Abstract
Diagnosing Multiple Sclerosis (MS) is challenging because of many diseases can be confused with it. MRI helps neurologists to do a right diagnosis of MS fulfilling the dissemination in space and time criteria (2017 McDonald criteria) and there must be no other explanation for the patient’s symptoms. However, with the increasing MRI availability, there has also been an increase in abnormal incidental radiological findings so close to the typical MS lesions, but these patients have no signs or symptoms of the disease; this entity is named Radiologically Isolated Syndrome (RIS). It is very important to know these RIS patients are at high risk of developing MS, although there are no approved treatments for this asymptomatic phase of the disease. Here we are reporting a clinical case showing the RIS concept and how the patient has developed a tumefactive MS in the early time course of the disease. We suggest that onset early treatment in this presymptomatic phase is a good clinical practice to prevent a new relapse of MS.
Keywords
Multiple sclerosis; Radiologically isolated syndrome; Clinically isolated syndrome; Tumectative multiple sclerosis; Disease-modifying drugs; Barkhof criteria
Introduction
Radiologically Isolated Syndrome (RIS) consists of the presence of hyperintensities on the brain of a patient observed by magnetic resonance imaging (MRI), carried out by the most recent clinical recommendations. Hyperintensities that are morphologically compatible with demyelinating lesions and fulfill Barkhof ’s criteria for anatomic dissemination, but in absence of suggestive clinical signs, are suggestive of demyelinating disease. It is well-known that a third of patients with RIS will convert to Clinically Isolated Syndrome (CIS) within 5 years of follow-up [1]. For many authors, RIS is the asymptomatic phase of multiple sclerosis (MS), where inflammatory phenomena are already present and the axonal damage characteristic of the illness begins [2,3]. It is important to identify patients at risk of conversion to CIS [1,4-6] and to consider if it is worth initiating treatment with disease modifying drugs (DMD) in this asymptomatic phase (before the first clinical event) [4,7,8] We present the case of a patient in which we observed RIS that underwent transformation into aggressive MS according to MRI criteria, with multiple tumefactive lesions.
Case Presentation
A 46 year-old woman was referred to the Unit of Neurology because of a problem with dysphagia. An abnormal brain MRI showed multiple T2 and FLAIR hyperintensities/lesions which met three of four Barkhof ’s criteria for dissemination in space (Figure 1). She had a personality disorder and had been treated in the Othorhinolaringology Service because of spasmodic dysphonia. Interrogated specifically, she had never presented suggestive clinical signs of demyelinating disease. The dysphagia improved after a change was made in the treatment of her psychiatric disorder. We decided to carry out a control MRI after 6 months, in which new lesions were not detected. Ten months after that consultation, the patient was admitted to the Mental Health Service because of a worsening of her personality disorder, with the deterioration of family dynamics, reduced impulse control and regressive behavior. The neurologic examination revealed an altered mental status, left facial palsy, hypoesthesia in her left limbs and an unstable gait. The score of the Expanded Disability Status Scale (EDSS) was 6. At this time, a new brain MRI was conducted that showed several tumefactive lesions enhanced with gadolinium (Figure 2). Cervical spinal cord MRI was normal. The visual evoked potentials (VEP) showed a bilateral demyelinating optic neuropathy and IgG oligoclonal bands were present in her cerebrospinal fluid (CSF). The patient was put on treatment with natalizumab. After 6 months of this treatment, her state of health had improved (EDDS score of 3) and a new MRI showed a reduction in the size of the lesions, which were not enhanced with gadolinium.
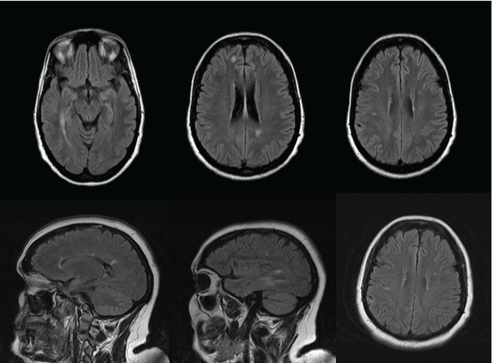
Figure 1: Brain MRI (FLAIR) shows several hyperintensity lesions that meet the dissemination in space of Barkhof criteria: 1) ≥ 9 T2 lesions that are at least 3 mm in size or ≥ 1 gadolinium-enhancing lesions, 2) ≥ 3 periventricular lesions, 3) ≥ 1 juxtacortical lesions and 4) ≥ 1 infratentorial lesions.
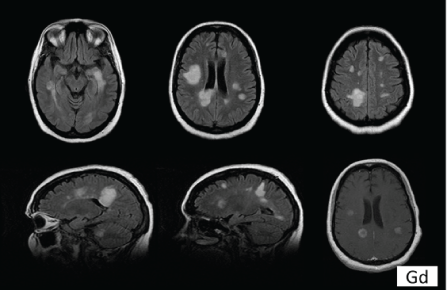
Figure 2: Brain MRI (FLAIR and Gd-T1). Multiple supra and infratentorial hyperintensity lesions, some of them showing a tumefactive appearance, the biggest ones with a diameter of 4.2 and 3.6 cm. Several lesions showed enhanced with gadolinium (Gd).
Discussion
The present case may seem merely anecdotal, but the reality is completely different from a simplistic consideration. We refer to the fact that RIS does not only imply the visualization of a brain MRI with typical lesions of demyelinating disease that meet Barkhof ’s criteria for dissemination in space. Once a demyelinating etiology of these cerebral hyperintensities was suspected, we decided to complete the patient’s evaluation with a neurophysiological examination (VEP), an immunological study of the CSF and spinal cord MRI, with the objective of identifying the presence of risk markers of conversion to CIS. At this time, we changed the scenario of the patient, from purely radiological to strictly clinical (asymptomatic multiple sclerosis). The current study is informed by a conversion risk to CIS in 33% of patients with RIS within a follow-up period of five years, with a greater risk in men, patients under the age of 37 years and with a cervical or thoracic spinal cord lesion detected by MRI[1]. Therefore, it seems reasonable to identify these patients with the purpose of avoiding or retarding this conversion, similar to what happens in CIS and subsequent conversion to clinically defined multiple sclerosis (CDMS). Studies on the natural history of MS show that this illness follows a continuum, with an asymptomatic phase represented by RIS, followed by CIS (that is to say, the first event of MS), then by a relapsing-remitting phase and finally a secondary progressive phase. The most important concept should be recognized that, in the asymptomatic phase, both inflammatory phenomena, and axonal degeneration are already present [3]. These processes are not transitory phenomena, and their deleterious effects will generate an increase in disability over the course of time. At the moment, it is helpful that axonal damage is a pathologically precocious characteristic in MS as it is the basis of neurodegeneration. This damage leads to brain atrophy in patients that present with RIS, when compared with normal subjects[2], 33% of whom demonstrate cognitive impairment which is qualitatively comparable to that present in patients with CDMS [9,10].
After carrying out the conceptual effort of putting RIS on the same level in the clinical stadium as presymptomatic MS, this is when the therapeutic dilemma arises; that is to say, treat[7] or not to treat[8] a patient with asymptomatic disease with an inflammatory-autoimmune pathophysiology and a chronic course. The administration of a drug should be informed by the norms of evidence-based medicine, considering the risks and benefits. The use of a drug outside of its clinical indications should be restricted to well-designed clinical trials, valuing as much the drug’s effectiveness as its safety. In the event of opting to treat, there are two questions: which treatment is best to administer, and, in the absence of clinical symptoms, how should the therapeutic response be monitored? In principle, and again similar to CIS and its conversion to CDMS, an option would be the employment of a DMD with the purpose of retarding conversion to CIS. From the point of view of neurodegeneration, this concept should guide the therapeutic options. In this sense, glatiramer acetate has demonstrated positive effects on brain atrophy starting from the second year of treatment; based on MR spectroscopy, an increase in the N-acetyl aspartate (NAA)/creatine ratio has been shown, suggesting a neuroregenerative effect [11]. A recent study demonstrated a significant effect of treatment with sc interferon-β1a in terms of a reduction in cortical lesions and the progression of cortical atrophy in MS, which could also be applied to RIS [12]. As an alternative, in spite of being considered a second line treatment at the moment -at least in Spain, fingolimod has been suggested as a possible drug to use in this phase due to its demonstrated effect on brain atrophy and its superiority vs. intramuscular interferon-β1a [13]. In this sense, during the 67th annual meeting of the AAN in 2015 Okuda and colleagues submitted an abstract reporting the start of a randomized doubleblinded clinical trial in RIS subjects using dimethyl fumarate as disease-modifying drug, but the results have not been reported [14]. Unfortunately, at the current moment, there are no other clinical trials or prospective studies that have approached this complex scenario to shed light on this therapeutic dilemma.
Conclusion
Here we are reporting a clinical case showing the RIS concept and how a patient has developed a tumefactive MS in the early time course of the disease. We suggest that onset early treatment in this presymptomatic phase is a good clinical practice to prevent a new relapse of MS, but the choice of treatment must be agreed with the patient who must be aware of the actual treatment expectations.
Declaration of Interest
There is no conflict of interest to declare.
Download Provisional PDF Here
Article Information
Article Type: CASE REPORT
Citation: Estévez DAG (2018) Transformation of Radiologically Isolated Syndrome into Tumefactive Multiple Sclerosis. J Neurol Neurobiol 4(2): dx.doi.org/10.16966/2379-7150.150
Copyright: © 2018 Estevez DAG. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
Received date: 11 Sep, 2018
Accepted date: 21 Sep, 2018
Published date: 27 Sep, 2018