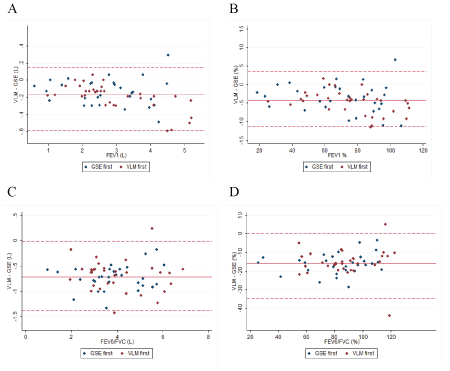
Figure 1: Bland Altman plot. The solid line represents the mean difference in litre. The dotted line represents the limits of agreement.


Soren Sperling Haugen11* Majbritt Jeppesen1 Hanne Vebert Olesen2 Signe Fog Sonderup1 Lotte Orneborg Rodkjaer1 Soren Fangel-Jensen1
1Department of Infectious Diseases, Aarhus University Hospital, Denmark*Corresponding author: Søren Sperling Haugen, Department of Infectious Diseases, Aarhus University Hospital, Denmark, Tel: 81.263.33.7215; E-mail: sonpde@rm.dk
Cystic fibrosis (CF) is a systemic life-shortening autosomal recessive disease that mainly affects the respiratory and digestive systems. Pathogens cause infections and inflammation in the lungs, leading to progressive respiratory failure, which is the major cause of morbidity and mortality among patients with CF [1-4]. In Denmark, the treatment of CF is centralised to two centres located at Aarhus University Hospital and Rigshospitalet Copenhagen. Most patients with CF are seen in the outpatient clinic every 4-6 week, where an experienced physician will evaluate the patient’s general health, nutritional status, diabetes, infections and lung function. The monthly visit to the hospital requires great flexibility for patients and their families. Telemedicine has the possibility of contributing to a more flexible outpatient clinic.
Recommended lung function tests are used to evaluate the overall physiological condition of the lungs, the effect of ongoing treatment and the need of additional treatment [5,6]. Forced expiratory volume in one second (FEV1) and Forced Vital Capacity (FVC) are golden standard measurements for lung function testing [7,8].
The interest in home monitoring of patients with CF has increased over the past decade [9]. In order to monitor patients at home, several studies have investigated the use of small handheld devices for lung function testing at home.
Grzincich et al. explored the experience and adherence of real-time monitoring of spirometry for four weeks among 30 patients with CF. 70% of the patients were satisfied with the possibility to evaluate their own health [10]. Jarad et al. explored adherence to daily measurement for 6 months. 63% of patients with CF were withdrawn from the study due to lack of adherence [11]. Poor adherence to therapy is well identified among patients with CF and poor adherence is known to be associated with higher diseases burden [12-14]. Two studies have also tried to predict pulmonary exacerbation using FEV1 without convincing results [15,16].
Most studies regarding CF have failed to test the accuracy of the device for home monitoring. Any differences between devices should be investigated and considered when exploring home monitoring.
We hypothesize that home monitoring could contribute to a more flexible outpatient clinic and increases patient empowerment for patients with CF.
In this study, we aim to explore the validity of equipment for home monitoring of lung functions. In addition, we wish to investigate adherence to daily monitoring and patients’ experiences with home monitoring. We explore whether administration of intravenous antibiotics caused a change in FEV1 four weeks during and after treatment in their own home.
We measure changes in quality of life and health status to explore if home monitoring affects health parameters as health perception and treatment burden.
The project was approved by the Danish Research Ethics Committee and The Danish Data Protection Agency (file number 2012-58-006).
The project consisted of two parts.
Study design: 63 patients were randomised in a cross-over study. The aim was to compare the handheld Vitalograph model 4000 lung monitor (40750) (VLM) for home use with The CareFusion Jaeger Vyntus SPIRO, which is used as golden standard equipment (GSE) in the outpatient clinic.
Data collection: Data were collected from adult patients (i.e. 18 years or older) with CF at the Department of Infectious diseases, Aarhus University Hospital, Denmark. Lung function testing was performed in the outpatient clinic and supervised by experienced personal.
A pause between tests was given to avoid fatigue influencing the results. According to ATS/ERS guidelines one lung function test consisted of three to eight manoeuvres. The highest values of FEV1 and Forced expiratory volume in six second (FEV6) in litres were collected from the VLM (VLMFEV1 VLM FEV6) and the highest values of FEV1 and FVC in litres and percent were collected from GSE (GSEFEV1 GSEFVC). For each test the GSE calculates an expected FEV1 and FVC based on each participant’s gender, height, weight and race. Based on the expected lung function a value in percent is then calculated by the GSE. The ratio from GSE endpoints in litre and percent were used to calculate endpoint in percent from VLM. All lung function parameters were collected as recommended by ERS/ ATS guidelines [17].
Statistical analysis: Comparison of lung function measurements were analyzed as paired continuous data in STATA13. VLMFEV1 was compared to GSEFEV1. The VLM measures FEV6 as a surrogate for FVC, why VLMFEV6 is compared to GSEFVC.
The differences between FEV1 from GSE and VLM were illustrated using Bland-Altman plots. All data on lung function parameters is illustrated as mean values with 95% confidence intervals.
Participants were sub grouped into gender and order of randomisation to investigate if any of these factors determined the difference in lung function parameters.
Study design: A case-series report on 10 adults with CF, who were instructed to perform one daily lung function test for four weeks during and after a two-week intravenous antibiotic treatment in their own home.
Data collection: Patients were included around the beginning of an intravenous treatment with antibiotics in the outpatient clinic. Health status and quality of life (QoL) was measured using Cystic Fibrosis Questionnaire (CFQ-R14+) for adults on randomization and after the study period [18]. The questionnaire consists of 50 self-reported items. Answers are transformed into scaled scores from 0-100 within twelve domains. At the end of the study period patients were invited to participate in a semi-structured interview regarding their experience with home monitoring focusing on the following issues: convenience, utility, usability, training and technical support. Data was analyzed using Meaning Condensation [19]. Meaning condensation entails a summary of the meanings expressed by the interviewees into shorter formulation. Transcripts from the interviews were read and analyzed and then organized in central themes. Long statements are compressed into briefer statements in which the main sense of what is said is rephrased in a few words. Themes from each interview were compared and variations and similarities were identified. The analysis involves five steps [19].
Statistical analysis: A simple linear regression model was used on each patient’s home measurements. Variation around the estimated regression line was calculated as a 95% prediction interval.
The T-test was used to investigate if adherence from day 1-14 changed compared to mean adherence from day 15-28. Results from the overall QoL and all twelve subcategories before and after the study period were compared. Change in QoL for each participant were analyzed as paired data, while mean QoL from baseline and QoL after home monitoring were analyzed as unpaired data.
Altogether, 63 adults participated, which corresponds to 85% of all eligible patients at the center. Trail profile in appendix.
Characteristics of the study population.
| Gender | Age | Randomization | FEV1 | FVC |
| Female 36 Male 27 | Mean 28 (range, 18 to 50) | GSE before VLM 30 VLM before GSE 33 | Mean FEV1 in litre: 2.94 L (range 0.64 to 5.42) Mean FEV1 in %: 75.0% (range 19.7 to 114.7) | Mean FVC in litre: 4.29 L (range 1.25 to 7.17) Mean FVC in %: 93.2% (range 33.5 to 141.2) |
The mean VLMFEV1 was 2.77 L (CI 95%: 2.49; 3.05) and the mean GSEFEV1 was 2.94 L (CI 95%: 2.64; 3.23). The mean difference between VLMFEV1 and GSEFEV1 was -0.17L (CI 95%: -0.21; -0.13), p<0.001 corresponding to a difference of -4.24% (CI 95%: -5.13; -3.43). The mean difference between the devices, ± limits of agreement corresponding to 95% prediction intervals are shown in a BlandAltman plot (BA-plot) (Figure 1A+B).

Figure 1: Bland Altman plot. The solid line represents the mean difference in litre. The dotted line represents the limits of agreement.
The mean VLMFEV6 was 3.58 L (CI 95%: 3.24; 3.91) and the mean GSEFVC was 4.29 L (CI 95%: 3.95; 4.62). The mean difference between VLMFEV6 and GSEFVC was -0.71 L (CI 95%: -0.77; -0.64), p<0.001corresponding to a mean difference between the VLMFEV6 and GSEFVC of -15.7% (CI 95%: -17.4; -14.1).
The difference in FEV1 was not associated with randomization (p=0.12) or gender (p=0.13) (Figure 1A-1D).
All lung function tests performed in the outpatient clinic were monitored by a member of the research group. The VLM required a slightly different technique than GSE. The difference in the technique seemed to trouble some of the patients resulting in larger differences between VLM and GSE.
Patients’ characteristics: Ten patients were enrolled in the Part B. In the study period, 13 eligible patients received IV antibiotic treatment. Two patients refused to participate, and one patient was excluded due to severe psychological condition.
The mean VLMFEV1 was 2.39 L with a range from 1.24 to 3.67 L. One patient, ID 01 failed to hand in the VLM after the study period. 8 out of 9 patients completed the CFQ-R14+ after the study period.
5 out of 9 patients were interviewed after the study period.
Home measurements: Regression analyses were made on all patients’ home measurements (Table 1). ID 1, 4 and 8 did not produce enough data to the analysism day 1 until day 14 the mean adherence was 60% (CI 95%: 35; 86) contrary to day 15 until day 28 where the mean adherence was 33% (CI 95%: 4; 61). Difference in adherence from the first 14 days of the study compared to the last 14 days was significant p=0.023 (Table 1).
| ID | Overall tests performed | Tests performed from day 1-14 |
Tests performed from day 15-28 |
Coefficient r (CI 95%) | Variation around r | T-test (r=0) |
| 1 | - | - | - | |||
| 2 | 19 (68) | 13 (93) | 6 (43) | 0.001 (-0.003; 0.005) | 0.139 | 0.569 |
| 3 | 9 (32) | 6 (43) | 3 (21) | 0.015 (-0.005; 0.034) | 0.334 | 0.118 |
| 4 | 2 (7) | 2 (14) | 0 (0) | - | - | - |
| 5 | 11 (39) | 8 (57) | 3 (21) | -0.006 (-0.016; 0.005) | 0.229 | 0.254 |
| 6 | 23 (82) | 11 (79) | 12 (86) | 0.005 (0.002; 0.007) | 0.086 | 0.001* |
| 7 | 10 (36) | 7 (50) | 3 (21) | 0.007 (0.000; 0.013) | 0.0146 | 0.049* |
| 8 | 2 (7) | 2 (14) | 0 (0) | - | - | - |
| 9 | 28 (100) | 14 (100) | 14 (100) | -0.021 (-0.034; -0.008) | 0.545 | 0.003* |
| 10 | 14 (50) | 13 (93) | 1 (1) | 0.001 (-0.011; 0.012) | 0.222 | 0.863 |
Table 1: Test performed: x(y). x represents number of days. Y represents percent of total tests. Coefficient (litres/day) based on a simple linear model. Variation estimated as a PI 95% interval. T-test, r=0.
Questionnaire: Baseline questionnaires were completed on the first day of the IV treatment. Questionnaires after home monitoring were on average handed in 47 days after baseline questionnaire, ranging from 28 to 65 days.
The mean baseline score was 70.3 (CI 95%: 61.5; 79.2). The mean score after home monitoring was 66.4 (CI 95%: 52.7; 80). No significant change in overall QoL was found, p=0.19. Change in overall QoL is presented in figure 2. The CFQ-R14+ QoL score contains 12 subcategories, which are presented in figure 3. No significant difference was found in any subcategory.
Patient interview: Five participants completed the interview. Overall participants experienced monitoring their lung function as “A new visual partner”. The following inter-connected themes emerged.
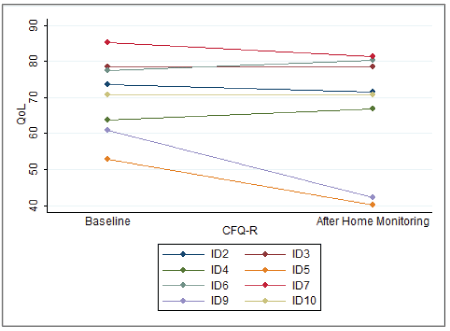
Figure 2: Overall change in QoL from Baseline until After Home Monitoring.
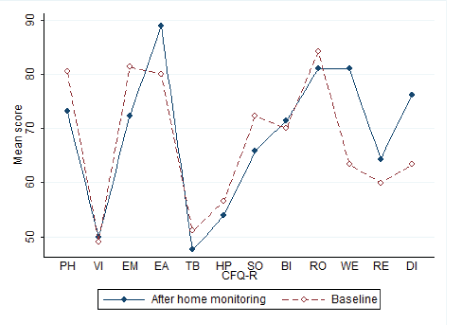
Figure 3: Change in subcategory.
Advantages: gives security and consciousness about disease, measurements could change perception of disease status, gives extra sense of security, disease becomes more visible, more control, and larger responsibility.
Disadvantages: conscious about disease, worries about measurements, psychological impact if the lung function decreases, increased focus on disease, takes more focus in everyday life, breach of daily routines, requires energy and time.
Most of the previous studies on home monitoring of patients with CF have been small descriptive studies with limited external validity. A request for studies with CF patients receiving intravenous treatment and a measure of QoL during home monitoring has been made [9]. We have conducted a trial that cover both these requests. In addition to this, we have conducted a randomized trial that gives important insights into the validity of a hand-held device for home monitoring.
Comparison of VLM and GSE: The mean difference in FEV1 between the two devices was -0.17L, p<0.001. Similar results have been found in studies with other handheld spirometers [20].
The Bland-Altman plot shows that the difference between the VLM and GSE was biased; meaning that the VLM almost consistently measured a lower FEV1 . In total, 55 of 63 participants performed a lower VLMFEV1 compared to GSEFEV1. Clinically this could prove to be an advantage, because it will allow clinicians to always take a negative difference into account when assessing home measurements. However, any difference in parameters makes it more difficult and uncertain to use home measurement.
The mean difference between VLMFEV6 and GSEFVC was -0.71 L. FEV6 has been investigated as a surrogate for FVC in primary care to detect COPD [21]. The difference between FEV6 and FVC has also been proven to be larger among patients with obstructive airways [22]. Patients with CF almost always have a degree of airway obstruction which could explain the large difference found in this study.
The reasons for the difference in lung functions parameters were not investigated. Observations on technique were not scored but should be considered in future use and studies. Future studies on how training on the VLM affects the difference in FEV1 would be of great importance for both clinicians and patients.
After the study period of 28 days, all patients received a reminder to hand in their VLM on their following visit. Still, one patient failed to bring the VLM on two following visits in the outpatient clinic.
Home measurements: Only 3 out of 9 patients showed a significant change in FEV1 during the study (Table 1). The CI 95% from these patients (ID 6, 7, 9) indicate that their change in FEV1 seems clinically irrelevant, considering expected daily variation [23]. Hence, during study period we were not able to visualize any clinically relevant change in FEV1 during and after 2 weeks of IV antibiotic treatment.
In this study one patient responded to a larger drop in FEV1 at home by contacting the outpatient clinic. The patient was seen in the outpatient clinic and hospitalized to further treatment. This example illustrates that even though VLM differs from GSE, changes in FEV1 is useful when FEV1 values from the VLM are compared to previous FEV1 home measurements.
Adherence: This study shows that home monitoring of lung function is possible during and after IV treatments of antibiotics. In fact, our data shows that adherence during treatment was significantly higher than adherence after treatment (60% compared to 33%, p=0.023).The novelty value of new equipment might also contribute to a larger adherence in the first part of the study period. The results from patients’ lung function tests were visualized with a FEV1 in litre and in percent (supplementary). Patients’ were not able to visualize their FEV1 values on a graph until the end of the study. A continuing visualization of FEV1 over time could motivate for ongoing tests and increase adherence.
Adherence to daily monitoring varied greatly from 7% to 100%. This indicates that clinicians should be aware, that not all patients are qualified for home monitoring. Low adherence could facilitate negative feelings of guilt. On the other hand, patients performing daily monitoring for a longer period might be in risk of excessive monitoring. Further studies are needed in order to define which patients could benefit from home monitoring.
Questionnaire: Reports from the CFQ-R14+ showed lowest scores in the 3 domains on vitality, treatment burden and health perception. This corresponds well with earlier findings in Danish patients with CF [24]. Since our data on QoL is not compared to a control group, we cannot conclude whether QoL is affected by the IV treatment or home monitoring. Home monitoring has previously been shown to be a benign intervention for patients with CF [25]. Still no significant change in overall QoL or any of the twelve domains was found in our study.
ID 5 and ID 9 had a large drop in overall QoL during the study. These two patients were the only with a negative correlation coefficient (r) (Table 1), although this was not statistically significant for ID 5. Both patients reported poor effect from their IV treatment and received additional treatment after the study period. The lack of effect from their treatment corresponds with their negative correlation coefficient and could explain their drop in overall QoL.
Interview: Overall patients were comfortable with the use of VLM. The interviews provided clinicians with insights on how patients’ experiences their diseases, how home monitoring can support and affect their daily living in a positive or negative direction. To avoid unnecessary worries regarding measurements, patients must be giving information on fluctuation in FEV1 and interpretation of measurements before starting home monitoring.
Download Provisional PDF Here
Article Type: RESEARCH ARTICLE
Citation: Haugen SS, Jeppesen M, Olesen HV, Sønderup SF, Rodkjær LO, et al. (2018) Evaluation of Home Monitoring for Patients with Cystic Fibrosis: A Feasibility Study. J Infect Pulm Dis 4(2): dx.doi.org/10.16966/2470-3176.136
Copyright: © 2018 Haugen SS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access