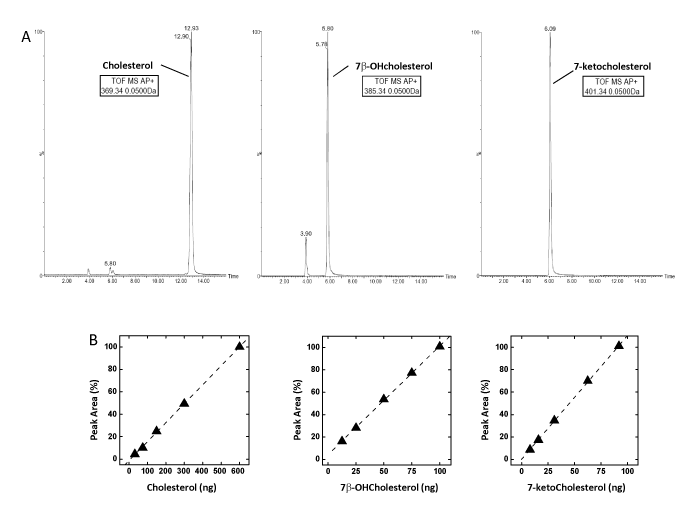
Figure 3: A. LC-MS analysis of a standards mixture of Cholesterol, 7β-OHcholesterol and 7-ketocholesterol 1:1:1 (250-350 pmoles) eluted from a reversed phase column under isocratic conditions (acetonitrile : methanol, 65:35 v/v) and recorded at each molecular ion (respectively 369.35 m/z for cholesterol, 385.33 m/z 7β -OHcholesterol and 401.33 m/z for 7 –KC)
B. Calibration curves for the quantitative determination of cholesterol (30-600 ng), 7β-OHC (12-100 ng) and 7-KC (8-100 ng). Curves were obtained by injecting increasing amount of each (oxy)sterol standard in the LC-MS apparatus and plotting the integrated area of the corresponding mass peak vs the amount of the injected analyte. The peak area relative to the largest amount of analyte injected was taken as 100%. The equations of the calibration curves were obtained by linear interpolation of the LC-MS data.
Application of the method to real samples
Representative LC-MS analysis of oxysterols in sperm cells from healthy donors is reported in Figure 4A. The developed method largely allowed the unambiguous identification of 7β-OHC and 7-KC and the correspondent quantification. A scatter plot of the levels of the three sterols detected in our group of healthy volunteers is reported in Figure 3B. Results are shown as ng of sterol per million of cells. A wide distribution of these sterol species was observed, ranging respectively from 2.9 to 25.9 ng/106 cell (mean value 9.5 ± 7.8 ng/106 cell) for Chol, from 0.64 to 2.88 ng/106 cell (mean value 1.61 ± 0.70 ng/106 cell) for 7β-OHC and 0.23 to 1.26 ng/106 cell (mean value 0.82 ± 0.34 ng/106 cell) for 7-KC. Subjects with diagnosis of normozoospermia had significantly lower sperm levels of Chol compared to oligozoospermic featured by total sperm count ≤ 39 × 106 cells/ejaculate (respectively 3.5 ± 0.7 ng/106 cell vs 11,7 ± 7,3 ng/106 cell, P=0.048). Also oxidized derivatives of Chol were found at higher levels in sperm cells from oligozoospermic subject compared to normozoospermic (respectively: for 7β-OHC 0.81 ± 0.17 ng/106 cell normozoospermic vs 1.82 ± 0.58 ng/106 cell oligozoospermic, P=0.006; for 7-KC 0.36 ± 0.16 ng/106 cell normozoospermic vs 0.93 ± 0.23 ng/106 cell oligozoospermic, P=0.001).
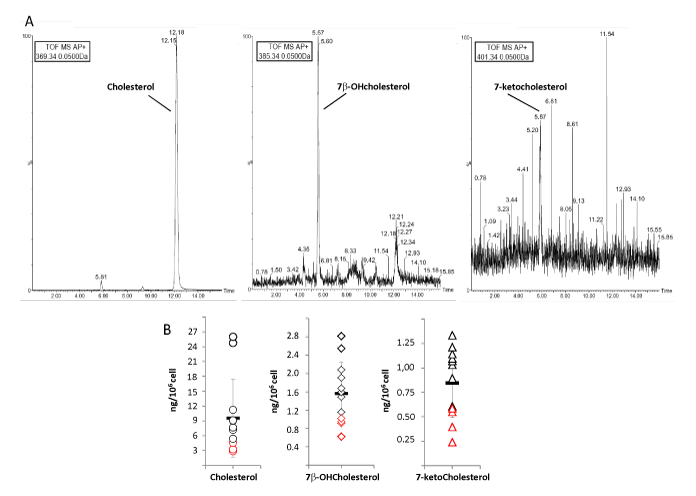
Figure 4: A. Representative LC-MS analysis of Cholesterol, 7β -OHcholesterol and 7-ketocholesterol extracted from a real sperm sample obtained from a male donor. LC traces are recorded at each molecular ion (respectively 369.35 m/z for cholesterol, 385.33 m/z 7β -OHcholesterol and 401.33 m/z for 7 –KC).
B. Scatterplots showing the quantification of cholesterol (circles), 7β -OHcholesterol (squares) and 7-ketocholesterol (triangles) in sperm samples from 12 male donors. Data are reported as ng of sterol/106 sperm cells. In each scatterplot are also reported the mean value (horizontal bar), standard deviation (whiskers) and data from normozoospernic samples (red indicators).
Discussion
In this study we describe the development of a fast and reliable method, using LC-MS analysis, for the quantification of Chol and main oxysterols in human sperm cells. Furthermore, despite the small sample size, these preliminary data suggest that disorders of the spermatogenic process exert major impacts on the composition, architecture and function of the sperm membrane through the modulation of the sterol pattern. Furthermore, considering that 7β-OHC and 7-KC are oxidative derivatives of Chol suggested to have a role in sperm function [3, 13], their quantification in sperm cells may represent an attractive marker for the evaluation of the exposure of the test is to environmental factors of toxicological importance such as pollutants or heat stress [18,19] or conversely, to verify the efficacy of a therapy with antioxidants. To this regard, further studies on larger cohorts of finely characterized patients are warranted to address these hypotheses.
Conclusion
We describe a robust LC-MC method for the detection on cholesterol and main oxysterols in human sperm cells. This method represents a useful tool to investigate the role of oxysterols, as oxidative derivatives of Chol, in sperm physiology.
Acknowledgment
The expert technical assistance of Dr. Marino Bellini (Department of Pharmaceutical and Pharmacological Sciences, University of Padova) with MS analyses is gratefully acknowledged.
Article Information
Article Type: Research Article
Citation: Šabović I, De Toni L, Tescari S, De Filippis V, Menegazzo M, et al. (2017) Detection of Cholesterol and its Oxidized Derivatives in Human Sperm Membranes through a Fast and Reliable LCMS Method. J Clin Lab Med 2(1): doi http://dx.doi. org/10.16966/2572-9578.109
Copyright: © 2017 Šabović I, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
Received date: 09 Feb 2017
Accepted date: 02 Mar 2017
Published date: 07 Mar 2017