Figure 1: Aerial view of the field location - Coleman landfill site, Saint Kilda, South Australia


Raghupathi Matheyarasu1* Balaji Seshadri2 Pankaj Kumar3 Sonia Shilpi2 Nanthi S Bolan2 Ravi Naidu4
1 Future Industries Institute, Australia*Corresponding author:Raghupathi Matheyarasu, Future Industries Institute (FII) Building ‘X’, Room -20-B04 University of South Australia Mawson Lakes, Adelaide - 5095, Australia, Tel: +61 0424892992; E-mail: matry020@mymail.unisa.edu.au
This study investigated the rate, intensity and types of wastewater irrigation on dry matter yield of selected crops under field conditions (St Kilda, South Australia). A completely randomised block design was employed for the plant growth experiment, where seven field crops (sunflower (Helianthus annuus), sugar beet (Beta vulgaris), canola (Brassica napus L.), alfalfa (Medicago sativa), maize (Zea mays), napier grass (Pennisetum purpureum) and giant reed (Arundo donax) were employed to evaluate the influence of wastewater application on dry matter production. Two types of wastewater (abattoir wastewater (AWW) and municipal wastewater (MWW) and tap water (TW) as a control treatment) and two different loading rates were applied throughout the planting period based on the irrigation requirements (400 mm yr-1 ha-1 and 800 mmyr-1 ha-1). Overall, the plots irrigated with 800 mm wastewater (AWW) showed significantly higher yield than the plots irrigated with 400 mm wastewater (MWW) and TW in all the plots of seven plant species used. The overall trend in biomass yield for three water types and loading rates followed AWW 800 mm>AWW 400 mm>MWW 800 mm > MWW 400 mm > and TW 800 mm treatment, for all the crops. However, the percentage DM yield varied significantly among the crops.
Wastewater; Irrigation; Poor fertile; Dry matter; Relative agronomic effectiveness; Relative yield increases
Global arable land per person has reduced from 0.42% in 1950 to 0.2% in 2000 and is expected to become 0.1% in 2020 [1]. This illustrates the need to produce more grain per unit area to accomplish future global food requirements. With the demand for food crops increasing, fertilisation of agricultural soil is inevitable. As a result, chemical fertiliser consumption has also peaked in the last few decades [2]. Globally, fertiliser consumption for agriculture continues to increase; in 2012 the global fertiliser requirement was 178 million tonnes (N, P and K), which is forecasted to reach a peak of approximately 194 million tonnes in 2017 [3]. The Australian agriculture sector requires about 1 million tonnes of nitrogenous fertiliser and half a million tonnes of phosphorus fertiliser annually for crop production [4]. It is predicted that by 2035, there will be an immense need for P fertilisers globally due to the continuous exploitation of P resources from the existing reserves [5]. Apart from food crops, the rising human population is also in need of energy [6], which is already placing pressure on the environment by emitting greenhouse gases and generating lots of solid (e.g. flyash) and liquid wastes (effluents) [7-9].
Therefore, resource recycling will enable us to meet water, fertiliser and energy needs, thereby enabling land reclamation and income generation by producing valuable bio-energy from wastewater treated crops [10,11].
Growing plants on contaminated land with wastewater will not only use the wastewater and treat the contaminated land but also provide the valuable biomass, which can be used in the paper production, fodder and bio-energy production [12,13]. Contaminated sites (landfill and abandoned mine sites) could be ideal for wastewater enhanced plant growth due to the supply of nutrients and alkaline pH of some farm wastewaters including abattoir wastewater [14,15]. Moreover, the fertility of such sites is typically poor mostly due to pH and the lower availability of nutrients [16]. Although some landfill sites are alkaline [17] due to the waste dumping [18], most of the abandoned mine sites are acidic result of acid mine drainage [19]. Therefore, irrigating those acidic sites with effluents like abattoir wastewater (AWW) will neutralise the soil and supply the required nutrients for plant growth and hence help to increase biomass production.
Biomass-derived energy can help minimise the effects of human activities such as nutrient leaching (e.g. phosphorus), emission of toxic gasses (e.g. nitrous oxide) and high concentration of heavy metals. In our earlier studies, we found that the continuous irrigation of AWW can increase the soil fertility and also the plant biomass yield under greenhouse conditions [20]. However, it is important to examine both positive and negative impacts of AWW irrigation under field conditions. Mittal [21] reported that land application of AWW can cause severe environmental degradation. This is due to flooding field with nutrient rich wastewater, but it is minimal in the presence of vegetation. Hence, growing biomass under wastewater irrigation can be the best option to ameliorate contaminated soil. There are crops that can uptake more amount of nutrients from soil such as Arundo donax and Pennisetum purpureum which are grass species grown across the Australia and worldwide [22,23]. Moreover, these crops can be effective in increasing biomass production under wastewater irrigation [20]. The objectives of this study were as follows: (a) the effect of different types of wastewater (abattoir wastewater -AWW; municipal wastewater -MWW and Tap water -TW as control), irrigation intensity on the increase in biomass of selected plant species; (b) to study the effect of WW irrigation and nutrient cycling in a soil with poor fertility, their nutrient uptake (N and P) efficiency; (c) to determine the most effective plant species (among selected high biomass producing plants), irrigation intensity, irrigation type and productivity under local climatic conditions.
The experimental (Coleman land treatment site =17 hectares) site is located at St Kilda, 23 km NNE of the capital of South Australia. This municipal (Salisbury city) landfill site was operated and maintained for disposal of general waste, trees and other plant stubbles from the avenue plantation and parks since 1950. The soil type at the study site was Anthroposol /Technosol (Australian soil classification system) [24] (Figure 1).

Figure 1: Aerial view of the field location - Coleman landfill site, Saint Kilda, South Australia
The field trial area was marked using the measuring tape and weeds were removed manually before ploughing. A week after weeding, the land was ploughed using the mechanical power tiller and the final dimensions of the cleared area were approximately 30 m length by 17 m width. The field design was prepared before clearing the field plots (Plate 1). The field was marked after ploughing and levelling. The selected site area was further subdivided into small plots. Each plot was 1 m2 and the spacing between the sub plots was 1 m for intercultural operation. Each plot was planted according to the treatment plan, starting from the north direction, AWW (400 mm yr-1 ha-1; 800 mm yr-1 ha-1); MW (400 mm yr-1 ha-1; 800 mm yr-1 ha-1) and TW (800 mm yr-1 ha-1). With three replication of all the seven species starting from sunflower (Helianthus annuus), sugar beet (Beta vulgaris), canola (Brassica napus L.), alfalfa (Medicago sativa), maize (Zea mays), napier grass (Pennisetum purpureum) and giant reed (Arundo donax), were planted accordingly. Seeds were sown at the demarked area of each treatment plot after field preparation. The seeds started germinating a week after sowing. In total, 105 experimental sub plots were employed in this experiment (7 crop species* 5 loading rate* 3 replicated=105 plots). Each subplot was monitored carefully to study the rate of wastewater irrigation on plant productivity. The plots were irrigated on daily basis (based on the water requirements of the crops and irrigated based on the treatment plan) and gap filling was done in 1st and 2nd week of the planting cycle to maintain the equal population. Similarly, weeding and plant thinning were carried out to maintain the substantial population in each plot in the 3rd and 5th weeks, respectively. All the plots were harvested at the same time; three months after planting and fresh biomass of the harvested plants were measured and dried in a hot air oven (at 70°C), to measure the dry matter yield. The plant samples were taken to the laboratory to measure the plant tissue concentration.

Plate 1: Field trial descriptions - This study conducted at Coleman landfill site
The stored soil samples from the different location were analysed for micro and macro nutrients. Soil characterisation was performed based on the Australasia [25] manual. Soil pH and EC was measured using 1:5 soilwater solution. LECO 2000 CNS analyser [26] was used for estimating soil total C and total N. Olsen P was measured by molybdenum blue method [27]. Acid digested soil samples [28] were used for total P and micronutrients determination using Inductively Coupled Plasma - Optical Emission Spectrometry (ICP-OES).
The effect of wastewater irrigation on plant productivity under the field condition was also measured using Relative agronomic effectiveness (RAE). The agronomic effectiveness of the different nutrient loading on the dry matter yield [29,30] was determined from the nutrient concentration of plant tissues of seven crops.
The RAE was calculated using Equations (1) and (2)
\[{\rm{RAE(\% ) = }}\frac{{{\rm{(yield forAWW)}}}}{{{\rm{(yield for TW)}}}} \times 100\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(1)\]
\[{\rm{RAE(\% ) = }}\frac{{{\rm{(yield forMWW)}}}}{{{\rm{(yield for TW)}}}} \times 100\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(2)\]
In this study; the effect of wastewater irrigation on plant productivity under the field condition was also measured using Relative yield increase (RYI).
The RYI was calculated using Equations (3) and (4).
\[RYI{\rm{(\% ) = }}\frac{{\left( {AWW - TWW} \right)}}{{\left( {TW} \right)}} \times 100\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(3)\]
\[RYI{\rm{(\% ) = }}\frac{{\left( {MWW - TW} \right)}}{{\left( {TW} \right)}} \times 100\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(4)\]
Statistical software (SPSS) was used for the analysis of data. The relationships between wastewaters types, nutrient loadings, and plant yield were using Pearson correlation coefficients, 2 sample t-tests and the significance level was P<0.001.
The study area was well drained, levelled and made suitable for growing plants. The soil pH of the study region measured between 8.1 and 8.3. Similarly, electrical conductivity was low (313 µS cm-²) which is most suitable for growing bio-energy crops [31]. The total nutrient concentartion of the soil was measured before planting. The total N and P concentrations were 960 and 46.4 mg/kg, respectively and the total C of the soil was approximately 10,000 mg/kg.
Overall, the plots irrigated with AWW wastewater (800 mm) showed significantly higher yield than the plots irrigated with 400mm and TW in all the plots of seven plant species used. The effect of the loading rates (800mm and 400mm) showed significant changes in terms of dry matter yield (DM) yield of sunflower under field conditions (Figure 2). This may be influenced by the differences in nutrient supply and irrigation rates between these treatments [32]. There was a significant difference between loading rates, types of wastewater irrigation in the overall DM production. All of the crops followed similar trends on DM yield as per water types and irrigation rates. The overall trend in biomass yield for three water types and loading rates followed AWW 800 mm>AWW 400 mm> MWW 800 mm>MWW 400 mm> and TW 800mm treatment, for all the crops. However, the percentage DM yield varied significantly among the crops (Figure 3). This depends on readily available plants nutrients (available -N, available-P) supplied from the application of wastewater [33] and also the uptake of nutrients by the plant species [34].
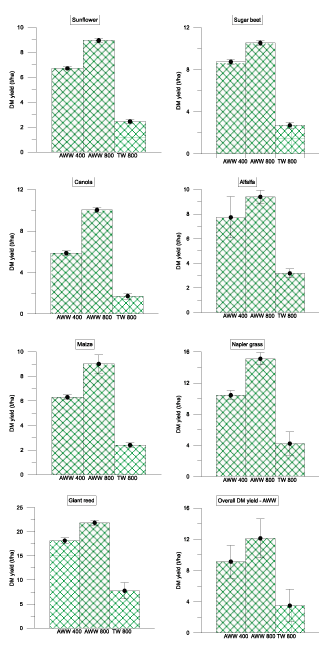
Figure 2: Effects of various treatment methods (AWW 400, AWW 800 mm/yr and TW 800 mm/yr) on dry matter yield of seven different crops (sunflower, sugar beet, canola, alfalfa, maize, napier grass and giant reed) in Coleman land treatment site at St. Kilda. Error bars represents the standard deviation of replicates
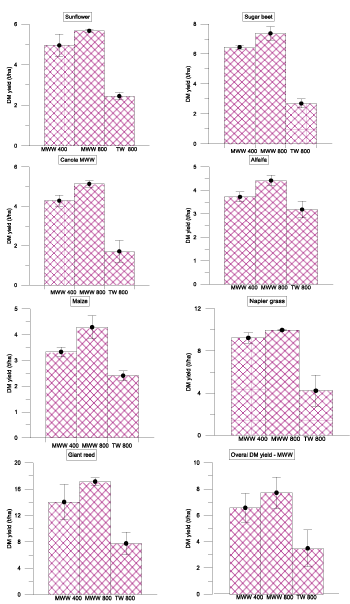
Figure 3: Effects of various treatment methods (MWW 400, MWW 800 mm/yr and TW 800 mm/yr) on dry matter yield of 7 different crops (sunflower, sugar beet, canola, alfalfa, maize, and napier grass and giant reed) in Coleman land treatment site at St. Kilda. Error bars represents the standard deviation of replicates
The percentage of yield (RYI– relative yield increases) increase was calculated for the all the treatments. In comparison with TW irrigation (800 mm), AWW (800 mm) showed a 270% higher DM yield. There was a considerable increase in biomass yield for the two loading rates of MWW 400 mm (26.7%) and 800 mm (39.5%) compared to TW treatment in sunflower plots. The overall biomass data for the sunflower suggests that the trend in yield is as follows: AWW 800 mm>AWW 400 mm>MWW 800 mm>MWW 400 mm>TW 800 mm treatment (Figure 4).
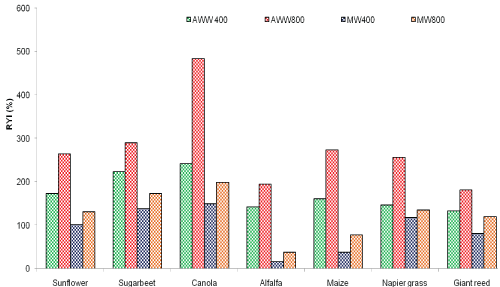
Figure 4: Relative yields of seven different crops grown under five treatments (AWW 800, AWW 400, MWW 800 and MWW 400) compared to tap water (TW) irrigation. (RYI = [(AWW or MWW – TW)/TW]*100)
The percentage increase in DM yield for sugar beet showed a 74.3% higher biomass yield in AWW (800 mm) and MWW showed a 41% more biomass compared to TW. In canola, compared to TW irrigation (800 mm), AWW (800 mm) showed about 82% higher DM yield, which was highest among the seven crops used. In the case of MWW, the biomass yield was 51% higher than TW. The AWW (800 mm) irrigation was 66% higher in terms of biomass production compared to TW and MWW at the same rate showed just 59% increase in yields against TW in alfalfa. Maize showed higher yields of 73.2% for AWW and 59 % for MWW, respectively compared to the biomass yields on TW irrigation rate at 800mm. Napier grass, known worldwide as a high biomass producing plant, responded well to the various water types and loading rates. At 800 mm/year rate, the biomass yield increased 71 % and MWW increased 35 % higher biomass yield compared to TW. Giant reed showed an increase of 228% in terms of biomass yield among all crops for AWW (800 mm/year) treatment compared to TW irrigation. The MWW treatment at the same loading rate showed a 142% increased biomass yield compared to TW irrigation (Figure 4).
Among the seven species tested in this field trial, the overall nutrient uptake was measured as the amount of plant tissue nutrient contents. Figure 5 provide a comparison of plant tissue nutrient contents between the wastewater types and TW. The maximum nutrient uptake was found in the AWW treated plots and the nutrient uptake order as follows AWW>MWW>TW. The maximum nutrient concentration (%) was recorded in the following treatments listed in the increasing concentration of TN: AWW irrigation 3.1%; MWW 2.5% and TW 2.4%. Similarly for phosphorus, AWW irrigation 0.5%; MWW 0.4% and TW 0.3%; AWW irrigation 2.4%; MWW 2.1% and TW 1.9% for K concentration of the crop species used in this study (Figure 5).

Figure 5: Effects of various treatment methods (MWW 400, MWW 800 mm/yr and TW 800 mm/yr) on plant tissue concentration of 7 different crops (sunflower, sugar beet, canola, alfalfa, maize, and napier grass and giant reed) in Coleman land treatment site at St. Kilda. Error bars represents the standard deviation of replicates
A Pearson correlation analyses were conducted for all the plant species irrigated with two different loadings of three different types of water (AWW; MWW and TW); to study the differences in the soil and plant parameters with varying total and plant available nutrients. Overall, the three water types were positively correlated with major nutrients in the soil. For example, Olsen P, TP, DM yields, nitrate-N and ammonia-N was positively correlated with soil TN concentration. The individual treatments and their correlation between the major and micronutrients are discussed below.
The plant productivity of the soil with low fertility was highly determined by the soils parameters such as TN, TP and plant available forms of nutrients such as ammonia-N, Olsen- P and nitrate-N. In terms of AWW (combined effects of both 400 and 800 mm) treatments: the plant productivity of AWW irrigated soil was significantly positively correlated with the plant available forms of nutrients such as Olsen P, nitrate-N and ammonia N. (Table 1). Dry matter yield data obtained from the field trial (all seven crops) increased with increasing wastewater irrigation. The plots irrigated with AWW 400 and 800 mm showed significantly (p<0.001) higher yield than the MWW (400 and 800 mm) and TW (800 mm) irrigated plots (Table 2). This was contributed to the supply of high rate of irrigation and nutrients; wastewater supplies readily available plant essential nutrients for the better growth and development of the plants [35]. The nutrient loading and rate of the application always impact the soil properties significantly thereby improving the fertility of soils [36]. In the current study, AWW irrigation significantly increased the plant productivity compared to the TW and MWW by increasing the nitrate–N, Olsen P, TN, ammonia–N and TP content of the soil (p<0.001) (Table 3).
| Properties | DM yield (t/ha) | Tissue N (%) | Tissue P (%) | Tissue K (%) | Total N (mg/kg) | Nitrate N (mg/kg) | Ammonia N (mg/kg) | Total P (mg/kg) | Olsen P (mg/kg) |
| DM yield (t/ha) | 1 | ||||||||
| Tissue N (%) | 0.236 | 1 | |||||||
| Tissue P (%) | 0.387* | -0.262 | 1 | ||||||
| Tissue K (%) | 0.475** | -0.131 | 0.831** | 1 | |||||
| Total N (mg/kg) | 0.621** | 0.073 | -0.009 | 0.229 | 1 | ||||
| Nitrate N (mg/kg) | 0.452** | -0.19 | 0.448** | 0.371* | 0.612** | 1 | |||
| Ammonia N (mg/kg) | 0.765** | 0.351* | 0.285 | 0.321* | 0.566** | 0.538** | 1 | ||
| Total P (mg/kg) | 0.525** | 0.242 | 0.334* | 0.261 | 0.424** | 0.312* | 0.301 | 1 | |
| Olsen P (mg/kg) | 0.355* | 0.138 | 0.199 | 0.127 | 0.426** | 0.296 | 0.106 | 0.823** | 1 |
Table 1: Pearson correlation coefficients of wastewater irrigated soil properties and plant growth parameters (n=105)
* Correlation is significant at the 0.05 level
** Correlation is significant at the 0.01 level
Table 2: Effects of wastewater irrigation on soil properties and plant growth and development (mean ± s.d)
n.s. not significant
| Properties | TW | AWW | Sig.diff. | MWW | Sig.diff. |
| DM yield (t/ha) | 3.5 ± 2.1 | 10.6 ± 4.6 | p<0.001 | 7.1 ± 0.1 | p<0.001 |
| Tissue N (%) | 2.4 ± 0.7 | 3.1 ± 0.7 | p<0.001 | 2.5 ± 0.5 | n.s |
| Tissue P (%) | 0.3 ± 0.2 | 0.5 ± 0.3 | n.s | 0.4 ± 0.1 | n.s |
| Tissue K (%) | 1.9 ± 1.3 | 2.4 ± 1.2 | n.s | 2.1 ± 0.2 | n.s |
| Total N (mg/kg) | 537.5 ± 18.1 | 1747.6 ± 15.4 | p<0.001 | 1274.9 ± 17.6 | p<0.001 |
| Nitrate N(mg/kg) | 13.6 ± 5.3 | 51.7 ± 1.4 | p<0.001 | 19.6 ± 0.7 | p<0.001 |
| Ammonia N (mg/kg) | 1.1 ± 0.1 | 14.8 ± 1.2 | p<0.001 | 3.6 ± 1.5 | p<0.001 |
| Total P(mg/kg) | 163.4 ± 5.7 | 516.5 ± 17.3 | p<0.001 | 321.5 ± 4.7 | p<0.001 |
| Olsen P(mg/kg) | 47.5 ± 1.9 | 155.8 ± 4.6 | p<0.001 | 101.3 ± 1.8 | p<0.001 |
Table 3: Effects of wastewater irrigation on soil properties and plant growth and development (mean ± s.d. n=105; comparison of overall effects of three types of irrigation loading – in a land treatment site)
The overall effects of wastewater irrigation and TW on the productivity and fertility of low fertile soils were determined using Principal Component Analysis (PCA) for the individual component. The effects of irrigation loading rate (AWW400, AWW 800, MWW 400, MWW 800 and TW 800) on low fertile soil and plant properties were clearly exposed using PCA. The wastewater loading rates (n=105) of both wastewater (AWW and MWW) show distinct separation from the TW treatments. The abattoir wastewater irrigated plots (both level) greatly varied from the TW irrigation (both irrigation levels) reflecting the quantity of wastewater and nutrient loads. The effects of wastewater irrigation on the soil properties showed clearly in the factor loading float. Factors 1 (DM yield) and 2 (N uptake) explained 40% and 21% of the variation, respectively (Figure 6).

Figure 6: PCA analysis: Effects of various treatment methods (MWW 400, MWW 800 mm/yr and TW 800 mm/yr) on plant tissue concentration of 7 different crops (sunflower, sugar beet, canola, alfalfa, maize, and napier grass and giant reed) in Coleman land treatment site at St. Kilda
The AWW application has influenced the RAE in all the other treatments for both levels of treatments in all the seven plant species used. The results showed that AWW irrigation to low fertile soils in the study area can increase the soil productiveness in terms of an agronomic point of view (Figure 7). The RAE increased for all the treatments (MWW application) for all the seven crops grown. The agronomic effectiveness of the treatments was in the following manner: AWW 800>AWW 400 >MWW 800>MWW 400>TW 800. In the case of irrigation intensity, the RAE was highest for the wastewater treatment for both AWW and MWW. In TW (assumed as a control) less than 100% RAE suggests poor agronomic efficiency compared to the AWW and RAE higher than 100% in AWW and MWW treatment indicates the agronomic potential of the amendment is significant.
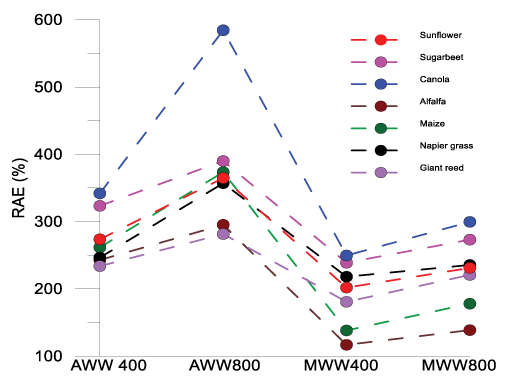
Figure 7: Relative agronomic effectiveness of seven different crops (sunflower, sugar beet, canola, alfalfa, maize, and napier grass and giant reed) grown under four wastewater treatments (AWW 800, AWW 400, MWW 800 and MWW 400) compared to tap water (TW) irrigation in Coleman land treatment site at St. Kilda
The influence of abattoir wastewater irrigation on soil fertility and plant dry matter yield of selected plant species under field conditions were as follows: Effluent irrigation was shown to be a major alternative nutrient source for soil with low fertility as soil fertility can be increased by wastewater application. Abattoir wastewater irrigation can supply 100 % of nutrients required for production of bio-energy crops in a cropping cycle. Improved management can reduce the incidence of nutrient loss from wastewater irrigated soils by calculating input and output ratio to avoid nutrient loss and seepage. Growing plants at the land treatment site with nutrient rich wastewater can be a sustainable and economic method for disposal and management of wastes. Biomass produced from wastewater irrigated source can further be utilised for paper production, bio-energy generation and as animal fodder.
I would like to thank (first author) the University of South Australia for the UPS scholarships and CRC for Contamination Assessment and Remediation of the Environment for the CRC-CARE scholarships to undertake research on Nutrient management in abattoir wastewater discharged soil.
Download Provisional PDF Here
Article Type: Research Article
Citation: Matheyarasu R, Seshadri B, Kumar P, Shilpi S, Bolan NS, et al. (2017) The Effect of Wastewater Irrigation Rate on Dry Matter Yield of Selected Field Crops. Int J Water Wastewater Treat 3(3): doi http://dx.doi.org/10.16966/2381-5299.142
Copyright: © 2017 Matheyarasu R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access