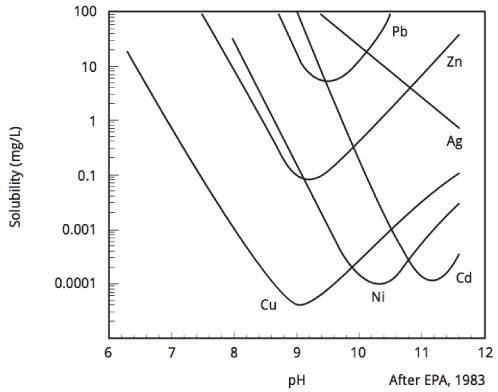
Figure 1: Solubility of hydroxide metals


Habib Karimi*
MSc of Environmental health engineering, Shahrekord, Iran*Corresponding author:Habib Karimi, MSc of Environmental health engineering, Shahrekord, Iran, Tel: (+98) 9136278823; E-mail: ehe15400@gmail.com
The effect of pH and initial concentration of lead ions on the removal of Pb(II) using Ca(OH)2 as precipitant agent were investigated. In order to analyze the precipitation process, seven initial Pb(II) concentration (100, 200, 300, 400, 500 and 600 mg/L) and five pH levels (3,5,7,9 and 11) were studied. The precipitation experiments were carried out using jar test equipment. This study was a batch method that involves the mixing of Ca(OH)2 with Pb(II) containing solutions to occur nucleation and solid growth and subsequent separation of solids (precipitates) from solution by filtration. The experiments were conducted for both actual and synthetic wastewater. The result showed that the removal efficiency was increased by increasing of pH and initial concentration of lead. The high pH levels showed better performance than the low levels in the removal of lead ions from both actual and synthetic wastewater. The highest removal efficiencies for actual and synthetic wastewater were 88% and 95% at pH 11, respectively. The result of this study showed that the Precipitation process using Ca(OH)2 is a suitable and efficient technique for the removal of lead ions from industrial wastewater in the pH range of 9-11.
Lead; Industrial wastewater: Heavy metals: Precipitation
Industrial wastewaters are effluents that result from human activities which are associated with raw-material processing and manufacturing. Water pollution occurs when potential pollutants in these streams reach certain amounts causing undesired alterations to a receiving water body [1]. Some industrial processes generate wastewater containing heavy metal contaminants. The common Heavy metals include Lead, Cadmium, Chromium, Nickel, Zinc, and Arsenic. Heavy metals can cause environmental and health effects. Also because of the high solubility of heavy metals in the aquatic environments, these pollutants can be absorbed by living organisms. If the metals are ingested beyond the permitted concentration, they can cause serious health disorders. Since most of the heavy metals are non-degradable into non-toxic end products, their concentrations must be reduced to acceptable levels before discharging them into the environment [2,3].
Lead is one of the heavy metals that present in the wastewater of many industries including pigments, dying, leaded glass, painting, and manufacture of storage batteries. The concentration of lead in these industries is higher than permissible discharge level. For example, the wastewater derived from storage battery manufacturing may contain lead concentrations in the range of 40.8 to 319.4 mg/L [4,5]. Lead is included in the list of probable carcinogen of the Environmental Protection Agency (EPA). The main target for lead toxicity is the nervous system, both in adult and children. At a high level of exposure, lead can severely damage the brain and kidneys. Lead exposure can cause weakness in fingers, small increases in blood pressure, anemia and miscarriage in pregnant woman [6]. The World Health Organization (WHO) recommends 0.05 mg/L as the maximum acceptable concentration in drinking water [7].
So, it is necessary to wastewater containing lead refined before discharge to the environment. Numerous physical and chemical processes including Adsorption [8], Electro dialysis [9], flocculation [10], ion flotation [11], and ion exchange [12] have been applied for removal of lead from industrial wastewater.
Precipitation process is one of the common treatment methods that used for removal of heavy metals and other pollutants. All ions with positive and negative charge can be removed from wastewater by a precipitation process.
The most common precipitation processes are three types including hydroxide precipitation, carbonate precipitation, and sulfide precipitation. In the hydroxide precipitation, the hydroxide sources including CaO, Ca(OH)2 , Mg(OH)2 , and NH4 OH can be used for removal of heavy metal cations from wastewater [13]. Metal precipitation as hydroxide, which is described in Equation 1:
\[{M^{2 + }} + O{H^ - } \to {\rm{ }}M{\left( {OH} \right)_2}\]
Hydroxide precipitation has the advantage of removing many of the pollutant parameters existing in metal finishing wastewaters without pretreatment. The process operates at ambient conditions and its operation is easy and suited to automatic control. The most important advantage of the process is its low cost [14].
In order to remove heavy metals by the precipitation method, it is necessary to achieve optimum conditions. The aim and emphasis of this study were to identify the effect of pH and initial concentration of Pb (II) on the lead removal efficiency and determination of an optimum condition for Pb (II) removal using Ca(OH)2.
In general, the precipitation process has two stages-the first nucleation and the second particle growth. Nucleation involves the adherence of Small particles to each other and particle growth consists of an enlargement of the nucleus through the addition of more atoms into the particle structure. In the nucleation process, the rapid solid formation occurs. In the second step, the agglomeration of small particles in the suspension was occurred to form larger aggregates. In the nucleation process, the tiny solid masses are formed and whereby theses masses or nucleus starts a crystallization process. In the hydroxide precipitation, the hydroxide ions react with the lead ions and produce the lead hydroxide precipitates as Equation 2:
\[P{b^{2 + }} + O{H^ - } \to {\rm{ }}Pb{\left( {OH} \right)_{^2}}\]
In the precipitation process, pH and initial concentration of metal directly influence removal efficiency. The solubility of metal hydroxides is presented in Figure 1. As is clear in Figure 1, the solubility of most metals in the pH range of 9 to 11 is minimal. Nevertheless, an accurate selection of the operating conditions (such as pH) is necessary. Because the wastewaters have a variable nature from one industry to another and complexes of pollutants contains in them. So, the focus of this study was to determine the optimal condition for the removal of lead from battery industries wastewater.

Figure 1: Solubility of hydroxide metals
All materials that used in this study were analytical grade and purchased from Merck. A stock solution of Pb(II) was prepared by weighing out a 1.615 grams Lead(II) nitrate (Pb(NO3)2) and diluting to 1000 ml volume. Pb(NO3)2 is a colorless powder and soluble in water. The density and solubility of Lead (II) nitrate in water is 4.53 g/cm3 and 52 g/100 mL at 20°C, respectively. The solutions with a required concentration of Pb(II) were prepared by diluting a stock solution.
The real wastewater samples were collected from SEPAHAN battery Co. that located in Isfahan, Iran. Prior sampling, the sample bottles washed with distilled water to remove the possible contaminants. To prevent the hydrolysis of samples, a few drops of nitric acid was dropped on them.
The precipitation experiments were performed using jar test equipment comprising five 2000 ml jars. The experiments were carried out using synthetic wastewater as well as actual battery industries wastewater. The concentration of lead in the actual wastewater was 8.8 mg/L. The precipitation experiments carried out in a 1 on 1 mole ratio [i.e., one mole of OH- added for each mole of Pb(II)].
The effect of initial concentration on the removal efficiency was assessed by determining the removal efficiency at 100, 200, 300, 400, 500 and 600 mg/L. Also, In order to evaluate the effect of pH on the lead removal efficiency, precipitation experiments were conducted with different pH conditions in a range from 3 to 11. The pH adjustment carried out using 1M NaOH and HCl. For all experiments, the mixing process was conducted in two steps. In the first stage, the mixing was performed for 2 minutes at 100 rpm. In order to complete the crystallization process, in the second stage mixing speed was reduced to 30 rpm and mixing time increased to 10 min. For sedimentation of precipitates, the samples were maintained at rest for 15 minutes. Then 50 ml of the supernatant was collected from each jar and passed through the filter. The Whatman NO.45 filter was used to remove precipitates. In all experiments, one liter of samples was used.
The amount of initial and final concentration of lead ions was quantified using an atomic absorption spectrophotometer. The drawing of the calibration curve was performed using a 2.5, 5 and 10 mg/L standard solutions of Pb(II). The following equation was used for calculation of removal efficiency:
\[R{\rm{ }} = \frac{{{C_0}{\rm{ }} - {C_1}}}{{{C_0}}} \times 100\]
Where R is the removal efficiency of a percent (%). C0 is the initial concentration of Pb(II) and C1 is the concentration after precipitation experiments and the passing of the supernatant from the filter (as mg/L).
The pH is the main factor in the removal of heavy metals by various methods such as chemical precipitation, adsorption, and biological method. In this study, it was found that by increasing the pH, the lead removal efficiency increases. The Pb(II) removal efficiency as a function of pH are presented in Figure 2 and clearly indicate that the low pH levels were not effective for removal of lead ions from wastewater.
The graphs of Figure 2 related to synthetic wastewater. Whereas the optimum condition for removal of lead ions form synthetic wastewater was obtained in pH 9 to 11, therefore the experiments for actual wastewater were conducted for mentioned pHs. The result showed that the removal efficiencies for actual wastewater are very high and was about 95%.
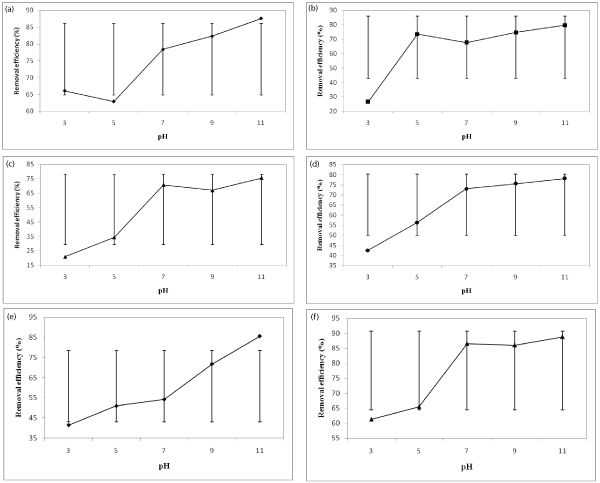
Figure 2: Effect of pH and initial concentration on the removal efficiency (a) 100 mg/L, (b) 200 mg/L, (c) 300 mg/L, (d) 400 mg/L, (e) 500 mg/L, and (f) 600 mg/L
The obtained results analyzed by T-test in SPSS software. The result of analyses showed that the removal efficiencies in pH 3 have a significant difference with removal efficiencies in pHs of 7, 9 and 11(p-value<0.05). However, the differences between removal efficiencies in pH 3 and pH 5 were not significant (p-value>0.05).
In the case of concentrations, were not observed the significant difference in 100 and 200 mg/L removal efficiencies. However, the removal efficiencies for 100 mg/L Pb(II) have a significant difference with the removal efficiencies in the concentrations of 400 and 500 mg/L.
As can be seen in the graph (a) the removal efficiency in pH 5 lower than removal efficiency in pH 3. It seems that the small reduction of removal efficiency (about 3%) in pH 5 don’t relate to the formation of smaller crystals in pH 5 and lower removal of lead ions in this pH. Since this reduction is very low, therefore this phenomenon probably related to measurement errors and quality of paper filters that used for removal of precipitants from the supernatant. There are also similar situations in (c) and (f) graphs. The reduction of removal efficiencies in these cases related to same probabilities. What certainly can be said is that the solubility of lead hydroxide in the pH range of 9 to 11 is minimal and the highest removal efficiency achieved in these pHs.
The error bars in graphs indicated the averages and standard deviations of removal efficiencies for each concentration. In general, the Pb(II) removal efficiency increased as the pH of the solution increased. For example, in the initial concentration of 300 mg/L and at pH of 3, the Pb(II) removal about 21% and this amount received to 75.5% at pH of 11. For a concentration of 200 mg/L, The Pb(II) removal efficiencies were changed from 26.5% to 79.7% at pH 3 to 11.
As can be seen from Figure 2, the removal efficiency was decreased about 3 times by reducing the pH from 11 to 3 at 200 mg/L initial concentration. A similar situation can be observed for 400 mg/L initial concentration. The removal efficiency increased from 56.3 to 75.5 % for 5 to 9 pH values, respectively. As can be seen in Figure 1, the lead removal efficiencies at 9-11 values are very close together. The higher removal efficiencies in high pH related to high concentrations of OH- ions in solution. These ions react with Pb(II) ions and convert them to insoluble precipitates and removed lead ions from solution.
At a concentration of 500 mg/L, with 8 units increasing in pH, the removal efficiency has increased two times. The maximum removal efficiencies for actual and synthetic wastewater were 95% and 88% at pH 11, respectively.
The previous studies confirmed these results. In one study, the removal of iron, chromium, copper, lead, nickel, and cadmium using chemical precipitation by CaO and MgO was investigated by Mahmood m. Brooti et al. [15]. In this study, the lead removal efficiencies in the pH of 6 and 10 were reported 20% and 55%, respectively.
In another study, the precipitation of nickel, zinc, and cadmium was studied by Peters and Ku [16]. Based on results of this study, in the higher pH values, the rate of nucleation and precipitates size was increased.
The initial concentration of lead has a similar effect of pH on the removal efficiency. From the result of this study, we can see that the removal efficiency of Pb(II) increased with increasing of initial concentration of lead ions in a sample.
As can be seen in Figure 1, the removal efficiency for 600and 300 mg/L Pb(II) concentration in pH of 11 were 75.5% and 88.8%, respectively. It seems that the higher removal efficiency at higher concentrations related to the formation of more and larger precipitates and agglomeration of these solids together.
The maximum Lead removal efficiency for synthetic wastewater was obtained at pH 11 and lead initial concentration 600 mg/L that equivalent to 88.8 percent. However, the removal efficiency for actual wastewater at same pH was significantly higher than synthetic wastewater was equivalent to 95 percent. The concentration of lead in actual wastewater is lower than synthetic wastewater and we expected that the removal efficiency is low. In the actual wastewater, a complex of ions is present. As a rule, these ions can compete with together in precipitation process and therefore can reduce the Pb(II) removal efficiency in actual wastewater. But it should be noted that the impurities and ions in the real wastewater can assist to precipitates coagulation, increasing the particle size and formation of larger agglomerates. Therefore in the actual wastewaters, we can achieve the higher removal efficiencies with a lesser amount of precipitant materials.
In a study that was conducted by Charerntanyarak L [17], the similar results were obtained. In this study, the synthetic wastewater containing Zn, Cd, Mn and Mg were used. The results of this study showed that the optimum pH for chemical coagulation and precipitation by lime treatment was more than 9.5. He reported that when polymer added to solution the removal efficiency increased.
Calcium hydroxide (Ca(OH)2 ) was found to be an effective and relatively low cost precipitant agent for the removal of Pb(II) from synthetic and actual wastewater. It was concluded that the Pb(II) removal efficiency is strongly influenced by initial concentration and pH. The optimum removal efficiency for actual and synthetic wastewater was at pH 9 to 11. The lead removal efficiency increased with increases in initial Pb(II) concentration and pH. Hydroxide precipitation process was found to be successful in removal of Pb(II) ions from wastewater especially in actual wastewater. The maximum lead removal efficiencies for actual and synthetic wastewater using Ca(OH)2 were 95 and 88% at pH 11, respectively. Ultimately this study demonstrates that calcium carbonate as a precipitant agent can effectively be used to the removal of lead ions from industrial wastewater under pH 9 to 11 pH ranges. For obtain higher removal efficiency, the dosage of lime should be increased.
The author would like to express her upmost gratitude to the SEPAHAN battery Co. for providing samples. The author would also like to acknowledge the research support from the Shahrekord University of Medical Sciences.
Download Provisional PDF Here
Article Type: Research Article
Citation: Karimi H (2017) Effect of pH and Initial pb(II) Concentration on The Lead Removal Efficiency from Industrial Wastewater Using Ca(OH)2 . Int J Water Wastewater Treat 3(2): doi http://dx.doi. org/10.16966/2381-5299.139
Copyright: © 2017 Karimi H, This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access