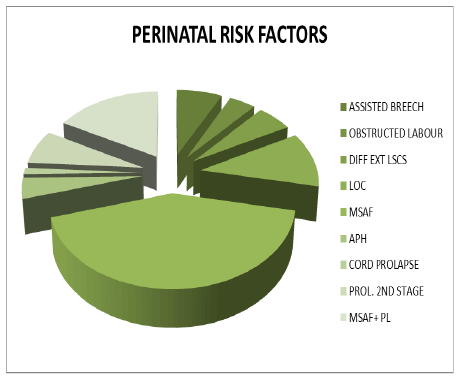
Figure 1: Perinatal risk factors for asphyxia
Swathi Chacham1,2 Jakkampudi Nagasravani* Uppin Narayan Reddy
Department of Pediatrics, Princess Esra Hospital, Owaisi Group of Hospitals, Deccan College of Medical Sciences, Hyderabad, Andhra Pradesh, India*Corresponding author: Jakkampudi Nagasravani, Department of Pediatrics, Princess Esra Hospital , Owaisi group of hospitals, Deccan college of Medical sciences, Hyderabad , Andhra Pradesh, India, Tel: +91 9908618333; E-mail: nagasravanij@gmail.com
Introduction: Perinatal asphyxia, the prime cause for neonatal mortality and morbidity, results in hypoxic damage to almost all organs of the neonate, kidneys being most frequently (50%) affected. However, neonatal renal failure poses diagnostic as well as therapeutic challenge as clinical and laboratory parameters are unpredictable in this age group. Paucity of studies determining the incidence and outcomes of renal failure in neonates with perinatal asphyxia has impelled the present study.
Objectives: To determine the incidence of acute renal failure and its predisposing risk factors in term neonates with perinatal asphyxia and to correlate its severity with hypoxic ischemic encephalopathy (HIE) staging.
Materials and methods: This Prospective case control study was conducted in a tertiary level neonatal intensive care unit, in Deccan college of Medical Sciences, Hyderabad, Telangana, India.
Inclusion and exclusion criteria: Consecutive term neonates admitted to NICU with perinatal asphyxia were enrolled as cases and 50 term neonates without it were selected as controls, after obtaining written informed consent. Neonates with major congenital anomalies were excluded.
Methods: Relevant perinatal history and examination findings along with biochemical results were recorded in a predesigned, pretested performa and the asphyxiated neonates were managed as per the standard guidelines.
Results: There were total 75 cases with perinatal asphyxia and 50 non asphyxiated controls (1.5:1 ratio). Over all incidence of perinatal asphyxia was 7%. Baseline variables were similar among cases and controls. Extramural neonates had significantly higher incidence of perinatal asphyxia (P=0.001). Overall, 60% of asphyxiated neonates had HIE stage II and 17.3% had stage III. Acute renal failure was evident in 70.7% of cases and 2% of controls, with statistical significance. Commonest risk factors for perinatal asphyxia were MSAF (40%) and positive sepsis screen (64%). While, 64% (48/75) of cases had screen positive sepsis, it was seen only in 18% (9/50) of controls, with statistical significance (P value: 0.001). 40% (30/75) of the cases had shock and 18.7% (14/75) of cases required ventilator support. Thus, shock and respiratory failure were significantly more common in asphyxiated neonates than those without asphyxia. Incidence of renal failure amongst the cases with perinatal asphyxia was 70.67% (53/75) and 2% (1/50) in controls, with statistical significance(X 2 -57.65, p value of 0.001) and 90.5% (48/53) of cases had pre renal azotemia, and non oliguric renal failure was seen in 73.5% of cases. Thus, the predominant type of renal failure among the cases was pre renal, non oliguric and intrinsic renal failure was evident in 9.4% of cases. 86.7% of Neonates with HIE II and100% of those with stage III had acute renal failure. Neonates with HIE stage II and III were more prone for shock and respiratory failure (multi organ dysfunction) and in turn had higher incidence of renal failure.
Conclusions: Incidence of renal failure was significantly high among the cases than controls and neonates with severe perinatal asphyxia had more frequent acute kidney injury. The predominant type of renal involvement was pre renal and non oliguric. Neonates with HIE stage II & III had significantly higher incidence of acute renal failure. Screen positive sepsis, shock and respiratory failure were major correlates of renal failure in asphyxiated neonates.
Perinatal asphyxia; Acute renal failure; HIE staging; Pre renal, Oliguria; Shock
ARF: Acute Renal Failure; ACOG: American Congress of Obstetricians and Gynecologists; APH: Ante Partum Hemorrhage; AAP: American Academy of Pediatrics; HIE: Hypoxic Ischemic Encephalopathy; WHO: World Health Organization; MSAF: Meconium Stained Amniotic Fluid
Perinatal asphyxia is the key factor attributable to neonatal mortality and long term neurological morbidity with an estimated incidence of 1-10 /1000 live births [1].
It results in hypoxic damage to almost all organs of the neonate, with kidneys being most frequently (40%) involved [1]. The neonatal kidney is anatomically, functionally immature and has been described as ‘halfway to acute renal failure’. Renal insufficiency manifests as early as 24 hrs of life leading to irreversible cortical necrosis when prolonged. Hence, timely detection of renal failure is vital in neonates with hypoxic ischemic encephalopathy (HIE) to sustain a stable biochemical milieu and initiate apt treatment [2]. However, neonatal renal failure poses diagnostic as well as therapeutic challenge, as clinical and laboratory parameters are unpredictable in this age group. Paucity of studies determining the incidence and outcomes of renal failure in neonates with perinatal asphyxia has impelled the present study. As per the latest estimates by World Health Organization (WHO), 4 million babies die each year before they reach the age of one month out of which ninety-eight percent of these neonatal deaths take place in the developing countries. Perinatal asphyxia and birth injuries together contribute to almost 29% of these deaths. WHO defined birth asphyxia as “failure to initiate and sustain breathing at birth” with Apgar score of <7 at one minute of life [3,4] . Similarly, ACOG & AAP have laid down essential criteria to diagnose perinatal asphyxia. These include, prolonged metabolic or mixed acedemia (pH <7.0 on cord arterial blood sample), persistence of an Apgar score of <3 for 5 min or longer, clinical neurologic manifestation as seizures, hypotonia, coma or HIE in the immediate neonatal period coupled with multiorgan dysfunction [1,5]. Antepartum or intrapartum asphyxia due to placental insufficiency contributes to ninety percent of perinatal asphyxia. The rest of the 10% results from post-partum events, secondary to pulmonary, and cardiovascular or neurologic insufficiency.
To determine the incidence of perinatal asphyxia, to evaluate acute renal failure and predisposing risk factors for it, in term asphyxiated neonates along with correlating the severity of renal failure with hypoxic ischemic encephalopathy (HIE) staging.
This Prospective case control study was conducted in a tertiary level neonatal intensive care unit, from Jul 2012 – Jul 2013, over a period of one year in Princess Esra hospital and Owaisi Group of Hospitals, Deccan college of Medical Sciences, (DCMS) Hyderabad, Telangana, India.
Consecutive term (38-42 weeks) neonates with perinatal asphyxia were enrolled as cases and simultaneously 50 term (38-42 weeks) neonates admitted to NICU for other morbidities (without any risk factors for perinatal asphyxia) were selected as controls after obtaining written informed consent (study population included both inborn as well as out born neonates).
Neonates with confounding factors believed to alter renal function such as septicemia, respiratory distress syndrome, necrotizing enterocolitis, major congenital anomalies and administration of nephrotoxic drugs either to baby or to mother were excluded from the study. Gestational age, birth weight, relevant perinatal history, examination findings and urine output were recorded in a predesigned, pretested proforma. The post asphyxiated neonates were managed as per the standard guidelines and monitored for derangements in the clinical, metabolic and hemodynamic milieu so as to intervene promptly. Neurological status was assessed using Sarnat and Sarnat staging, every 12th hour and seizures were treated with supportive care and anti epileptics. At 72 hours of life blood urea (Berthelot method), serum creatinine (Jaffe’s test) and serum electrolytes (Calorimetric method) were analysed.
Acute renal failure in neonates is diagnosed as oliguria <0.5 ml/kg/hr or serum creatinine of >2 SD above mean value for gestational age (>1.19 mg/dl).
75 cases and 50 controls were enrolled.
From the institute’s Ethics committee (Dissertation regd No: M120902113).
Results on continuous variables were presented as Mean ± SD (MinMax) and results on categorical variables were presented as percentage (%). Significance is assessed at 5% level of significance. Student T test (two tailed, independent), mann-whitney test has been used to find the significance of study parameters on continuous scale between two groups, Chi-square/Fisher Exact test has been used to find the significance of study parameters on categorical scale between two or more groups and P value <0.05 has been taken as significant. Statistical analysis was performed using SPSS version 20.
There were a total of 75 cases and 50 controls with case control ratio of 1.5:1. Incidence of perinatal asphyxia was 7%. Baseline variables like gestational age, birth weight and the gender preponderance were equally distributed among the two groups. Extramural neonates had significantly higher incidence of perinatal asphyxia (70.7%:53/75, P: 0.001) than intra mural neonates (29.3%:22/75). Commonest risk factors for perinatal asphyxia were Meconium stained amniotic fluid (MSAF) (40%) and positive sepsis screen (64%).While, 64%( 48/75) of cases had screen positive sepsis, only 18% (9/50) of controls had it, with statistical significance (P value :0.001) as shown in figure 1. Similarly, 18.7% (14/75) of cases required ventilator support, while none of the controls needed it. Likewise, 40% (30/75) of the cases had shock and none of the controls manifested it. 71.4% (10/14) of ventilated neonates had shock with statistical significance (P: 0.01). Majority of the cases who required respiratory support also had shock. Hence, there was a positive correlation between respiratory support and shock as shown in table 1. Incidence of renal failure amongst the cases with perinatal asphyxia was 70.67% (53/ 75) and 2% (1/50) in controls, with statistical significance (X2 -57.65, p value of 0.001). Likewise, 90.5% (48/53) of cases had pre renal azotemia and only 9.4% (5/53) of the cases had intrinsic renal failure. Thus the incidence of intrinsic renal failure in the present study was 9.4% and none of the controls had it. Among 53 cases with acute renal failure (ARF), 73.5% (39/53) had non oliguric ARF and 26.4% (14/53) had oliguric type. Thus, non oliguric azotemia was the predominant type of renal failure among the cases, with statistical significance (P value of 0.001) (pierson chi square coefficient =11.6). None of the controls had oliguric renal failure. Even though, 26.4% of cases had oliguric renal failure only 9.4% had intrinsic renal failure. Thus, unmonitored oliguric renal failure can lead to intrinsic renal failure, necessitating prompt recognition and intervention of oliguric renal failure to prevent its progression to intrinsic renal failure. 40% (2/5) of those with intrinsic renal failure required peritoneal dialysis. Among the 30 cases with shock 26 had ARF (86.6%) (X2 :6.2, p: 0.01) as shown in figure 2. Also, 4% (3/75) of the cases with ARF in this study had antepartum hemorrhage while none of the controls had.

Figure 1: Perinatal risk factors for asphyxia
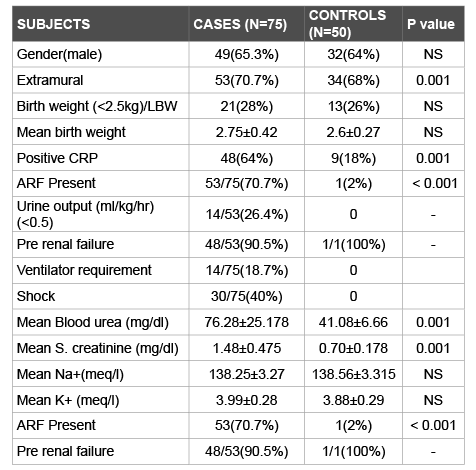
Table 1: Comparision of baseline variables and perinatal risk factors among cases and controls
Among cases Mean blood urea was 76.28 ± 0.25 with SEM: 2.91. Mean blood urea in controls was 41.08 ± 6.7 with SEM: 0.94.Similarly, mean serum creatinine levels were higher (1.48 ± 0.48; SEM: 0.1) in cases than controls (0.7 ± 0.18; SEM: 0.25) with statistical significance. Electrolytes were equally distributed in both the groups.
22.7% (17/75) of cases had HIE-I, 60% (45/75) had HIE-II and 17.3% (13/75) had HIE stage III. 29.4% (5/17) of neonates with HIE stage - I, 33.3% (15/45) of neonates with HIE stage-II and 76.9% of HIE-III had shock. Thus, neonates with HIE-I had significantly (p value of 0.01) less incidence of shock than those with higher stages. Similarly, 6% of Neonates with HIE- I, 86.7% of HIE stage-II and 100% of neonates with HIE stage-III had ARF with P value of 0.001as shown in figures 3-5. Hence, higher sarnat and sarnat stages (severe HIE) were associated with significantly higher incidence of renal failure. Important determinants of acute renal failure were perinatal asphyxia, positive sepsis screen, shock and respiratory failure, requiring ventilator support.
Mortality: 5% (4/75) cases died and all of them were of HIE stage III, with intrinsic renal failure. Two of them required peritoneal dialysis. None of the neonates with pre renal azotemia and non oliguric renal failure died. Likewise, none of the controls died.
Perinatal asphyxia is an insult during the intrauterine or immediate extrauterine period to the fetus or the newborn due to hypoxic and/or ischemic damage to various organs of greater magnitude which leads to transitory or permanent functional and biochemical changes.
Though, hypoxia and ischemia can result in impairment of every tissue and organ of the body, kidneys are extremely sensitive to oxygen deprivation. Hence, renal insufficiency can manifest within 24 hours of a hypoxic ischemic episode, and if prolonged, may even lead to irreversible cortical necrosis [1].
The present prospective, observational, case control study was conducted to determine the incidence of perinatal asphyxia and renal failure. It also evaluated the type of ARF in asphyxia and correlated the severity of renal failure with HIE staging. The results obtained in this study were compared with previous studies [2-6]. Incidence of perinatal asphyxia was 7% in the present study which was similar to previous studies [1,2]. Among the total 75 cases and 50 controls, baseline variables like gestational age, birth weight and the gender preponderance were similar. Other studies used varying proportion of cases and controls [2,7-11].
Extramural neonates had significantly higher incidence of perinatal asphyxia - 70.7% (53/75) (P=0.001) than intra mural neonates (29.3%:22/75) in this study. This can be explained by the fact that better foetal monitoring in intramural pregnancies might have lead to lesser incidence of perinatal asphyxia in them. Commonest perinatal risk factor was MSAF (40%). MSAF indicates intrauterine asphyxia and thus a well known risk factor for perinatal asphyxia [2]. The principal determinants of acute renal failure in neonates include either absolute or relative fall in effective circulating blood volume. Perinatal asphyxia is the prime etiological factor for acute renal injury, which was established in the present study as 70.7% of cases had ARF and only 2% of the controls manifested it (p-value:0.000). Also, significantly higher proportion of cases (64%) had positive sepsis screen when compared to controls (18%). Chorioamnionitis can lead to in utero asphyxia and its long term consequences while, postnatal sepsis with meningitis can result in postnatal hypoxic insult to the brain. 4% (3/75) of the cases with ARF had antepartum hemorrhage while none of the controls had and it was not seen in neonates with perinatal asphyxia who did not develop ARF. Thus ante partum hemorrhage could be a contributing factor to ARF [1,5,6].
Over all 30 neonates (40%) had shock. Among these 30 neonates, 26 (to 86.67%) had acute renal failure and there was a statistical significance (x2 =6.175; P=0.011). Also none of the controls had shock. Thus, the present study ascertains the shock as a key determinant to acute kidney injury, more so among asphyxiated neonates. None of the cases and controls received nephrotoxic drugs and did not require cardiac surgery which could also contribute to acute kidney injury. 3 cases (4%) had congestive cardiac failure and had ARF, while none of the controls had it. Cause for pre renal ARF among one control (2%) was dehydration due to inadequate feeding. None of the controls had intrinsic renal failure. 18.7% of cases required ventilator support, while none of the controls needed it. Hentschel et al. described pre renal factors, dehydration and cardiac surgery as important determinants for renal insufficiency in neonates [6]. The results of our study are comparable to the reports by Jayashree et al. [7] who also studied 12 asphyxiated neonates who had shock and 8 (66.6%) of them had ARF showing that the incidence of ARF in patients with shock was significantly higher (P<0.05). Our study shows that, out of 30 neonates with shock, 26 (86.6%) had ARF and among them 2 (7.6%) did not respond to fluid challenge, inotropes and had intrinsic renal failure. The remaining 24 (92.3%) had pre renal failure and had 100% recovery. Thus, early recognition of neonates with shock and appropriate fluid management can definitely help in the management of pre renal ARF and prevent the subsequent complications and development of intrinsic renal failure [5,6]. Stapleton et al. reported the incidence of oliguric renal failure of 8% [5]. 71.4% (10/14) of ventilated neonates had shock with statistical significance (p: 0.01). Thus, majority of the cases who had respiratory failure requiring ventilator support also had shock, ascertaining the positive correlation between respiratory support and shock[5,6]. This fact can be explained as severe perinatal asphyxia is often associated with multiorgan dysfunction leading to respiratory failure and tissue Hypoperfusion [5-7]. Oliguric renal failure manifested in cases with perinatal asphyxia and was absent among controls. (P value of 0.001) (Pierson chi square =11.6)
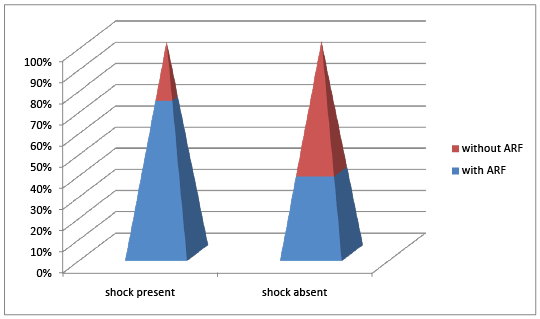
Figure 2: Correlation between Shock and Arf
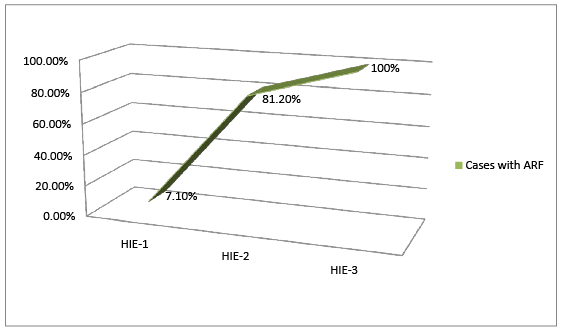
Figure 3: Correlation Between Hie Staging and Arf
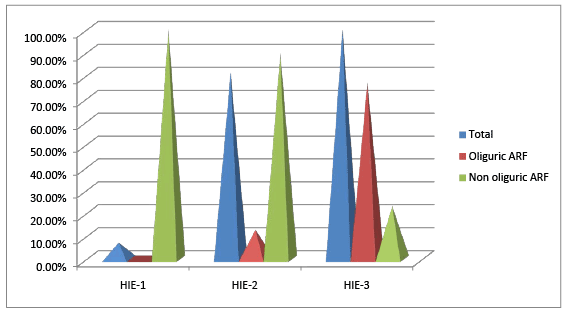
Figure 4: Correlation among types of Arf and Hie staging
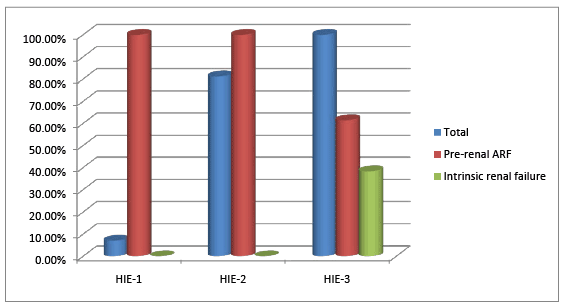
Figure 5: Correlation of severity of renal failure and asphyxia
Incidence of ARF amongst the cases was 70.67% and 2% in controls, with statistical significance 90.5% had pre renal azotemia. Among 53 neonates with ARF, 26.4% had oliguric renal failure and 73.5% had non oliguric ARF. Thus, non oliguric azotemia was the predominant type of renal failure among the cases. None of the controls had oliguric renal failure and intrinsic renal failure. Incidence of intrinsic renal failure was 9.4%, and 40% of those with intrinsic renal failure required peritoneal dialysis. Oliguric renal failure, when progressed lead to intrinsic renal failure.
Perinatal asphyxia leads to acute renal failure by two means. First pre renal impairment due to hypotension and tissue hypoperfusion leading to reduced circulating blood volume. Secondly, prolonged ischemia to the renal tubules results in acute tubular necrosis and intrinsic renal failure [5,6].
The incidence of ARF is similar to that of Pammi et al. [9] and close to Girish et al. [8] and Karlowicz et al. [10]. Others had lower incidence. Girish et al. had higher incidence of ARF in both stage – I & II than the present study. Predominant type of ARF was non oliguric and findings reported by Gupta et al. [2] were closer to this study. All the other studies had non oliguric ARF except Jayashree et al. [7]. Biochemical parameters of the cases were more comparable to Jayashree et al. in the present study, while those of controls were similar to all other studies [9-11]. The mean blood urea levels among the cases and controls in our study showed that it was significantly higher in cases as compared to controls. The results of our study are comparable to previous studies [2,7,8,11]. Serum electrolytes were similarly distributed between two groups in the present study and this was comparable to previous studies. There were three neonates with hyperkalemia and none had sodium abnormalities.
Reported incidence of ARF varies from 43%- 72% in asphyxiated neonates and non oliguric renal failure was more prevalent as in this study. The incidence reported by pammi et al. was similar to the present study while others had lesser incidence due to difference in methodology. In the present study all asphyxiated neonates with features of HIE in all 3 stages were studied, second the criteria adopted for defining acute renal failure in neonates is oliguria <0.5 ml/kg/hr or serum creatinine of more than 2 SD above of mean value for gestational age [1] which is more than 1.19 mg/dl, which no other previous studies had used, which helped in the management of the neonates at the early stages where the neonates had pre renal failure and responded well to the fluid challenge and had 100% recovery highlighting that in HIE, kidney despite being the best oxygenated organ, it is the most susceptible to ischemic–hypoxic injury because of redistribution of the blood flow to other vital organs and unique vascular supply of renal medulla and results in transient loss of renal concentrating capacity. More prolonged injury produces wide spread tubular dysfunction which may progress to intrinsic renal failure. The asphyxiated neonates have to be picked up when they are in stage of pre renal failure and managed with adequate fluids, so that they don’t progress to intrinsic renal failure as they have high mortality.
In par with previous studies, non-oliguric renal failure was more prevalent, this can be explained as due to decreased pituitary release of vasopressin or renal responsiveness to it and heterogeneous response and variable damage to the tubular epithelium that results in single nephron GFR and decrease in fractional re-absorption from tubules. This ascertains that non oliguric ARF is a major accompaniment of perinatal asphyxia and needs to be looked for.
In the current study, severe HIE / higher sarnat-sarnat stages were associated with higher incidence of ARF, and also intrinsic renal failure. These findings were comparable to those of Girish et al. [8].
In this study, mortality was 5%, which was less than previous studies. As neonates with oliguric ARF were managed aggressively, the mortality was less. None of the neonates with pre renal azotemia, non oliguric renal failure died similarly, none of the controls died. Pammi et al. [9] reported a mortality of was 36.1%, as most of them had associated morbidity and non oliguric ARF. Gupta et al. [2] found 14.1% mortality while Jayashree et al. [7] published a very high mortality of 61.5% and the deaths were attributed to HIE, shock and renal failure.
In our study mortality was less than previous studies (5.3%), all cases intrinsic renal failure i.e. total of 5 cases did not improve with fluid therapy. Four neonates died among them, 2 inspite of peritoneal dialysis died as they had associated co morbid condition, shock, MODS.
The asphyxiated neonates have to be screened for ARF at the earliest so that they can be managed at the pre renal failure stage only, without letting them to progress to intrinsic renal failure as they have high mortality.
In this prospective case control study the case-control ratio was 1.5:1 and baseline variables were similar among 75 cases and 50 controls. Incidence of perinatal asphyxia was 7%.
Extramural neonates, MSAF, screen positive sepsis, APH were major risk factors for perinatal asphyxia. Respiratory failure, shock, higher HIE stages were significant determinants of ARF in asphyxiated neonates. Incidence of renal failure amongst was 70.67% and 2%in cases and controls respectively. The predominant type of ARF was non oliguric ARF and HIE stage II & III, coupled with oliguric ARF were major determinants to intrinsic ARF, which was 9.4%. Oliguric renal failure and intrinsic renal failure manifested only in cases with perinatal asphyxia and was absent among controls. Among cases Mean blood urea, mean serum creatinine levels were higher among cases than controls. Incidence of mortality was 5% with 100% of them being HIE III and intrinsic renal failure. As non-oliguric renal failure is predominant, in neonates with perinatal asphyxia, urine output monitoring alone is not sufficient and mandates biochemical monitoring. Screen positive sepsis, shock and respiratory failure were major correlates of renal failure in asphyxiated neonates. In neonates with HIE prevention of intrinsic renal failure is vital as it is associated with MODS and has higher morbidity and mortality, as seen in the present study.
We thank Dr. Syed Yadullah Mehdi for helping with manuscript editing.
Authors declare no conflicts of interest regarding this article.
Download Provisional PDF Here
Aritcle Type: Research Article
Citation: Chacham S, Nagasravani J, Reddy UN (2016) Acute Renal Failure in Neonates with Perinatal Asphyxia and its Correlation with HIE Staging: A Prospective Case Control Study. J Neurol Neurobiol 2(2): doi http://dx.doi. org/10.16966/2379-7150.119
Copyright: © 2016 Chacham S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access