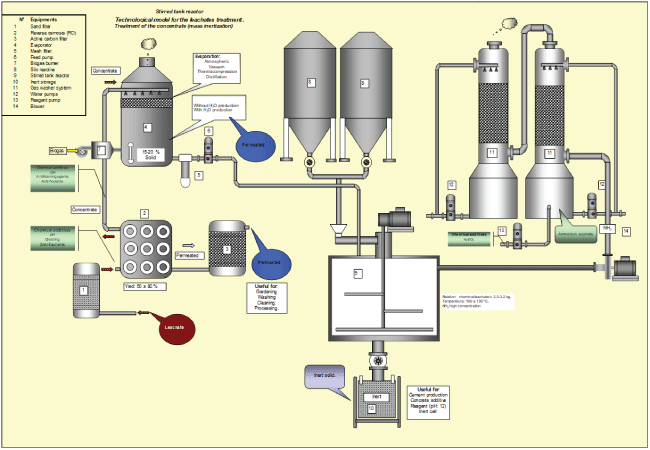
Figure 1: Flow diagram: Bulk Inertization Technology (BIP)-Stirred tank reactor


Joaquin Reina Hernández*
Water Treatment Department, Energy & Waste Company, Condorchem Group, Spain*Corresponding author: Joaquin Reina Hernández, Water Treatment Department, Energy & Waste Company, Condorchem Group, Spain, E-mail: jreina@ewtech-ing.com
One of the most complex aspects of the global management of a Municipal Solid Waste (MSW) landfill is the leachates treatment. This liquid is mainly obtained from the degradation of organic matter. This added to other liquids present in the waste, including rainwater and others, constitutes a complex and highly polluted water. There are many techniques available for leachates treatment but the only technique that enables an inert waste to be obtained is inertization. The present article is focused on the study of a new technology for the inertization of leachates derived from landfills. This technology is based on the contact between dispersed phases. A pilot plant was built to undertake the study and with the aim of observing the behavior of the process and its yield. The overall results of this study indicate that this technology facilitates the contact between the flows (leached-reactive), significantly improving the processes of heat transport and mass transfers that take place during the development of the reactions. This allows operating costs to be minimized by reducing the amount of reagents used making the process more attractive from an economic point of view.
Wastewater; Technologies; Leachate; Inertization
One of the most complex aspects in the comprehensive management of a landfill or treatment plant for Municipal Solid Waste (MSW) is leachate. Leachate makes up the liquid part that filters through solid waste disposed at the landfill, and extracts materials in solution or suspension. The leachate in turn is determined by the combination of elements found in the composition of the solid waste, the way in which the landfill is operated and the climate conditions at the site. Its composition varies according to the age of the landfill, type of waste deposited and prior any sampling. Therefore, landfill leachate must be suitably managed, according to its physical and chemical characteristics and its composition. The composition of landfill leachate depends on the type of landfill (hazardous, non-hazardous and inert) and within the same landfill this varies depending on its time in operation [1].
In general, leachate shows high levels of contamination, mainly due to:
- High concentrations of organic material
- Nitrogen concentrations, mainly in the form of ammonium.
- Other salt concentrations, mainly chlorides and sulphates.
-Presence of heavy metals which level depends on the age on landfill. High when the landfill is young and low when the landfill is old.
Furthermore, issues surrounding the suitable handling of solid waste does not just affect human health, but are also related to environmental contamination, as well as the pollution of soil and watercourses both on the surface and underground, which are often the source of drinking water.
Therefore, this wastewater must be treated to reduce its level of contamination before being released into natural sources and prevent damage of the underground water.
In Spain the leachate situation hasn’t changed since more 10 years. For this reason, there is a serious problem with this topic yet, not only, for the big number of the landfill that exits also for the different technologies used. In the last ten-year techniques like Inverse Osmosis (RO) and evaporation and its combination with other procedures are very common.
Despite them, the technology before mentioned don’t give a final solution to the leachate problem, only are useful for reduce the leachate level, due to produce a concentrated with high level of contamination that need other types of treatment. This concentrated in many cases is carry to a Wastewater Treatment Plant (WWTP) where it treats together with the urban water and, in this way; many landfills give a solution at this problem.
However, the inertization process, as technique for the leachate treatment has had a low application in Spain. The main problem presented with application the inertization technology has been its high operation cost due to the reagents used quantity and the quality of the final product given the technology used (bulk mix).
Based on this has been developed this preliminary, study at pilot plant scale about a new leachate inertization process based on Dispersed Phase contact (DIP) with the aim to improve the inertization process in terms of operational costs and product quality.
Leachate treatment is very similar to waste water treatment, although with some difference due to its high organic load. Financial and technical aspects dictate the most suitable type of treatment in each specific case, and a combination of many is quite common.
The most used technologies use different combinations of biological and physical-chemical treatments/techniques. The flow rates to be treated, the pollution load, and the limits of the discharge regulations, are the parameters that finally determine whether a single technique may be enough, or if it will be necessary to resort to more complete techniques/treatments, such as a membrane filtering process after the biological process [2-5].
Recently, concentration into different components through evaporation and spray drying has been introduced as a treatment technique.
Independently of the treatment/technique applied, be it membrane or evaporation, two fractions will be obtained: one clean, the permeate and one dirty, the rejected. The latter is where all the impurities of the treated water reside. Therefore, the rejected or concentrate has a greater environmental impact than the water from which it comes; hence, it must be treated in order to minimize its volume, re-evaluate its comprising sub products, or inertize it for its future handling and disposal within the facility itself.
All the aforementioned technologies can be considered secondary treatments, given that they are intended to minimize this type of waste (residual water) and not convert it into a material that is apt for handling and future disposal.
Hence, today there is talk of three types of landfill leachate treatments. The first are intended to prepare and condition the leachate for its entry into a second treatment stage. The second are aimed at minimizing them either physically, i.e. membrane filtration, where inverse osmosis is noteworthy, or by thermal means such as evaporation and spray drying, or a combination of both.
The third treatments are responsible for directly transforming leachate (with prior conditioning) or concentrates coming from secondary treatments, into an inert solid for handling and dumping at the landfill itself or facilities prepared for this activity. Within these treatments inertization is found.
The purpose of this work is to carry out a pilot plant scale study of the new technology for leachate inertization based on the contact between disperse phases (liquid-solid), with the aim of discovering the feasibility of this technology and the efficiency of the inertization process.
Inertization is a physical-chemical process to convert effluent into a solid waste with structural integrity, while preventing the migration of contaminants by reducing their mobility.
Inertization is a technique widely used in the management of hazardous waste and includes two operations:
Process that uses a series of reagents to:
- Reduce the hazardous nature of the waste
- Minimize the migration speed of contaminants to the environment
- Reduce the toxicity of its components
- The concept of securing is commonly used as a synonym for stabilization. Stabilization is carried out by adding reagents which:
- Improve the handling and physical characteristics of the waste
- Decrease the surface area through which the transfer or loss of contaminants can occur
- Limit the solubility of any contaminant present in the waste
- Reduce the toxicity of contaminants
The process of adding reagents is described with the aim of:
Solidifying the waste, increasing its resistance and decreasing its compressibility and permeability.
In the broadest sense of the word, inertization can be defined as the technique used to transform hazardous waste into inert waste; understood as waste that does not experience significant physical, chemical or biological transformations [6-7].
In common practice, the aim is to achieve waste that can be managed as non-hazardous waste.
Inertization is mainly applied to inorganic paste-like and solid hazardous waste, and in a lesser extent to organic waste for the following reasons:
When an organic waste cannot be reused or recycled, the energy contained therein is usually used as fuel for special facilities such as cement and clay work.
Organic components hinder the solidification process of cement (most used reagent for said purpose).
In the case of leachate and/or leachate concentrate from landfills or municipal solid waste treatment plants, inertization can be carried out by adding Calcium Oxides (CaO) and cement to the same (reagents commonly used for this purpose), in equipment that allows it to be mixed.
The main purpose of the inertization process using calcium oxide (CaO) is to obtain calcium hydroxide or slaked lime (CaOH). Through the formation of this compound (CaOH) reactions take place that enable chlorine (Cl), sulphates (SO4) and other compounds present in the leachate to be captured.
The reactions taking place in the leachate inertization process the following are noteworthy.
1. CaO + H2O ↔ Ca (OH)2∆H=-1140 kJ/kgCaO
a) Ca (OH)2 + Cl2 → CaOCl2 + H2O
b) Ca (OH)2 + H2SO4 → CaSO4 + 2H2O
The first reaction corresponds to the hydration of calcium oxide, i.e. the formation of calcium hydroxide or slaked lime, which is very common in this kind of process. This reaction is exothermic. The slaked lime production process is carried out at a temperature of around 150°C.
This reaction heat causes a rise in temperature, both in the solid product created (inertized product) and the gas phase resulting from the process. At the same time, this temperature rise is an indicator of the quality with which the process is taking place.
Due to the ammonium (NH+4) content of the leachate and the process’s operating conditions, i.e. rise in temperature and pH, ammonium (NH+4) is released in the form of ammonia (NH3) due to the rupture of the chemical balance taking place. This ammonia is released as gas, forming a gas flow that must be treated.
Reactions a and b are secondary reactions, which appear in the inertization process and enable the capture of chlorine (Cl) and sulphates present in the leachate, forming part of the crystalline net of the calcium sulphate (CaSO4) and calcium chloride CaOCl2 components formed, which are retained in this form in these solid compounds. Both the chlorine (Cl) and sulphates (SO4) remain trapped in the crystalline net of these components, which prevents them from moving or migrating to any other medium.
In today’s market within leachate inertization technologies, bulk inertization can be found. Bulk inertization phase (BIP). Mixture of leachate (liquid phase) + bulk reagents (solid phase). (Figure 1,2).

Figure 1: Flow diagram: Bulk Inertization Technology (BIP)-Stirred tank reactor
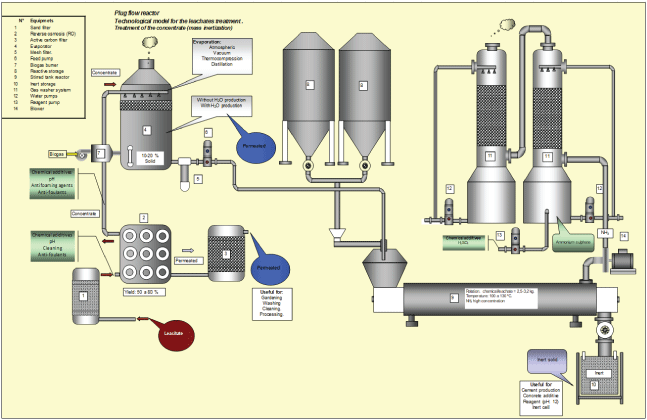
Figure 2: Flow diagram Bulk Inertization Technology (BIP)-Plug flow reactor.
This type of inertization is characterized by the leachate get into contact with a reagent for its inertization inside equipment (reactor). In this case, the phases (liquid-solid) are mixed in suitable proportions to form the inertized product. Some of the most common equipment/reactors are: drum mixers or screw conveyor that running continuous or discontinuous (Figure 1 stirred tank reactor) or screw conveyor (Figure 2 plug flow reactor). In the case of bulk flow reactors, the efficiency of contact between phases (solid-liquid) is limited by the intensity of the mixture (contact between phases), so mass transport and heat coefficients are limited, in order to considerably decrease (high resistance to the development of reactions) what leads to long residence times and in general low inertization efficiency. To tackle this disadvantage and others related to solid-liquid contact, high amounts of reagents are used per unit mass of waste (leachate-liquid).
In the study of bulk inertization processes, reagent/leachate weight ratios are reported by different Spanish entities ranging from 2.5 to 3.2. These depend in a great extent to the prior treatments received by the leachate and the type of reactor used (complete mixture or piston flow).
All of this involves an increase in technological operating costs due to the high amount of reagent needed to carry out the inertization process. This aspect, combined with low quality inertization, makes the application of this technology for treating landfill leachate less attractive in many cases.
This reaction results in the release of a gas with a high concentration of NH3 accompanied by H2O vapor. This is due to the high concentration of ammonium (NH+4) held by this type of water (leachate), caused by the displacement of balance, both chemical (rise in pH) and technical (rise in temperature) that occurs, which favors the release of ammonium (NH+4) in the form of gas (NH3).
This gas is washed to eliminate the NH3 and any other type of component present in the gas flow formed by the absorptiondesorption system before being discharged to the atmosphere. Figures 1,2 show flow diagrams of this technology.
In both cases, inverse osmosis is combined with evaporation. The latter stage is to make the concentrate from the membrane separation more concentrated. The resulting concentrate passes to the inertization process, where using the appropriate reagents it is converted in an inert solid product for handling and discharge to the landfill.
Among other advantages of this type of inertization, the following should be noted: Constructional simplicity and low fixed costs.
Among its disadvantages the following should be noted: high Kg Reagent/Kg Leachate ratio which leads to high operating costs, given the type of reactor (mixed) used has high power consumption, and due to the contact established between the phases (liquid-solid), low mass transport and heat coefficients arise which increase the resistances to the reactions taking place and thereby affect the efficiency of the inertization process. In general, this type of technology does not comply with suitable operating conditions for carrying out the process that takes place.
Another disadvantage that should be noted, is the high concentration of NH3 appearing in the gas phase generated by the process, due to the rupture of the chemical balance due to the rise in pH and temperature of the mixture. The rise in temperature is caused by the heat given off during the formation of the slaked lime (CaOH). Exothermic reaction.
In order to avoid all the disadvantages of bulk inertization, mainly the amount of solid reagents to be used, a new model of contact between the phases, involved on the process, is proposed. In this case the contact model in dispersed phase. This model consists of atomizing the liquid and spraying the reagents inside a chamber heated with air to the reaction temperature recommended by the process [8-10].
A pilot plant is a processing plant on a reduced scale. The aim of designing, constructing and operating a pilot plant is to obtain information about a determined physical or chemical process, which enables the determination of whether the process is technically and financially viable, as well as the establishment of the optimal operating parameters of said process for the later design and construction of the plant on an industrial scale. The aim is also to obtain models that can simulate the behavior of the process in determined operating conditions.
The experiment was carried out with three leachate concentrations from three different landfills managed by the Ferrovial Group. Each concentrate was obtained differently, i.e. from different kinds of treatments including the following: inverse osmosis, two-stage inverse osmosis and inverse osmosis plus evaporation by thermalcompression.
With prior mass inertization tests at laboratory scale that serve to determine the different operating parameters of the process; the pilot plant was constructed to carry out this study, with the aim of determining:
- Whether the process is technically and financially viable.
- The results reported by inertization when disperse phases come into contact.
- Information about carrying out the process.
- The operating parameters of said process for posterior design and construction of the plant at an industrial scale.
- Models that can simulate the behavior of the process in determined operating conditions.
Figure 3 shows the facility constructed for this purpose. It shows the different parts included in the facility.

Figure 3: Pilot plant for testing.
Part components of ILFD technology
1. Inertization chamber.
- Diameter × height=800 × 4000 mm.
2. Liquid atomization nozzle.
- Cone type nozzle narrow angle opening=45°
- Average drop size 85 micron.
3. Reagent spray
- Diameter × height=80 × 400 mm.
4. Reagent dispenser.
- Volumetric type.
- Speed adjuster
5. High pressure pump.
- Peristaltic type.
6. Gas filtering system.
- Laple cyclone=250 × 500 mm.
7. Air heating chamber.
- Diameter × length=600 × 1000 mm
8. Burner
Fuel=diesel.
9. Fan.
-Centrifuge type.
10. Control and automation system. The pilot plant has a control panel that enables the control of the different
operating variables of the plant including:
- Input air temperature in the inertization chamber and process output.
- Flow of leachate, injected air and generated gases.
- Inertization chamber pressure.
- Amount of reagent to dispense.
In general, the reagent used for the inertization of landfill leachate is a mixture of calcium oxide+cement at 50%, in the case of inertization in disperse phase micronized calcium oxide is used, which facilitates the spraying of reagent by the heating agent inside the inertization/ mixing chamber.
The atomizer used for the atomization of the leachate, i.e., the formation of drops, is a hollow cone nozzle with a narrow angle, through which the reagent is sprayed with the help of the incoming hot air flow. This sprayer has 300 mm on the nozzle.
The concentrate (leachate) used for the tests was obtained from different landfills and obtained in different ways, i.e., from different kinds of treatments included the following: inverse osmosis, two-stage inverse osmosis and inverse osmosis plus evaporation by thermalcompression.
Figures 4-6 show flow diagrams of the treatments to which the different leachate were subject according to their origin (purpose of these tests).
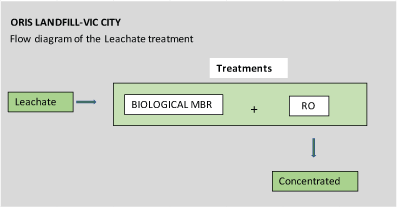
Figure 4: Flow diagram Treatment of leachate from the Orís landfillVic City.
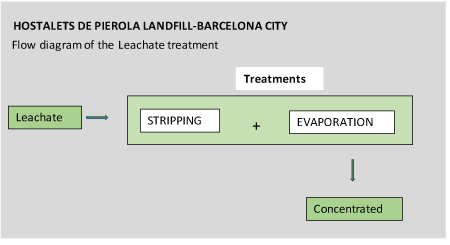
Figure 5: Flow diagram Treatment of leachate from the Hostalets de Pierola landfill. Barcelona
Figure 6: Flow diagram Treatment of leachate from the Santa María de Palautordera landfill. Barcelona.
Table 1 shows the chemical characteristics of each leachate according to its origin and treatment type which is the purpose of this pilot plant scale study.
| Leachate for studying |
Lix 1 Ref: 3970 |
Lix 2 Ref: 3972 |
Lix 3 Ref: 3974 | |
| Parameters | Units | Results | Results | Results |
| Ammonium | mg NH4/l | 9.634 | 147 | 10.800 |
| Dry Waste | mg/l | 55.750 | 46.620 | 38.400 |
| Material in Suspension | mg/l | 713 | 126 | 10.040 |
| Conductivity (at 20°C) | µS/cm | 54.755 | 40.924 | 75.700 |
| pH | Units of pH | 8,0 | 7,0 | 5,3 |
| Chlorides | mg Cl/l | 20.904 | 16.040 | 48.584 |
| Sulphates | mg SO4/l | 11.008 | 803 | 36.013 |
| T.O.C | mg Cl/l | 13.230 | 2.487 | 14.628 |
Table 1: Shows the characteristics of each leachate according to its origin
and treatment type which is the purpose of this pilot plant scale study.
Leach 1=Palautordera; Leach 2= Oris; Leach 3=Hostalets de Pierola
The high level of ammonium, chloride and sulfate concentrations in the Leachate in these cases is due to mainly at four reasons. The high level of the organic matter content present in the waste, the age of the landfill, haven’t having done perform a previous a sorted of the waste before being deposit in the dump and the different process involved in its treatment like Reverse Osmosis (RO) and evaporation mainly.
From table 1 it can be noted too that the leachates that have been treated by evaporation are the ones with the highest concentrations of their hazardous components and conductivity
Test operating conditions: To carry out the experiment on a pilot plant scale, a set of tests were carried out prior to establishing the operating conditions, using the leachate from the Orís landfill as the leachate for the tests, due to its lower contaminant load with regards to the presence of chlorine (Cl-) and sulphates (SO4-2) and due to its completely neutral pH, i.e., equal to 7.
These prior tests were used to establish the operating conditions regarding the flow rate of leachate, reagent and air, and the working temperature for tests on the different leachate. For the leachate from the Orís landfill a flow rate of leachate 12 lit/h, reagent 7.05 kg/h and air 271 m3 /h at a temperature of 175°C were the suitable conditions for inertization of the leachate.
The reagent used is a mixture of 50% micronized calcium oxide with an average particle size of 85 micron (µm) and cement that is sprayed inside the inertization chamber by coming into contact with a hot air stream, and to atomize the leachate a hollow cone nozzle with a narrow angle of 60º was used that operates at a pressure of 18 bar and delivers an average drop size of 85 micron (µm).
Before entering the pump, the leachate is filtered with a rubber filter to remove any kind of solids that may obstruct the atomization nozzle and cause it to operate poorly. The supply flow rate for the different tests ranges from 10 to 15 lit/h.
Plant operation for tests: Plant operation begins by turning on the fan, which creates air flow through the heating chamber, inertization chamber and cyclone. Meanwhile, the depression needed to start up the burner that is supplied with diesel is created.
Before starting the burner, the air heating temperature is preset to 180°C (set point) on the control panel. Once this operation is carried out, the burner is turned on and the working temperature is awaited, i.e. 80°C.
Once operating conditions are reached the reagent dispenser is started, with its operating speed previously set, and in this way the amount of reagent supplied always is known. Once the dispenser is started, two (2) minutes are awaited to turn on the leachate injection pump, while controlling both its flow rate 12.5 lit/h and pumping pressure that is found in the range of 18 bar.
Analytic and control method: All parameters related to leachate, inertized products and gas flow generated by the inertization process, are determined by a laboratory accredited for the implementation of these tests, considering the standard methods established for the characterization of these kinds of effluents. Lab AT (Ambiente Tecnología Consultores). Barcelona
Analysis carried out on samples of inertized products from different landfills.
1) Loss 105°C: weight loss experienced by the original sample after drying at 105°C phase (PNA065).
2) Leachate test: According to standard UNE EN 12457- 4 (sample weight: 97.5 g, water volume: 892 ml). The initial pH (PNA004) and conductivity (PNA005) were determined during the first 15 minutes of agitation.
3) Chlorides: Analysis carried out for the valuation (PNA006). Determined on the leachate.
4) Total solids dissolved (STD)/dry residue: determination of the total solids of the leachate by drying and gravimetry (PNA086).
All tests had an implementation time of one (1) hour. During the tests the following working variables were measured:
- Supplied leachate flow rate
- Supplied reagent flow rate (rpm)
- Injected air flow
- Resulting gas flow
- Working temperature.
- Resulting product weight (inertized product) for said purpose, it was placed both at the bottom of the inertization chamber and the bottom of the cyclone, being recipients for the re-collection of the resulting product (inertized product). This product was weighed on a scale dedicated to this purpose at the end of each test.
- Volume of fuel used for air heating.
Figure 7 shows the results reached in the inertization of the different leachate using disperse phase contact technology. In all three cases, the result obtained was a white powder, although in different tones. The greatest proportion of inertized product was obtained at the bottom of the inertization chamber and another smaller fraction at the bottom of the cyclone. The gas flow result from said process had a bitter-acid odor and certain ammonia scent.

Figure 7: The results obtained in the inertization process of the different leachate under study.
To discover the efficiency of the inertization process on-site leachate tests were carried out. For said purpose, equal parts of water and inertized product were mixed and left to rest for some time. Then the color and transparency of the aqueous extract swimming on top of the surface of the solid fraction was observed as a quality indicator of the process.
The pH was also measured, which enabled us to discover the alkaline level of the extract.
These tests always enabled the amount of reagent to be injected into the inertization chamber to be adjusted.
According to the origin of the leachate, Figure 8 showed the coloration and transparency of the aqueous extract swimming on top of the solid fraction for a reagent/leachate weight ratio=0.77.

Figure 8: The results obtained from the on-site leachate process for the different leachate under study.
Figure 8 shows the results of the on-site leachate tests.
This proves the efficiency of the inertization was different in the three cases. All evidence indicated that the leachate from the landfill of Orís had the best inertization quality, while the remaining were of lesser quality, which proves the migration of components from the solid phase to the resulting aqueous extract.
Samples of the three inertized products from the different leachates were sent to a laboratory accredited for their analysis, while the gas phase analysis was carried out on site.
Tables 2 show the results of the analysis reported by the laboratory accredited to carry out these kinds of analyses, both inertized product and the gas phase it generated.
Table 2 shows that the inertized product of the Palautordera landfill exceeded the limits corresponding to the COD and Total Dissolved Solids (TDS) required as limit values for non-hazardous waste (for an aqueous extraction ratio of L/S=10 L/kg).
| Results: Pilot plant scale tests | Ref: 3951 | Lix 1 Ref: 3971 | Lix 2 Ref: 3973 | Lix 3 Ref: 3975 | |
| Parameters Waste Analysis | Units | Results | Results | Results | Results |
| Loss 105°C | % | 7,7 | <0,2 | <0,2 | <0,2 |
| Flash Point | °C | >75 | >75 | >75 | |
| Leachate Analysis | |||||
| Initial pH | pH Units | 12,5 | 12,5 | 12,5 | 12,5 |
| Initial Conductivity | µs/cm at 25°C | 13.180 | 14.920 | 13.480 | 27.930 |
| Final pH | pH Units | 12,6 | 12,6 | 12,6 | 12,6 |
| Final conductivity | µs/cm at 25°C | 13.370 | 14.980 | 13.560 | 13.550 |
| T.O.C | mgC/Kg s.m.s | 4.850 | 590 | 11.130 | |
| Antimony | mg Sb/Kg s.m.s | <0,2 | <0,2 | <0,2 | |
| Arsenic | mg As/Kg s.m.s | <0,2 | <0,2 | 0,8 | |
| Barium | mg Ba/Kg s.m.s | 4.7 | 7.1 | 3,4 | |
| Cadmium | mg Cd/Kg s.m.s | < 0,1 | < 0,1 | < 0,1 | |
| Copper | mg Cu/Kg s.m.s | < 0,6 | < 0,6 | < 0,6 | |
| Total Chromium | mg Cr/Kg s.m.s | 1,8 | 1,1 | 2.6 | |
| Mercury | mg Hg/Kg s.m.s | < 0,01 | < 0,01 | < 0,01 | |
| Molybednum | mg Mo/Kg s.m.s | 0,3 | 0,2 | 0,4 | |
| Nickel | mg Ni/Kg s.m.s | < 0,6 | < 0,6 | 1,4 | |
| Lead | mg Pb/Kg s.m.s | < 0,6 | < 0,6 | < 0,6 | |
| Selenium | mg Se/Kg s.m.s | < 0,2 | < 0,2 | < 0,2 | |
| Zinc | mg Zn/Kg s.m.s | < 0,6 | < 0,6 | < 0,6 | |
| Ammonium | mg NH4+/Kg s.m.s | 150 | < 40 | 940 | |
| Chlorides | mg cl-/Kg s.m.s | 7.930 | 11.220 | 6.350 | 58.460 |
| Homogenous CDO | mg O2/Kg s.m.s | 12.950 | 1.470 | 29.820 | |
| Sulphates | mg SO2-4/Kg s.m.s | 5.540 | 1.670 | 32.830 | |
| Total Dissolved Solids (TDS) | mg /Kg s.m.s | 59.000 | 76.000 | 47.000 | 210.000 |
Table 2: Results of the analysis of the leaching of the inertized product of the three samples (leachate) studied.
Leach 1=Palautodera; Leach 2=Oris; Leach 3: Hostalets de Pierola
Table 2 shows the results achieved when inertizing the three types of leachate, with the technology of contact between phases (DIP). This result allows corroborating that the technology is appropriate for the inertization of these leachates. From table 2 it is also noted that, the results achieved differ depending on the type of treatment received by the leachate, when the leachates were submitted to the inertization process using the same ratio of reagent, that is, Kg Reagent/Kg Leachate ratio=0.77.
The table 3 shows a comparison regarding the percentage of reduction of its main polluting components.
| Comparative Results | Lix 1 | Inertizated Ref: 3971 | Lix 2 | Inertizated Ref: 3971 | Lix 3 | Inertizated Ref: 3971 | ||||
| Parameters | Units | Result | Reduction | Result | Reduction | Result | Reduction | |||
| In | Out | (%) | In | Out | (%) | In | Out | (%) | ||
| T.O.C | mg C/Kg s.m.s | 13.230 | 4.850 | 63,34 | 2.487 | 590 | 76,28 | 14.628 | 11.130 | 23,91 |
| mg NH 4+/Kg s.m.s | 9.634 | 150 | 98,44 | 147 | 40 | 72,79 | 10.800 | 940 | 91,30 | |
| mg Cl-/Kg s.m.s | 20.940 | 11.220 | 46,33 | 16.040 | 6.350 | 60,41 | 48.584 | 58.460 | 16,89 | |
| mg SO 42-/Kg s.m.s | 11.008 | 5.540 | 49,67 | 803 | 1.670 | 51.92 | 36.013 | 32.830 | 8,84 | |
Table 3: Comparative results between the raw leachate and treated leachate by inertization process, based on the main harmful components.
Leach 1=Palautodera; Leach 2=Oris; Leach 3=Hostalets de Pierola
From table 3 the highest percentages of leachate contaminants reduction were obtained with the leachate from the Oris landfill, which is the leachate that had the lowest degree of concentration, since it had only been submitted to a process of concentration by reverse osmosis, followed by leachate from Palautordera and finally the one corresponding to Hostalet de Pierola.
However, only the Palautordera leachate is in which all harmful components were reduced, so we can speak of a complete inertization, while in the Oris leachate there was an increase in the Sulphates while in the Hostalet de Pierola there was an increase in the Chlorides. In these leachates we could not speak of a complete inertization.
It should also be noted that the pollutants with the highest degree of reduction were T.O.C, Ammonium, Chlorides and, to a lesser extent, the Sulphates. This showed that the yield of the different reactions that took place and their possible reaction rates were different.
Another of the analyses carried out was carried out on the gaseous phase resulting from the inertization process with a view to knowing the possible contaminants and their concentrations produced by the inertization process.
The table 4 shows the results of the analysis carried out on the gaseous phases resulting from the inertization process.
| LIX | Parameter | Units | Results |
| Lix 1 | Ammoniac | (mg/m3) | 266 |
| Lix 2 | Ammoniac | (mg/m3) | 4,8 |
| Lix 3 | Ammoniac | (mg/m3) | 293 |
| Lix 1 | Hydrogen Sulphide | (mg/m3) | 10,0 |
| Lix 2 | Hydrogen Sulphide | (mg/m3) | <1,0 |
| Lix 3 | Hydrogen Sulphide | (mg/m3) | <1,0 |
| Lix 1 | Olfactometry | UOE/ m3 | 11.585,00 |
Table 4: Results of the analysis of the gas phase generated during the inertization process of the three samples (leachate) studied. Determination of
ammoniac, hydrogen sulphide and olfactometry.
Leach 1=Palautodera; Leach 2=Oris; Leach 3=Hostalets de Pierola
From Table 4 it can be noted with respect to the emission of ammoniac (as ammonium) that the concentrations of these components in the gas phase were higher in those more concentrated leachates, i.e., those that had been subjected to an evaporation process. Therefore, the ammoniac (as ammonium) that was not trapped in the inertizating process was released in the gas phase due to the rupture of the equilibrium, both chemical and thermodynamic that took place.
Regarding the emission of hydrogen sulphide (as sulphide), it can be stated that they were low, therefore, it is possible to argue that most of the Sulphates were trapped in the inertizate which made the concentration of this component in the gas phase minimal.
This allows us to state, in a certain way, that the sulphates were more susceptible to this inertization process than the ammoniums, which could have been due to the differences in reaction rates.
Referring to (table 2) to the case of the inertized product of the Orís landfill, only the limit corresponding to the COD was exceeded, while in the case of the Hostalets de Pierola landfill the limits corresponding to chlorides, COD, sulphates and Total dissolved solids were exceeded.
It must be considered that the concentrate under study was obtained in different manners and from different sources, and from different landfills, so differences were expected in their results when treated under the same operating conditions, in this case the reagent/leachate weight ratios=0.77.
According to the data reported for the leaching tests, on table 2 it can be proposed that the best results were reached using the inertized product from the leachate from Orís, i.e., where the level of solid concentration was lower. In general, there was a reduction in the concentration of contaminants present in leachate from said inertized products.
Table 5 shows a quantitative and qualitative estimative comparison between the current technologies for leachates inertization from landfills (BIP vs DIP) related with different operational parameters, including investment and operation costs.
| Investment | Reagent Consumption Ratio | Energy Consumption | Operation Cost | ||
| Electric | Thermal | ||||
| BIP | Low | 2a3kgreact /Kglix2 | High | Low | High |
| DIP | High | O,75a 1,2 Kgreact /kglix2 | Low | High1 | Low |
Table 5: Quantitative and qualitative comparison between DIP and BIP technologies for leachates inertization in Spain.
1- It can be used biogas or heat of Engine exhaust gases. For that reason the cost could go down
2- Depend on the origin of the leachate (reverse osmosis or evaporation)
Table 5 shows the differences in investment costs, which are mainly due to the dimensions of the inertization chambers. This is related to the type of contact between the phases. In the case of Disperse Phase (DPI) high contact surface was required and for them equipment with a big work volume.
However, due to this large contact surface, which is related with way in which both the reactants and the leachate were introduced, the transport processes that took place (mass and heat) were favored and for that reason a smaller amount of reagents was required to reach the same product quality.
In the case of the consumption of electrical energy, due to the way in which the contact between the reagent and the leachate occurred in the DIP technology, the electricity consumption is much lower than the BIP (bulk phase) technology.
In the case of BIP technology, there is a stage of sludge formation that requires a lot of electrical energy for mixing and transport of the material. This sludge then dries up due to the heat that is released due to the reaction that takes place to produce the solid inertized.
The DIP technology requires hot air for its work, since the process takes place at a temperature between 150°C and 200°C. Therefore, the consumption of thermal energy is high. This consumption can be minimized if residual sources of calorific energy are used, such as exhaust gases from the cogeneration system or the combustion of the biogas.
All this causes that the operating costs of the DIP technology are lower than of the BIP technology.
Ammoniac and Hydrogen Sulphide: Table 4 shows the results of the determination of ammoniac (as ammonium), hydrogen sulphide (as sulphide) in the studied samples of the atmospheric emissions of the leachate inertization process.
So, regarding atmospheric emissions, it should be noted that the ammoniac levels (NH3) present in the gas phase from the inertization of leachate from the landfills of Palautordera and Hostalets de Pierola are apparent, and although there is no official limit for this parameter in current legislation, emission limits under 100 mg/m3 are usually required.
Regarding the hydrogen sulphide, the value obtained in the case of gases from Palautordera is 10 mg/m3, which coincides with the limit required in paragraph 27 of Appendix III of Decree 833/1975, while currently for many environmental infrastructures the limit is set at this value or lower.
Based on the results of this study, the following conclusions are drawn.
Disperse Phase Inertization (DIP) is a process suitable for the treatment of leachate coming from landfills and Municipal Solid Waste (MSW) treatment plants due to is useful to get an inert solid with a high stability and with lower operational cost than bulk phase inertization (BIP).
This is due to a.
- Lower reagent consumption.
- Lower energy consumption.
Everything indicates that the level of concentration or the origin of the leachate, have influence in the inertization quality and in the reagent/leachate weight ratio to achieve a suitable inertization process.
Of all the hazardous components studied, the most sensitive to the stabilization process are the T.O.C and Ammonium for this kind of reagent, while to a lesser extent, the Chlorides and Sulphates.
This technology offers a new solution to the leachates management generated in municipal solid waste landfills (MSW).
The author would like to express their gratitude to the Ferrovial Servicio Group and all staff who cooperated on the development of this project.
Download Provisional PDF Here
Article Type: RESEARCH ARTICLE
Citation: Hernández JR (2018) Preliminary Study at the Pilot Plant Scale of the Landfill Leachate Inertization Process Based on Dispersed Phase. Int J Water Wastewater Treat 4(2): dx.doi.org/10.16966/2381-5299.157
Copyright: © Hernández JR. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access