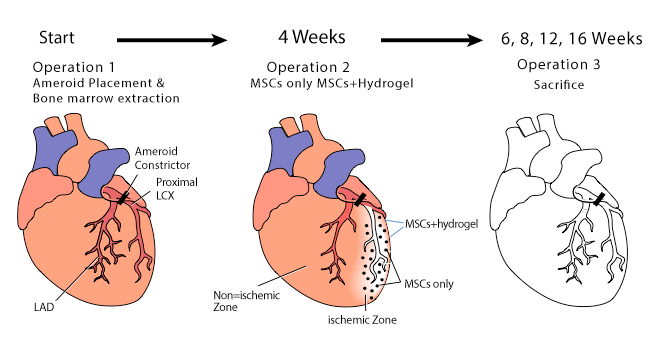
Figure 1: Experimental timeline demonstrating ameroid placement, injection of MSCs only or MSCs plus hydrogel, animal euthanasia and histologic evaluation at the end of the experiment.


Effect of Hydrogel in Cell-Based Therapy in a Porcine Model of Chronic Myocardial Ischemia
Zhou Y1* Wang S1 Yu Z1 Hoyt RF Jr1 Karakas MF2 Hunt T1 Chan A2 Kindzelski BA1 Abraham MR2 Horvath KA1
1Cardiothoracic Surgery Research Program, NHBLI, National Institutes of Health, Bethesda, USA*Corresponding author: Zhou Y, MD, Chief, Cellular Biology Section Cardiothoracic Surgery Research Program, National Heart Lung and Blood Institute, National Institutes of Health, Building 10, Room B1D47, 10 Center Drive, Bethesda, MD 20892, USA, Tel: 301-451-7101; Fax: 301-480- 1649; E-mail: zhouyifu@mail.nih.gov
Hyaluronic acid (HA)-based hydrogels have been reported to enhance stem cell survival following transplantation in rodent models. Here, we test the effects of encapsulating MSCs in HA-serum hydrogels (synthesized by cross-linking HA by autologous serum) in a large animal model. We performed the current study in a porcine model of chronic myocardial ischemia by injecting MSCs with and without hydrogel into chronically ischemic myocardium in order to compare cell survival at several time points following transplantation.
Autologous MSCs were injected with or without hydrogel in separate marked areas within the ischemic region (created by applying ameroid constrictors around the proximal left circumflex artery 4 weeks prior).Animals (n=2) were sacrificed at 2, 4, 6 and 12 weeks following cell transplantation and histopathologic assessment was conducted.
Transplantation of MSCs encapsulated in hydrogels boosted retention of transplanted cells in the injected area and stimulated angiogenesis compared to areas devoid of hydrogel injections in the short term. Of note, MSCs exited the hydrogel and migrated into the surrounding myocardium despite homogeneous dispersion of MSCs in the injected hydrogel. This suggests that once injected, MSCs have more affinity for the host tissues.
Intra-myocardial transplantation of MSCs encapsulated in HA-serum hydrogels enhanced myocardial cell retention at an early stage and promoted angiogenesis in a large animal model. Based on these results, future studies of encapsulated cell delivery in transmurally infarcted myocardium are warranted in order to optimize the use of HA-serum hydrogels in cell-based therapy.
Hydrogel; Mesenchymal stem cell; Chronic myocardial ischemia
Mesenchymal stem cells (MSCs) have been broadly used as a cellbased therapy to improve heart function in experimental animal studies and clinical patients with myocardial ischemia or infarction [1-6]. Yet, the issue of cell retention and survival after transplantation remains a major obstacle for cell-based therapy. Efforts have focused on stem cell gene manipulation [7,8] or utilizing materials, such as hydrogel [9-12], that aim at increasing cell survival and homing following transplantation.
Hyaluronic acid (HA) is one of the chief components of myocardial extracellular matrix. Cells bind HA via cell surface receptors, CD44 and CD168, which have been found to be important in cell adhesion and motility [10-12]. The HA degradation products play a role in angiogenesis [10], which is one of the mechanisms underlying cardiac regeneration. The carboxyl groups of HA are modified with N-hydroxysuccinimide (NHS) to yield HA-NHS groups. These HA-NHS groups react with amine groups in serum along with cells and tissue to form amide bonds. The hydrogel entraps stem cells and adheres to the tissue. HA: Serum hydrogel can be used for stem cell transplantation to the heart via intramyocardial injection or epicardial application. Our previous work has shown that hyaluronic acid (HA)-based hydrogels enhanced stem cell survival following transplantation in rodent models. Here, we tested the effects of encapsulating MSCs in HA-serum hydrogels (synthesized by crosslinking HA with autologous serum) by injecting MSCs with and without hydrogel into chronically ischemic myocardium in a porcine model with the goal of evaluating transplanted stem cell survival at several time points following transplantation
The experimental protocol was approved by the Institutional Animal Care and Use Committee of the National Heart, Lung, and Blood Institute, and the investigation conformed to the Guide for the Care and Use of Laboratory Animals (National Academy Press, 1996, Washington, D.C.). Eight Yorkshire domestic pigs, initially weighing 15-20 kg, were selected for this study. All animals were housed one per cage and allowed free access to water and commercial pig food.
As outlined in Figure 1, 8 animals underwent a small left thoracotomy under general anesthesia and had placement of an ameroid constrictor around the proximal left circumflex artery to create a model of chronic myocardial ischemia as described previously [5,6]. At this first operation, bone marrow (15 ml) was harvested for ex-vivo MSC expansion. Four weeks later, a second left thoracotomy was performed on each animal, the circumflex territory (ischemic zone) exposed and injected with exvivo expanded MSCs in one marked area and MSCs plus hydrogel in the other marked area, all within the ischemic zone. At 2, 4, 6, and 12 weeks following injection, cohorts of 2 animals were sacrificed and the hearts were then harvested and sectioned. The injected cell fate and angiogenic effects with or without hydrogel were tested by H & E staining and immunohistochemistry staining with anti-Von Willebrand’s factor antibody and smooth muscle actin.
Bone marrow-derived MSCs were prepared and cultured using the same methods described previously [5,6]. The MSCs were tested for CD markers, karyo type analysis, and their respective multi-potency differentiation abilities.
Chemical modification of carboxyl groups in HA to amine-reactive N-hydroxysuccinimide esters was achieved by reacting 10 w/v% HA (MW 16 kDa; LifeCore Biomedical) with 67 w/v% 1-ethyl-3-(3- diemthylaminopropyl) carbodiimide (EDC; Sigma), and 25 w/v% N-hydroxyl succinimide (NHS; Sigma) in phosphate-buffered saline (PBS) as previously described [9].
Peripheral blood from each pig was collected using serum tubes before surgery. For serum isolation, the blood was centrifuged at 1500 rpm for 10 min.
HA: Ser hydrogel was prepared by mixing in a ratio 10 w/v% HA-NHS dissolved in PBS with pig serum in a ratio of 1:1.5. All polymerization was carried out at pH 7-7.4 and room temperature for 5minutes. The cells were suspended in an appropriate amount of serum and the HA was dissolved in PBS. The two components were mixed for 30 seconds and the final volume of 600 µl of hydrogel was transferred to the barrel of a 0.5 mL sterile syringe using a viscous pipettes. We chose a 1:1.5 ratio for serum in a total of 600 µl and HA in order to obtain optimal viscosity of the hydrogel for injection and maximize adhesivity. In order to ensure functionality of the NHS groups following transplantation, hydrogels were synthesized within 5minutes of dissolving HA in PBS.

Figure 1: Experimental timeline demonstrating ameroid placement, injection of MSCs only or MSCs plus hydrogel, animal euthanasia and histologic evaluation at the end of the experiment.
Four weeks following ameroid placement, ex-vivo expanded autologous MSCs (1.2 × 108 ) were prepared and washed as described previously [5,6]. MSCs were suspended in 2.4 ml of normal saline and equally divided into two aliquots. One aliquot (MSCs only, 6 × 107 ) were injected intramyocardially in each animal using a 25 gauge needle (with an injection depth of 0.5 cm) within the suture-marked area within the left myocardial ischemic zone (12 injection sites, 100 µl in each site).The aliquot of MSCs was mixed with freshly prepared hydrogel as described above, and injected within another marked area within the ischemic zone the same way as described above.
At 2, 4, 6 and 12 weeks following injection, cohorts of 2 animals were sacrificed at each time point. The marked area of MSCs only and MSCs plus hydrogel were carefully cut off separately within ischemic zone. The specimens had the surrounding tissue trimmed off and were cut into 4 pieces. Each of these pieces was further cut into 3 parts of 5 × 5 mm thick slices and either collected in cassettes and fixed with 10% buffered formalin for paraffin embedding, or in OCT for frozen sections with no fixation. Paraffin embedded sections were stained with Hematoxyl in and Eosin and Masson trichrome for morphological analysis. Immunohistochemical or immunofluorescent staining was performed using mouse monoclonal anti-human antibodies. The incubations of primary antibodies were followed by detections of fluoresce in isothiocyanate or rhodamine conjugated with anti-mouse IgG and the nuclei were labeled with 4’, 6-diamidino-2-phenylindole (DAPI).
There were no signs of adverse effects after transplantation of MSCs encapsulated in hydrogels. Two unexpected animal deaths were found 1-2 weeks following ameroid placement due to large areas of myocardial infarction (one animal died due to severe surgical wound infection and another died due to pulmonary edema following ameroid placement as determined by autopsy examination). No post-cell injection related animal deaths were found in this preliminary study. Eight animals survived to scheduled euthanasia.
In this large animal model study, we found that the transplantation of MSCs encapsulated in hydrogels boosted the retention of transplanted cells in the injected area and stimulated angiogenesis compared to the MSC only injected areas in the early stage. At 2 weeks following injection, hydrogel injected areas demonstrated evidence of more cell aggregation compared to areas where only MSCs were injected. In the MSC plus hydrogel transplantation group, the MSCs migrated towards the periphery of the cell clusters close to the surrounding myocardium (Figure 2). This migration occurred despite homogeneous dispersion of the hydrogel and cells prior to injection. This suggests that once injected, MSCs show more aggregation near host tissues. Furthermore, a greater amount of cell clumps are still seen in the hydrogel group compared with MSC only injection during later time points (Figure 3 and Figure 4).
Hydrogel degradation: Once injected, hydrogel solidified and can be observed as the white colored area in H&E staining sections at the 2 week time point (Figure 2). Importantly, there was very little evidence of injected hydrogel at 4weeks (Figure 3), which was attributed to degradation.
Fibrosis: We observed greater amounts of fibrosis in hydrogel-injected areas, when compared with MSC only injections (Figure 3 and Figure 4) - the differences were most pronounced at 6 and 12 weeks. At 12 weeks, the fibrosis became much more severe and could be visualized on gross macroscopic inspection, demonstrating dramatic differences between MSC only and MSC plus hydrogel injection areas (Figure 5 and Figure 6).
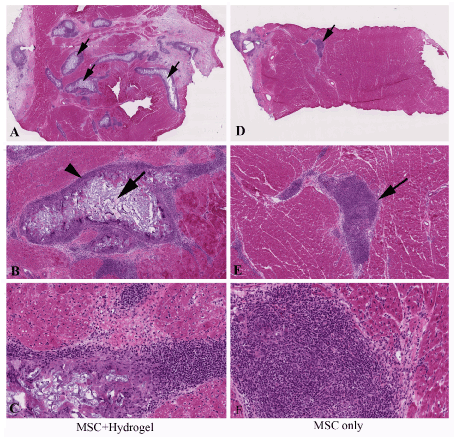
Figure 2:At 2 weeks following injection, hydrogel injected areas showed more cell clumps (A,B,C) compared with MSC only injections (D,E,F). Solidified hydrogel is seen in the center of the cell clumps (arrows in A and B) and injected cells migrated to the edge of the cell clumps (arrow head in B). Magnifications are AD=10X, BE=50X, and CF=200X.
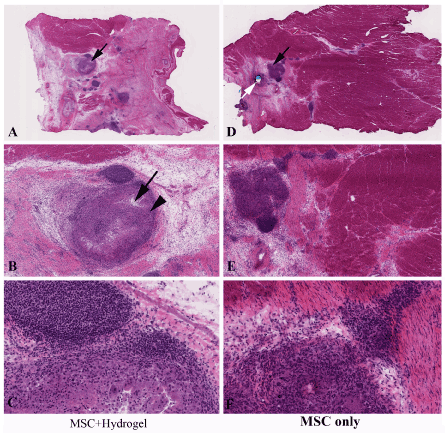
Figure 3: At 4 weeks, hydrogel gradually absorbed (A,B arrows), but the injected area showed more fibrosis compared with MSC only injections. In D, black arrow indicating an injected cell clump, and white arrow indicating a suture used for marker of the injection. Magnifications are AD=10X, BE=50X, and CF=200X.
Inflammatory cell infiltration/Angiogenesis: At the 2 and 4 week intervals, inflammatory cell infiltration in the hydrogel-containing region was observed as well. We performed the immunofluoresent staining of factor VIII and smooth muscle actin in frozen sections at 2, 4, 6, and 12 weeks after injection. In hydrogel injected animals, there appeared to be increased capillary density suggested by positive factor VIII and SMA staining around the spherical cell cluster at 2 weeks. These differences became less apparent at later time points (Figure 7).
Since cell-based therapy was introduced as a method to stimulate therapeutic angiogenesis for ischemic heart disease at the turn of the century [13-23], optimal cell delivery technique has remained a hotly contended topic. Intracoronary infusion was initially attempted for patients with ischemic coronary artery disease [3,13-18]. The major limitation of this practice was the need to obstruct coronary blood flow during cell infusion and a few seconds thereafter, which may cause the flushing out of cells after reperfusion. Similarly, intravenous infusion of allogeneic MSCs led to entrapment of most of the transplanted cells in the lungs instead of the heart, as reported by imaging [4]. Investigators have also tried to deliver the cells in tramyocardially using special injecting devices [20,21,24], or by direct epicardial injection during open heart surgery [2]. This partially solved the issue of cells homing to the target tissue. However, cell retention was less than ideal after being injected into the beating myocardium. Therefore, polysaccharide-based strategies have been studied in an effort to create a friendly environment to increase the survival and retention of cells after delivery [9-12]. Investigators believe that the lack of extracellular matrix is one of the main reasons for reduced cell retention after delivery [25].
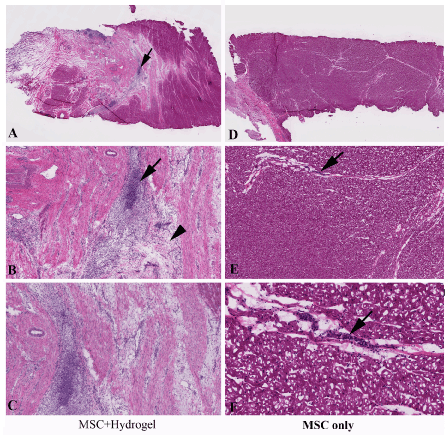
Figure 4: At 6 weeks, the fibrosis seems dramatically increased in the hydrogel injected area. More cell clumps are still seen in the hydrogel group compared with MSC only injection. Arrows indicating cell clumps, arrow head indicating fibrotic tissues. Magnifications are AD=10X, BE=50X, and CF=200X.
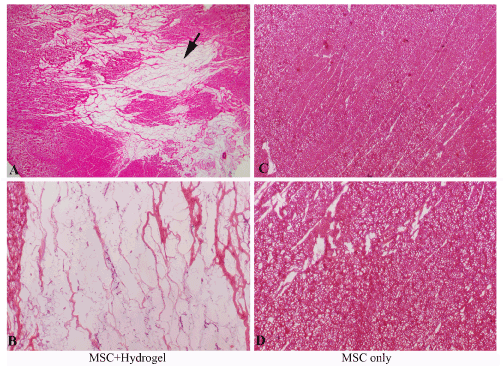
Figure 5: At 12weeks, the hydrogel injected area showed large areas of fibrotic tissue. Arrow is indicating one of the fibrotic areas. Magnifications are AC=100x, BD=200x.
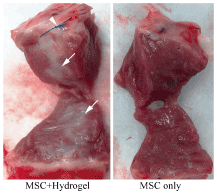
Figure 6: At 12 weeks, hydrogel injected area showed large area of fibrotic tissue, which can be seen on macroscopic inspection. White arrows indicating the fibrotic areas in the tissue clump that cutting out from marked injecting areas which showed dramatic differences between MSC only and MSC plus hydrogel injection areas.
Hyaluronic acid, or hyaluronate, is one of several kinds of polysaccharides that are important constituents of the extracellular matrix [10-12]. Since HA has strong viscosity and is able to retain water, it plays a very important role in tissue homeostasis and biomechanical integrity. We have used modified preparation of HA with serum, a product we called hydrogel, in a rodent model of acute myocardial infarction with cardiosphere-derived cells (CDCs) and found that these hydrogels stayed in the injected site for up to 4 weeks. CDC encapsulation with hydrogel has been shown to promote survival and proliferation and improve left ventricular ejection fraction following myocardial infarction [9].
In the current study, we tested the effects of hydrogel in a porcine model of chronic myocardial ischemia to see whether cell retention and survival improved at several time points following MSC and hydrogel transplantation. We found that the hydrogel dramatically increased cell retention and promoted angiogenesis at early stage, however the hydrogel also caused large amounts of fibrosis starting from 4 weeks and reaching a peak at 12 weeks. We carefully compared these results with our rodent model, which did not cause obvious fibrosis as the injections were made in myocardial scar tissue. In the large animal study model, we delivered the cells with hydrogel in ischemic but viable myocardium, which could result in destruction of the cardiomyocytes in the injection tract since the gel is retained in the myocardium. Therefore, it seems that hydrogel injection into viable myocardium may induce myocardial damage due to solidified hydrogel after injection and takes longer time to be absorbed. Whether the resultant fibrosis from the hydrogel injection has an effect on left ventricular function is not known. Functional studies are warranted to further delineate any benefit of encapsulated cell delivery in ischemic myocardium. These studies may provide more definitive data on whether HA-serum hydrogels lead to a synergistic benefit in injected stem cells and may further optimize the usage of HA-serum hydrogels in cellbased therapy. While differentiation of injected stem cells into functional cardiomyocytes is the ultimate goal of cell based therapies, several studies have shown poor cell differentiation following transplantation. Possible explanations for this include poor cell engraftment, scant survival and inadequate proliferation.

Figure 7: Immunofluoresent staining of factor VIII (green), smooth muscle actin (SMA) (red) and DAPI (blue) in frozen section at 2, 4, 6, and 12 weeks after MSC only and MSC plus hydrogel injection. In hydrogel injected animals, there appeared to be increased capillary density suggested by positive factor VIII and SMA staining around the spherical cell cluster at 2 weeks. These differences became less apparent at later time points. White arrows indicating cell clusters. Magnifications are 100X.
The authors are particularly grateful to their colleagues in the Laboratory of Animal Medicine and Surgery, NHLBI for assisting in animal surgery for the study.
Download Provisional PDF Here
Article Type: Research Article
Citation: Zhou Y, Wang S, Yu Z, Hoyt RF Jr, Karakas MF, et al. (2015) Effect of Hydrogel in Cell-Based Therapy in a Porcine Model of Chronic Myocardial Ischemia. Int J Stem Cell Res 1 (1): doi http://dx.doi. org/10.16966/2472-6990.102
Copyright: © 2015, Zhou Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access