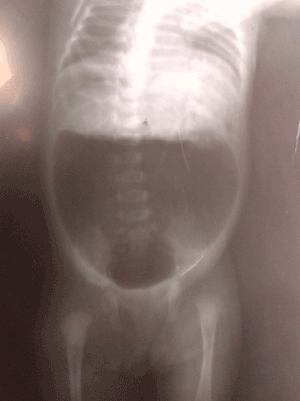
Figure 1: Invertogram Showing Large air-fluid level.

Raashid Hamid* Bhat Nisar Ajaz Baba Mufti Gowhar Sajad Wani
Department of Paediatric and neonatal Surgery, SKIMS, Srinager, Jammu and Kashmir, India*Corresponding author: Raashid Hamid, Department of Paediatric and neonatal Surgery, SKIMS Srinager, Jammu and Kashmir, India, Tel: 9469451875; E-mail: drraashidhamid@gmail.com
Background: Congenital pouch colon (CPC) is a rare disease associated with anorectal malformation (ARM). This study aimed to review cases in our institute with regard to clinical presentation, diagnostic features and management of this rare clinical entity.
Material and methods: From Dec- 2008 to Dec- 2013 records of 10 patients of CPC associated with ARM were reviewed. Information about demography, perineal examination, X-ray abdomen, invertogram, abdominal USG, voiding cysto-urethrogram, type of pouch and surgical management and outcome were retrieved and analysed.
Results: All 10 patients had Type- IV pouch colon, 8 patients underwent laparotomy with excision of colonic pouch after ligation of fistula and end colostomy as stage-I procedure. Abdominoperineal pull through was accomplished subsequently in these 8 patients. In remaining 2 patients loop colostomy was initially made and later division of the fistula with abdomino PSARP and colostomy closure in third stage. Preoperative diagnosis was made in 5 patients and remaining 5 patients were diagnosed intraoperatively. The mean age at presentation was 3.2 days ± 7.33 days. Eight patients were males and 2 females. No significant complications occurred postoperatively. There was no mortality in our series.
Conclusion: Although CPC is a rare condition associated with ARM, incidence in our state is similar to that found in other north-western states of our country. Evidence based management is needed to reduce morbidity and mortality.
Congenital pouch colon; Anorectal malformation; Fistula; Extrophia splanchnica; Congenital absence of colon and rectum; Colonic reservoir and Association of Imperforate Anus with Short colon (AIASS); Posterior Sagital Anorectoplasty (PSARP)
CPC is a rare anomaly in which whole or part of healthy colon is replaced by a pouch like structure that communicates with urogenital tract by means of a fistula [1,2]. This condition occurs more frequently in the northern, north-western and central parts of India, and neighbouring countries like Bangladesh, Pakistan and Nepal [3]. Few sporadic cases have been reported from other parts of the world [4]. Exact cause of this unique demographic distribution has not been yet ascertained. We present a series of 10 cases of CPC managed at this tertiary care hospital from Jammu and Kashmir, which is situated in northern India. Our primary aim was to see the clinical profile and operative results. The secondary aim was to look for functional outcome.
Our study was a retrospective, descriptive study of CPC cases. Between Dec- 2008 to Dec -2013, a total of 120 cases of ARM were managed in the department of Paediatric Surgery. Out of 120 cases of ARM, 10 cases were diagnosed to have associated CPC. Analysis of patient characteristics, diagnosis and management and postoperative outcome was made. For descriptive purpose, CPC was classified into 4 types as per Rao et al. classification of pouch colon [5] Rao et al. classified CPC cases into four types and are as follows:
Type I: Normal colon is absent and the ileum opens directly into the colonic pouch.
Type II: The ileum opens into a short segment of caecum which then opens into the pouch.
Type III: Presence of a significant length of normal colon between the ileum and the colonic pouch.
Type IV: Presence of near normal colon with only the terminal portion of colon (sigmoid and rectum) converted into a pouch.
Out of 120 cases of ARM, 80 were high type and 10 cases had associated CPC, with an incidence of 8.33 % of all cases of ARM, and 12.5% of high type ARM . Male to female ratio was 4:1. The mean age at presentation was 2.84 ± 4.3 days with range of 10 hours to 6 days. Weight ranged between 1.8 to 3.5 kgs with a mean of 2.7 ± 0.54 kg. In 5 (50 %) patients , preoperative diagnosis was made on erect and invertogram x- ray delineating the typical findings including a large air-fluid level which occupied more than half of the width of abdomen (Figure1). Intraoperative diagnosis was made in remaining 5 patients, and operative findings were thick walled abrupt change of calibre of distal colon to a pouch (Figure 2). The pouch had short mesentery in most cases (Figure 3). Histopathological report was available in 7 patients, which in most cases revealed thickening of sub mucosa and criss-cross pattern of muscle fibers. One female baby had colovesical fistula with hemivagina and another female baby had colovesical fistula only.
All the cases were Type -IV according to the Rao et al. classification. In 8 cases pouch was excised after the ligation of fistula and end colostomy was made. Abdominoperineal Posterior sagital anorectoplasty (PSARP) was accomplished in stage - II. Nine patients had pouch vesicle fistula; one had pouch prostatic urethral fistula. In two patients with low birth weight, loop colostomy was done initially to decrease the operative time. This wasfollowed by division of fistula, pouch excision with abdomino-PSARP. Subsequently, stoma closure was done after six weeks.

Figure 1: Invertogram Showing Large air-fluid level.
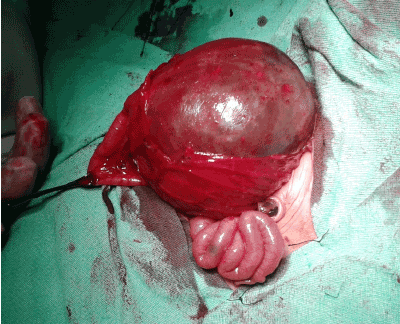
Figure 2: Intra operative picture showing Abrupt change in caliber of the colon.
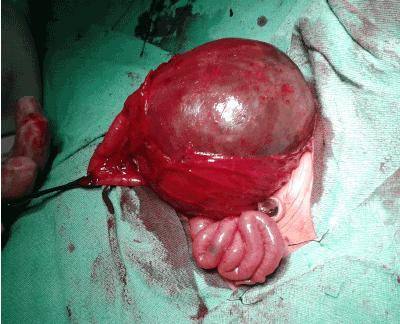
Figure 3: CPC showing Typical abnormal vasculature.
Ultrasonography detected bilateral hydroureteronephrosis in two patients, which were demonstrated as vesicoureteric reflux on voiding cystourethrogram. Six out of 10 patients had associated congenital anomalies including cleft lip in 1, polydactyly in 1, ASD in 1 and malrotation in 2 patients (Table 1). Subsequently, after a period of 3-4 months definitive abdominoperineal pull through was done as second stage repair. After definitive pull through procedure anal stenosis occurred in 3 patients, stricture in 1 and mucosal prolapses in 2 patients.
Patients were followed over a period ranging from 6 months to 4 years with a mean follow-up of 22.9 months after definitive repair. Anal stenosis responded to dilatation. Stricturoplasty was performed in 1 and mucosal prolapse excision in 1 patient. The records of patients who were assessed for fecal incontinence was available in 8 patients in follow-up. Continence was assessed after 3 years of age (as babies develop continence after 3 years, so that is why continence is assessed after 3 years of age). Continence assessment is a continuous process in which patient is assessed in surgeons office. Continence was good in 5 and fair in 3 patients assessed by Kelly’s method.
Various terms have been used to describe the pouch colon including pouch colon syndrome, extrophia splanchnica, congenital absence of colon and rectum, colonic reservoir and association of imperforate anus with short colon (AIASS) [6]. The incidence in our series was 8.33% and the reported incidence from other Indian case series is 5-18% [5]. The incidence could be higher in this part of the world which falls in Northern area of India, which is known to have higher incidence of CPC with ARM. In the recently conducted survey on CPC from various pediatric surgical centres in India, Pakistan, Bangladesh, Nepal, Sri Lanka, Italy, Sweden, and Japan, the incidence was reported to be the highest in north India (Kashmir, Chandigarh, Delhi, Lucknow, Varanasi), but decreased as we proceeded toward the east [6]. The majority of cases are reported from northern India. In Bangladesh and Pakistan incidence reported is 1.07% and 8-10% respectively. Incidence in the western world is about 1% [1]. A few reports have originated from China, Japan, and other parts of the world [5]. The cause of this unique geographical distribution has not yet been ascertained.
Sex ratio reported in a series outside Indian subcontinent has been almost equal (male –female ratio; 1.27:1) while it is 3-4:1 in India. In our series male –female ratio was 4:1. Interestingly, the sex ratio reported by authors outside India has been almost equal (1.27:1), while in India the reported incidence has been 3–4.3: 1 [6].
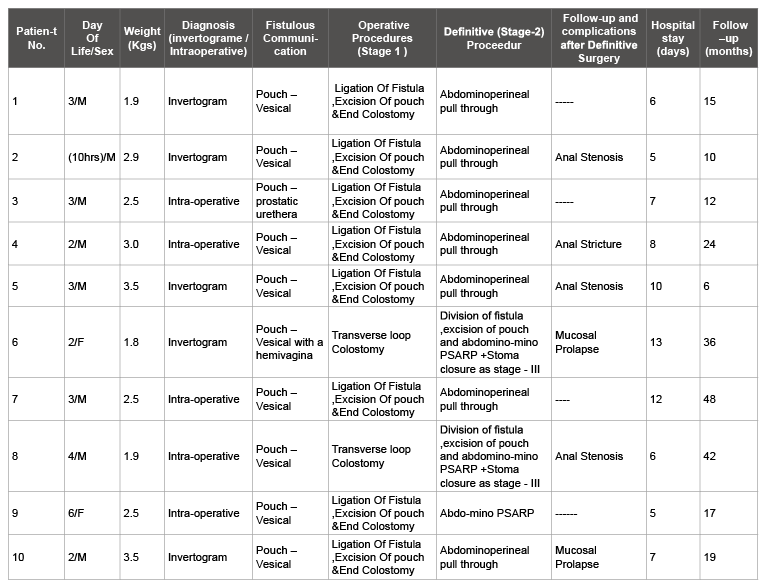
Table 1: Clinical characteristics of congenital pouch colon (n=10)
The possible aetiology and embryological basis of CPC include hindgut stimulation, vascular insult, faulty rotation and fixation of gut [1,21]. Trusler proposed that the dilatation was the result of chronic obstruction [7]. Another theory proposed was aborted hindgut development following obliteration of the inferior mesenteric artery early in fetal life [8]. Chatterjee proposed that the caecum and right colon develop normally from the postaxial midgut when this portion of the midgut is stimulated by normally developing hindgut [9]. Wu Yuejie suggested that faulty rotation and fixation of the colon leads consequently to a disturbed longitudinal growth [10]. Disturbance of the partitioning of the cloaca by the urorectal septum could explain the different types of the malformation [11]. Wakhlu et al. have postulated that CPC represents a stage in the development of cloacal exstrophy and is the combined effect of defective development of the splanchnic layer of the caudal fold and failure of rotation of the gut, causing defective longitudinal growth of the colon [12].
One different observation in our series is that all the 10 cases were Type - IV colonic pouch, which is not the distribution reported from the other authors. Mirza et al. reported 21 patients of CPC, out of them 9, belonged to Type- I, 5were Type- II, 2 had Type- III and 5 patients had Type IV CPC [14]. Another series of 97 cases of CPC by Ghritlaharey et al. had (31, Type -I), (10, Type- II), (27, Type- III), (29, Type- IV) pouch colon [15]. Type – IV CPC is now being increasingly reported by authors like Pavai et al. and Bhat, Pavai et al. reported four CPC cases associated with low anorectal anomaly, Bhat in a series reported 13 cases of Type- IV CPC out 17 cases [16,17].
Presentation may be early or delayed depending upon, the size of the fistula and type of CPC. The male baby presents early with anorectal agenesis and gross abdominal distension with or without passage of gas or meconium per urethra. Bilious vomiting with early gross distension of the abdomen in a case of ARM is strongly suggestive of congenital CPC. The female baby presents with passage of meconium from an abnormal opening, absent anus, and abdominal distension. A wide fistula could manifest late due to decompression by meconuria. Usually at that time these children are constipated and passing faeces from an abnormal opening. Fistula with genitourinary tract is characteristic of CPC. In males fistula is mostly with urinary bladder and in females fistula varies from perineum to vestibular to a cloacal malformation [18]. On examination a cloaca is usually found. Some authors have reported colouterine or colovestibular fistula also [12], although in our series there was no case of colouterine or colovestibular fistula. In cases of colonic perforation occurring early in patients with CPC, the baby may present with septicaemia, gross abdominal distension with prominent veins, fluid and electrolyte imbalance, and features of peritonitis. Sometimes a child may present with a colostomy performed by a surgeon who was unaware of this condition and subsequent development of complications like stenosis and prolapse, but none of our patient had prior colostomy by some other surgeon.
Preoperative diagnosis can be made early with radiography (erect and invertogram) [2]. A large loop of bowel filled with air and meconium, occupying more than half of the abdominal cavity is diagnostic of CPC in a patient with ARM. Sometimes air vesicogram may be evident in radiograph [14]. In our series among associated anomalies malrotation was most common, which is consistent with observation reported by other authors [1,6].
In our series most of the cases in our series presented in early neonatal period with abdominal distension. These babies present with an absent anal opening. There are two or more steps in the staged procedure depending upon the choice of procedure, which in turn depends upon the condition of the baby at presentation, the technical skill of the surgeon, and the availability of facilities for major neonatal surgery and postoperative care. Proximal diversion may take place in the form of: (1) End colostomy after division of the fistula and excision of the pouch in incomplete CPC (preferred approach). (2) End colostomy after division of the fistula and coloplasty in complete CPC. (3) Window colostomy, in which an opening is made on the anterior surface of the colonic pouch without attempting to ligate the fistulous connection. (4) Proximal ileostomy in complete CPC. (5) Transverse colostomy in incomplete CPC.
Excision of the pouch with an end colostomy is the procedure of choice. Window colostomy is simple surgery, can be performed with minimum anaesthesia time in a sick neonate, and provides adequate decompression and a time period to allow for weight gain and fitness for the second stage. However, the mortality following coloplasty with end colostomy in the newborn period is higher; hence this is better done at a later date.
In our study all the patients were managed by staged procedures. Eight patients underwent laparotomy with excision of colonic pouch after ligation of fistula and end colostomy as stage-I procedure (Figure 4). Colostomy was closed in second stage operation. Abdominoperineal pull through was accomplished subsequently in these 8 patients. In remaining 2 patients loop colostomy was initially made and later division of the fistula with abdomino PSARP and colostomy closure in third stage.
Advantages of staged procedure include sufficient time for proper investigations, can be performed in all types of CPC, procedure is well tolerated at few months of age, complications observed after definitive procedure are manageable as observed by Ghritlaharey et al. [20] minimum anaesthesia time in a sick neonate, and provides adequate decompression and a time period to allow for weight gain and fitness for the second stage. Staged procedure obviously has got complications like stomal prolapse, stenosis, and ulceration. In our series continence was good in 5 patients and fair in 3 patients which were consistent with the observations of Gopal et al. [21].
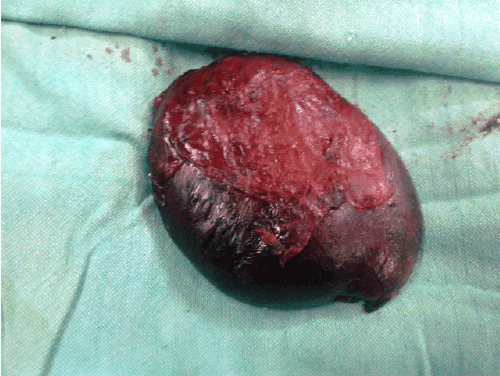
Figure 4:Excised specimen after ligation of Fistula.
We believe that there is also a role of pesticides, as most of our patients belong to the rural area where use of pesticides is common for spraying the orchards of apple. This theory has also been supported by Gupta et al. who incriminated Iodine and vitamin - B deficiency, use of pesticides and low socioeconomic status as a possible environmental factor leading to CPC [1]. The high density of cases in the northern belt of the Indian subcontinent points toward environmental factors, with deficiency of iodine or vitamin B as some of the possible factors contributing to this anomaly [1]. In north Indian belt, the land is very fertile and farming is the main occupation. Pesticides are used liberally and the population is mainly vegetarian. Also, most cases of ano-rectal anomalies belong to the low socio-economic status. All these factors suggest more of the environmental factors affecting or precipitating the anomaly. Still exact cause needs to be ascertained in a particular community like ours.
CPC is a rare entity, with a higher incidence in Northern India including our state. Adequate awareness about radiological, anatomical and histological features would lead to an exact diagnosis. Staged procedure is safe and has superior results.
Download Provisional PDF Here
Article Type: Research Article
Citation: Hamid R, Nisar B, Baba A, Gowhar M, Wani S (2015) Congenital Pouch Colon Syndrome in Jammu & Kashmir (India). Pediatr Neonatal Nurs Open Access 1(1): doi http://dx.doi.org/10.16966/2470-0983.104
Copyright: © 2015 Hamid R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access