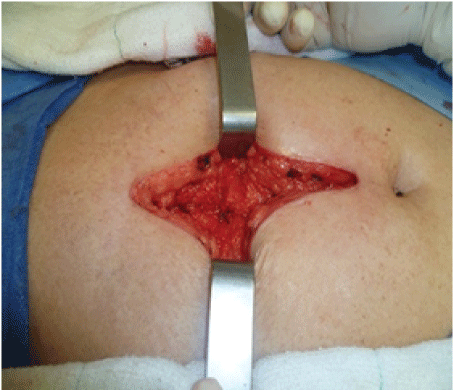
Figure 1: Surgical wound with sutured aponeurosis
Gasman Humberto Ochoa Alvarez1* Diego Fabricio Erazo Mogrovejo1 Benjamín Pérez Erazo2 Marlene Josefa DíazLópez2 David Gabriel RoseroArévalo3 Edison RobertoNúñez Moina3
1General Surgeon, Surgery Service, General Hospital IESS Riobamba, Ecuador/Rio Hospital Clinic, Center for General and Bariatric Surgery Riobamba, Ecuador*Corresponding author: Gasman Humberto Ochoa Alvarez, General Surgeon, Surgery Service, General Hospital IESS Riobamba, Ecuador/Rio Hospital Clinic, Center for General and Bariatric Surgery Riobamba, Ecuador, Tel: 0999003983; E-mail: gasmanoa@hotmail.com
Objective: To identify the effectiveness of the management of contaminated and dirty wounds with aspiration tubular drainage (GASMANVAC DRAINAJE) in subcutaneous cellular tissue in the General Surgery Service IESS Riobamba Hospital.
Location: IESS Riobamba General Hospital.
Design: Prospective, Transversal, Descriptive and Quasi-Experimental Study.
Statistical analysis: Measures of central tendency.
Patients and methods: A total of 675 postsurgical patients were analyzed in the IESS Riobamba Hospital between August 2009 and July 2013, who presented injuries classified as contaminated and dirty in which GASMANVAC DRAINAJE was used. The variables analyzed were age, gender, mass index surgery, surgery, aspiration tubular drainage, surgical wound infection and length of hospital stay.
Results: Of the 675 patients, 4% presented superficial infection of the surgical wound, while 96% did not, considering it a favorable evolution for the majority of patients.
Conclusions: The use of “GASMANVAC DRAINAJE” has demonstrated the reduction of surgical wound infection in the postoperative patient. If we compare the present study with the literature, which indicates that the infection in the case of contaminated wound is 15-30% and in the dirty one>30%, we can conclude that our investigation showed that this type of drainage is effective in the prevention of superficial infection of surgical wound presenting 4% of infection (1.6% contaminated wound and 2.4% dirty wound).
Surgical drainage; Surgical wound infection; Type of surgery
The infection of the surgical wound (IHC) and the complications derived from it have constituted an inseparable fact to the surgical practice since its inception multiple agents have been used in wounds as an attempt to promote healing and prevent death. These agents included resins such as turpentine, tar and fish, balms and ointments, myrrh and incense, honey, alcohol, glycerin, mercury chloride, silver nitrate, iodine, hypochlorites, creosote, ferric and zinc chlorides and carbolic acid [1-3].
In surgery, infections are always found, since the first lines of host defenses, the cutaneous or mucosal barrier, between environmental microorganisms and the host’s internal environment inevitably are disturbed [4]. For the infection to occur, the first step is the penetration of microorganisms into the tissues.
Throughout history and the first surgical procedures, the majority of deaths were the result of infections. It was until the end of the 19th century that the bacterial origin of surgical infections was demonstrated. Mortality from infections was so common after composite fractures or due to firearm shooting that the standard treatment was amputation [3,4].
Although the arrival of antibiotics seemed to end with the infection, time has shown that surgery continues as an effective therapeutic means. There are certain limits and disinformations that still persist: The surgical infection has been presented as nosocomial, forgetting that about 30% start in the community; almost exclusively refers to the infection of the surgical wound, without referring to other septic complications, which can reach greater significance [5-7].
The role of the patient himself as a reservoir of pathogenic germs is not sufficiently recognized, forgetting the nonbacterial, fungal or viral infections that can break out in an immuno depressed patient.
Sometimes excessive confidence is placed in antibiotics, forgetting that they only facilitate and give time to the defensive systems themselves.
The objective of antibiotic prophylaxis in surgery is to reduce the bacterial load when surgical damage begins, which has been shown to reduce the incidence of infection of the operative site, ranging from the skin to any parenchyma or anatomical cavity.
The most important bacteriology of surgical infections in recent decades is the high frequency of Staphylococcus aureus; to the impressive increase between 1985 and 1995 of severe soft tissue infections, especially of necrotizing fasciitis, productions by Streptococcus pyogenes, in which it was very relevant, in addition to the antibiotic therapy, the aggressive surgical action; gram-negative bacilli, especially enterobacteria; the increase of polymicrobial infections and of anaerobic, sporulated and nonsporulated bacilli [8-10].
It is worth mentioning that antimicrobials are adjuvants of the immune system. Thus, in a decompensated malnourished or diabetic patient, the risk of infection is greater regardless of the antibiotic, so the correction of all pre-operative factors is crucial to obtain good results [11,12].
The use of drainages dates back to the time of Hippocrates who described the use of cannulae. In 1895, Kellog described the precursor of aspiration drainage. Three years later Heaton applied constant aspiration to a siphon drain. At the beginning of the century, Yates concluded that the drainage of the peritoneal cavity is impossible from the physiological point of view and that the only function was to “peritonealize additionally” the affected area. To cite a local event, drainage to the “vesicular bed” after a cholecystectomy was used for a long time. With the advent of laparoscopic surgery, this drainage was used very selectively and it is currently a rarity to use drainage after a classic and laparoscopic cholecystectomy [13].
As you can see, the topic has been and continues to be controversial, the alternatives are very varied and the selection will depend largely on the local media and the surgical school [14].
From time immemorial, the infection of the surgical wound and the complications that derive from it have constituted an inseparable fact to the surgical practice [15]. The infection is the product of penetration, growth, metabolic activities and the consequent physical-pathological effects of the microorganisms in the tissues of the patient. Even with the perfection of the asepsis and antisepsis techniques, the infection will accompany the surgery, especially when surgical procedures defined as contaminated and dirty are approached.
These wounds have been handled in a traditional manner with late primary closure or second intention, a situation that constitutes a real physical and psychological pain for those who suffer from it, due to the doubling of their hospital stay associated with the delay in healing of wounds, increased morbidity and mortality and prolonging their early incorporation into the family, work or school environment, constituting a very high cost for society [16,17].
For this reason it is convenient to look for a useful alternative, comfortable for the patient, economical and effective that allows a better handling of the wounds.
The same that will reduce the incidence of surgical wound infection in contaminated and dirty surgeries; allowing the primary closure of the wound by incorporating aspiration tubular drainage in subcutaneous cellular tissue of patients operated on in the General Surgery Service of the IESS Hospital of Riobamba, allowing the quick recovery of the patient and lower hospital cost [18,19].
Patients, Materials and Methods
This is a Prospective, Transversal, Descriptive and QuasiExperimental study, in which all surgical patients were analyzed, an abdominal surgery with defined wounds as contaminated and dirty intervened in the General Surgery Service of the IESS Hospital of Riobamba for 5 years (2009-2013).
The following variables were analyzed: age, gender, body mass index, surgery, aspiration tubular drainage, surgical wound infection, length of hospital stay. We excluded patients with clean, clean contaminated wounds and who were not operated on in this hospital.
In the definition of study variables:
Time since the birth of an individual
Biological sex with which the individual is born.
It is a measure of association between weight and height. It is calculated according to the mathematical expression:
\[BMI = \frac{{Weight{\rm{ }}\left( {kg} \right)}}{{heigh{t^2}({m^2})}}\left( {metric} \right)\]
According to the trans-surgical findings and the type of surgery, injuries can be classified.
Contaminated: Traumatic wounds less than 6 hours, opening of the biliary tract or genito-urinary in the presence of bile or infected urine, important contamination by the content of the digestive tract; important asepsis rupture, interventions in the presence of acute inflammation without pus.
Dirty: Traumatic injuries contaminated or treated with a delay of more than 6 hours; in the presence of devitalized tissues, bacterial inflammation with pus, fecal contamination or foreign bodies, perforated viscera [20].
Infection of the surface of the surgical wound: Established by the surgeon who presents purulent discharge, even without microbiological confirmation, which affects the skin and subcutaneous tissue.
Time elapsed from admission to hospital discharge.
These are closed elements that perform a soft and permanent suction, which allows the evacuation of accumulated liquids or gases in areas of the organism after a surgical procedure; connected to a tank in which a vacuum has been made.
Patients were directly observed before and after their surgical intervention during their hospitalization and follow-up period through their clinical history in the AS-400 system of the IESS Hospital of Riobamba.
The study was approved by the Director of IESS Riobamba Hospital and the Local Health Research Committee. The confidentiality of the information obtained from the AS-400 System of the Hospital Records was respected at all times.
In the statistical analysis for qualitative variables, frequencies were used and for central variables, measures of central tendency were used as means and dispersion measures such as range and standard deviation. With the data obtained, an electronic sheet (EXCEL) was designed for tabulation, analysis and interpretation. The JMP software was used for the statistical analysis.
Results
A total of 675 patients were analyzed, their ages were between (15 and 97) years, as minimum and maximum respectively, a mean age of 49 years, standard deviation (D.E: 18.88). With injuries classified as contaminated (436 patients) and dirty (239 patients) in which “GASMANVAC DRAINAJE” was used; of these, 4% presented superficial infection of the surgical wound (1.6% contaminated wound, 2.4% dirty), while 96% did not, considering it a favorable evolution for the majority of patients [21].
The hospital stay was between 2 to 6 days, with an arithmetic mean of 4 days, standard deviation D.E: 3.06 with an interval of confidence 95% CI (3,73-4,74).
The following describes the steps for the placement of the DRAINAGE GASMANVAC (Figures 1-11).

Figure 1: Surgical wound with sutured aponeurosis
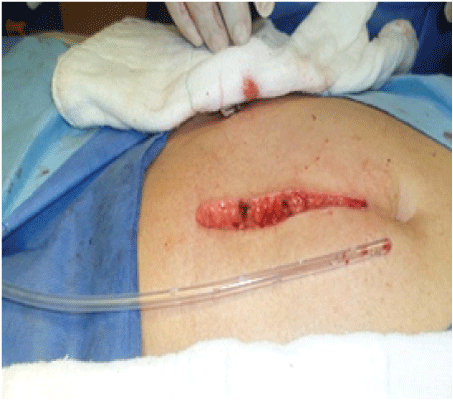
Figure 2: Measurement of the length of the nelaton probe to be used
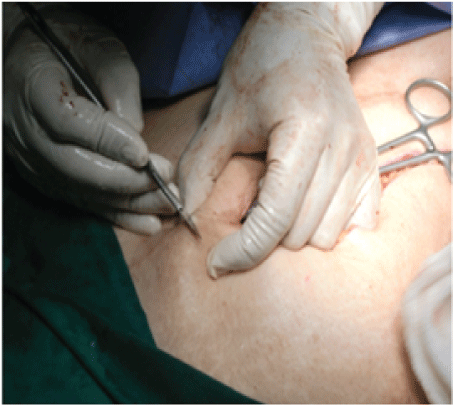
Figure 3: Anti-opening incision for exteriorization of the Nelaton probe
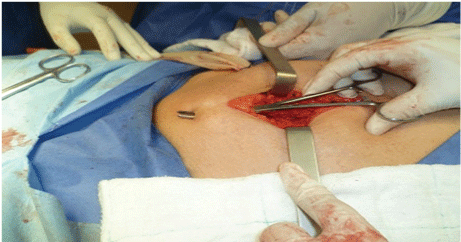
Figure 4: Kelly clamp exteriorization by opening wound
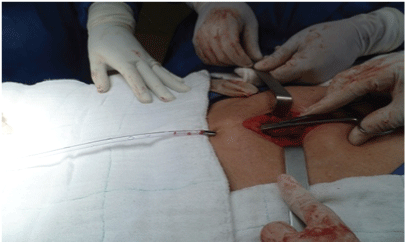
Figure 5: Null probe end entry per counter opening hole
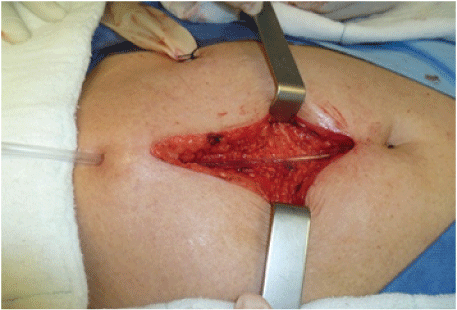
Figure 6: Placement of drainage in the subcutaneous tissue
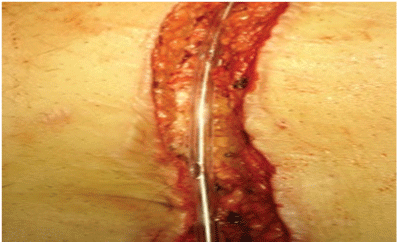
Figure 7: Holes in the entire path of the Nelaton probe
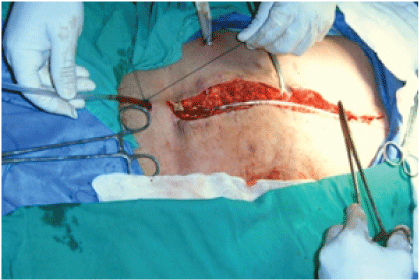
Figure 8: Nelaton probe fixation on skin to prevent displacement
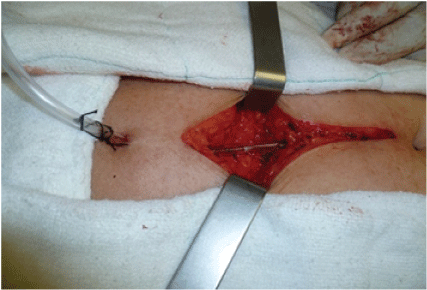
Figure 9: Nelaton probe coating with subcutaneous cellular tissue
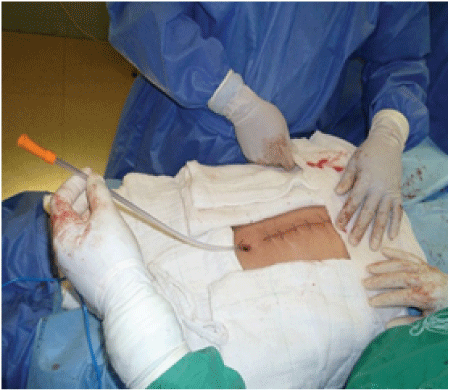
Figure 10: Primary airtight closure of surgical wound
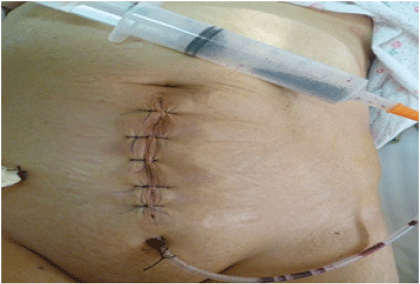
Figure 11: Primary airtight closure of surgical wound
In this study, aspiration tubular drainage was used in all patients as a prophylactic measure to avoid infection of the surgical wound, demonstrating a significant reduction in the percentage of infection in the two study groups for both contaminated and dirty wounds (Table 1).
| Type of Wound | Wound Infection | Total | |
| No N.° % | Yes N.° % | N° % | |
| Contaminated | 424 (97%) | 12 (3%) | 436 (65%) |
| Dirty | 221 (92%) | 18 (8%) | 239 (35%) |
| Total | 645 (96%) | 30 (4%) | 675 (100%) |
Table 1: Relationship between the type of Surgical Wound and the Infection Incidence H.Iess.Riobamba
The patients who presented infection of surgical wound were 30. Among the pathologies that were observed in this group, those of the table stand out, showing with great statistical significance that the percentage of complications increases as the more severe stages appear (Table 2).
| Post-Surgical Diagnosis | Number | Percentage |
| Complicated Appendicitis | 15 | 50 |
| Peritonitis | 6 | 20 |
| Hemoperitoneo | 5 | 17 |
| Colangitis/Hydropiocolecisto | 4 | 13 |
| Total | 30 | 100% |
Table 2: Distribution of Patients with Surgical Wound Infection According to Post-Surgical Diagnosis H.Iess.Riobamba
Of the 675 patients surgically treated with abdominal pathology and in whom aspirative tubular drainage was used in subcutaneous cellular tissue, it was determined that:
The minimum number of days of hospital stay is 2 with a maximum of 18 days
The majority of patients remained hospitalized for 2 to 6 days, the prolonged stay of a certain group of patients was due to intra-abdominal complications and the general condition, the underlying pathology itself identifying that tubular aspiration drainage more than effective does not prolong the hospital stay, contributed to a quick recovery of the patient and decreased associated costs [22,23].
It was determined statistically with Fisher’s test p: <0.0001 value that showed a direct relationship between age and female gender that predispose to surgical wound infection. This group identifies that the majority of women are in a postmenopausal period that is related to endocrine changes typical of this age, such as a decrease in the production of estrogen and progesterone, making them susceptible to surgical wound infection and other morbimortality (Table 3).
| Age | Gender | |
| Female | Male | |
| N° % | N° % | |
| 15-42 | 0 (0%) | 0 (0%) |
| 43-70 | 5 (17%) | 0 (0%) |
| 71-97 | 25 (83%) | 0 (0%) |
| Total | 30 (100%) | 0 (0%) |
Table 3: Distribution of patients with infection of surgical wounding according to gender and Ageh.Iess.Riobamba
Advanced age was identified as a risk factor for the appearance of surgical wound infection, this data was compared with different international studies where it is established that the older there is a higher incidence of surgical wound infection due to a significant deterioration of the defenses. Of the host, detriment in cellular repair and higher prevalence of comorbid conditions (Table 4) [24].
| IMC | Infection De Herida Quirúrgica | Total | |
| Yes N° % | No N° % | ||
| 18,5-24,9 | 0 (0%) | 220 (34%) | 18,5-24,9 |
| 25-29,9 | 0 (0%) | 290 (45%) | 25-29,9 |
| 30-40 | 30 (100%) | 135 (21%) | 30-40 |
| Total | 30(100%) | 645 (100%) | 675 (100%) |
Table 4: Distribution of Patients In Relation To the Body Mass Index and Surgical Wound Infection Hospital.Iess.Riobamba
These results have been compared with several studies that show that obesity predisposes to poor wound healing, failure of surgical techniques and hemorrhages. This is due to resistance to leptin, associated with obesity, which may contribute to the pathophysiology of involvement in wound repair and therefore to the appearance of infection due to lack of local circulation of the wound, longer surgical time and an increase in the area of exposure of the wound.
Download Provisional PDF Here
Article Type: RESEARCH ARTICLE
Citation: Alvarez GHO, Mogrovejo DFE, Erazo BP, DíazLópez MJ, RoseroArévalo DG, et al. (2018) Effectiveness in the Prevention of Superficial Infection of Polluted and Dirty Wounds with “Gasmanvac Drainaje”, in the General Surgery Service of Iess Riobamba Hospital since August 2009-July 2013. J Surg Open Access 4(1): dx.doi.org/10.16966/2470-0991.166
Copyright: © 2018 Alvarez GHO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access