Introduction
Previous studies have reported that 25% to 40% of surviving patients
with kidney graft failure eventually undergo allograft nephrectomy [1-6].
Indications for allograft nephrectomy include symptoms attributable to
the failed allograft (including pain, swelling or localized tenderness, fever,
hematuria or bleeding), thrombosis, infection, anemia with erythropoietin
resistance, failure to thrive and graft intolerance syndrome, which occurs
secondary to rejection and/or allograft ischemia. In other instances,
allograft nephrectomy may be performed for malignancy, as treatment
for diseases such as polyomavirus nephropathy or post-transplant
lymphoproliferative disease, or for space considerations in planning of a
subsequent kidney transplant. The rate of allograft nephrectomy in patients
with graft failure varies widely from 0.5-43% according to individual
center policies [3-9]. Although the role of allograft nephrectomy in the
management of kidney transplant recipients with graft failure remains
controversial, most clinicians agree that the presence of malignancy in the
allograft is usually a robust indication for nephrectomy coincident with a
requisite withdrawal of immunosuppression.
The aging donor and recipient populations have led to new challenges
in kidney transplantation. Current data demonstrate an increasing
proportion of elderly patients in an already rising end stage renal disease
(ESRD) population [10-13]. Both aging and chronic immunosuppression
are associated with an increased risk of malignancy [10-13].Renal cell
carcinoma (RCC) carries a higher prevalence in older individuals, in
patients receiving dialysis and in those with a kidney transplant compared
to the general population [10-12,14,15]. In addition, the overall risk of
malignancy in kidney transplant recipients ranges from 4 to 30-fold higher
depending on the type of malignancy analyzed [10-12,14,15]. Surprisingly,
there are few reports of allograft nephrectomy for malignancy in the
literature other than isolated case studies. The purpose of this study was to
review our overall experience with allograft nephrectomy for malignancy
at a single center including the use of pre-operative angiographic
embolization of the allograft as a bridge to planned nephrectomy to
reduce blood loss and prevent tumor dissemination.
Methods and Results
Over a 13 year period from 2002 to 2015, we retrospectively reviewed
indications for allograft nephrectomy in 74 consecutive cases. A total of
7 patients (9.5%) underwent nephrectomy for allograft malignancy. A
summary of case studies follows.
Case 1
A 39 year old Caucasian male with ESRD secondary to focal segmental
glomerulosclerosis-collapsing variant and diffuse nodular diabetic
glomerulosclerosis underwent living related donor kidney transplant
from his 44 year old human leukocyte antigen (HLA)-identical sister
in 1998. In 2010, this transplant failed secondary to chronic allograft
nephropathy and the patient started dialysis. As part of a retransplant
screening evaluation, the patient underwent a renal ultrasonography,
which demonstrated a 1.5 by 1.3 cm solid tumor in the allograft.
Subsequent ultrasound guided fine-need aspiration biopsy and cytology
demonstrated a papillary type I RCC, Fuhrman nuclear grade 3, 1.3 ×
1.1 × 1.0 cm tumor, which was well-circumscribed and confined to the
kidney (Figure 1A). Following angiographic embolization of the allograft,
the patient underwent an uncomplicated radical transplant nephrectomy.
Following a period of recovery and in the presence of a negative workup
for residual or metastatic disease, the patient underwent successful
living unrelated donor kidney retransplantation from a 25 year old donor
in August 2010 without being subjected to a mandatory waiting period
or disease-free interval because of the favorable histopathology and size
of the tumor. The second donor was a zero HLA-match and the patient
received alemtuzumab induction therapy. At nearly 5 years follow-up, the
patient continues to exhibit normal renal function (serum creatinine level
1.2 mg/dl and estimated glomerular filtration rate [GFR] >60 ml/min)
without any evidence of disease on an immunosuppressive maintenance
regimen of tacrolimus, mycophenolate and prednisone.
Case 2
A 31 year old Caucasian male with ESRD secondary to type 1 diabetes
mellitus underwent living related donor kidney transplant from his sister
in 1991; this kidney functioned for 11 years before failing secondary to
recurrent diabetic nephropathy. He was on dialysis for 3 months before
undergoing simultaneous kidney-pancreas transplantation from a 19 year
old male deceased donor in May, 2002. The second donor was a zero HLAmatch;
hence, the patient received rabbit anti-thymocyte globulin (rATG)
induction in combination with tacrolimus and mycophenolate. During
an evaluation for a ventral incisional hernia in 2011, a computerized
tomographic (CT) scan revealed an incidental 2.3 cm solid mass in the
failed left lower quadrant living donor kidney transplant, consistent with
the diagnosis of RCC (Figure 1B). Following angiographic embolization
of the failed allograft, the patient underwent uncomplicated radical
transplant nephrectomy in June2011. Final pathology demonstrated type
I papillary RCC, Fuhrman grade 2, with clear margins. Moreover, an
additional mass was identified on this allograft nephrectomy specimen- a
well circumscribed tumor with tubule-acinar architecture, most consistent
with acquired cystic disease-associated RCC. Thirteen years following
his second transplant and nearly four years following nephrectomy, the
patient continues to do well with a serum creatinine level of 0.9 mg/dl
and an estimated GFR of >60 ml/min. He also remains insulin-free and
disease-free. His current surveillance regimen consists of yearly CT
imaging.
Case 3
A 63 year old Caucasian male with ESRD secondary to lupus nephritis
was on peritoneal dialysis for nine months before receiving an ipsilateral
dual kidney transplant from a standard criteria donation after cardiac
death (DCD) donor in August 2011. The donor was a 49 year old white
male with a history of smoking, hypertension, weakness and unexplained
weight loss. The kidneys were considered for dual transplantation because
of the requisite warm ischemia associated with the DCD process in concert
with the kidney biopsy, which demonstrated 18% glomerulosclerosis
with mild vascular changes. Both kidneys appeared anatomically normal
except for atherosclerosis extending into the renal arteries. In May of
2012 (nine months from the index transplant), the patient developed
right lower quadrant fullness and pain in the setting of an elevated serum
creatinine level. A non-contrast abdominal and pelvic CT scan revealed
enlarged and edematous allograft kidneys with stranding in adjacent
soft tissues and a small amount of ill-defined perinephric fluid. A renal
transplant biopsy discovered a high-grade invasive urothelial carcinoma
with extensive squamous differentiation. Subsequent contrast-enhanced
CT scan showed extensive pelvic and retroperitoneal lymphadenopathy
with possible spread to the mediastinum, consistent with metastatic
urothelial carcinoma (Figure 2A). Cystoscopy of the bladder and the
native ureters showed no evidence of urothelial carcinoma. Positron
emission tomographic (PET) scan demonstrated local and metastatic
disease (Figure 3A). Following angiographic embolization of both
kidneys, the patient underwent attempted radical dual allograft nephroureterectomy,
which was complicated by thick scar tissue and an extensive
burden of extra-renal tumor that was not completely excised because it
was encasing the iliac vessels and extremely adherent to surrounding vital
structures. Final pathology revealed high-grade urothelial carcinoma
with sarcomatoid features and lymphovascular invasion with tumor at
the margins of resection and satellite lesions in the renal parenchyma.
Interestingly, histocompatibility typing of the tumor demonstrated both
donor and recipient elements. Following cessation of immunosuppression,
the patient received chemotherapy with paclitaxel for 6 months with
disease resolution. At 3 years follow-up, the patient is alive and doing well
on home hemodialysis four times per week with no evidence of recurrent
or metastatic disease (Figure 3B). Surveillance monitoring includes CT
imaging every 6 months.
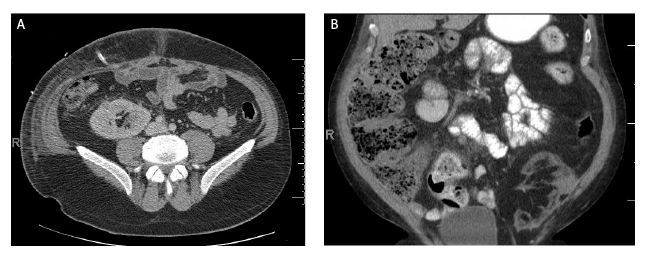
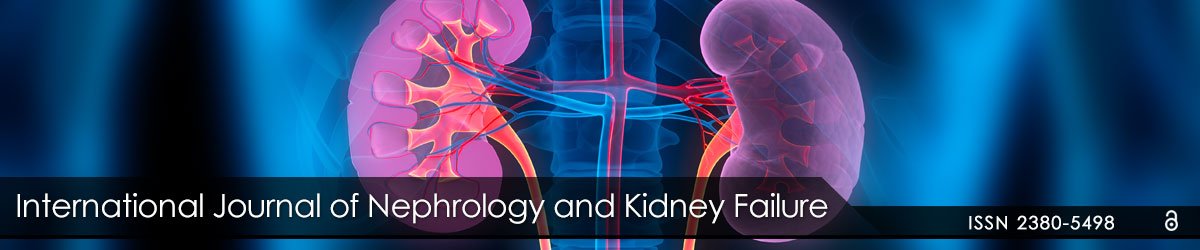
Figure 1: Small Renal Masses in Kidney Allografts
Figure 1A: (Case 1): 39 year old male with small renal mass found originally on ultrasound. CT scan demonstrated 1.5 cm x 1.3 cm solid tumor in
transplant allograft (arrow). Biopsy demonstrated RCC. Final pathology type 1 RCC, Fuhrman Grade 3.
Figure 1B: (Case 2): 31 year old male with a small renal mass found on CT scan. A 2.3 cm renal mass concerning for RCC in a failed left lower
quadrant living donor allograft (arrow). Final pathology type 1 RCC, Fuhrman grade 2.

Figure 2: Transitional Cell Carcinoma (TCC) in Kidney Allografts
Figure 2A: (Case 3): 63 year old male with ipsilateral dual kidney transplant underwent a renal transplant biopsy 9 months post-transplant for
possible rejection and high grade TCC was discovered. A subsequent contrast enhanced CT demonstrated metastatic urothelial carcinoma (arrow).
Figure 2B: (Case 7): 67 year old female who developed elevated creatinine following transplant. Non-contrast CT scan demonstrated ureteropelvic
junction obstruction and high density material within renal pelvis (arrow). Antegrade nephrostogram demonstrated concern for TCC (insert).
Cases 4 and 5
The next two cases involve donor-derived malignancies (myeloid
sarcoma or acute myeloid leukemia) in kidney recipients from the
same donor. The donor was a 38 year old female nursing home resident
whosustained brain death secondary to an intracerebralhemorrhage.
Her history was negative for any cancer or unintended weight loss.
Furthermore, her complete blood cell count performed at the time
of admission for brain hemorrhage did not reveal any significant
abnormalities and her peripheral blood smear did not have any evidence
for peripheral blasts. A preimplantation kidney biopsy revealed changes
consistent with long-standing diabetes mellitus.
Recipient 1: A 72 year old Caucasian male with a history of ESRD
secondary to long-standing type 2 diabetes mellitus and hypertension
wason hemodialysis for two years before undergoing uncomplicated single
kidney transplantation in October 2012. His past surgical history was
significant for thyroidectomy for papillary adenocarcinoma and radical
prostatectomy with pelvic lymphadenectomy for prostate cancer. The
recipient and donor were a two-HLA mismatch. He received alemtuzumab
induction in combination with tacrolimus and mycophenolate and
experienced slow graft function with a serum creatinine level nadir of 2.4
mg/dl. The patient’s serum creatinine level rose to 4.5 mg/dl 4 months after
transplant and renal ultrasonography and CT scan (Figure 4B) showed a
significant increase in the volume of the transplanted kidney and elevated
resistive indices. A subsequent renal allograft biopsy showed diffuse
parenchymal infiltration with immature mononuclear cells positive on
immunohistochemistry for CD34, CD117 and myeloperoxidase positive
blasts consistent with a diagnosis of myeloid sarcoma. Furthermore,
fluorescence in situ hybridization studies showed normal chromosomes
and confirmed 93% of the cells in the biopsy to be of donor origin (female,
XX) suggesting a donor-derived myeloid sarcoma transmitted with the
transplanted kidney. Therecipient’s bone marrow biopsy was negative
for leukemic involvement and a metaphase cytogenetic analysis revealed
a normal male karyotype with no apparent leukemic involvement. PET
scan did not show any foci of involvement beyond the renal allograft.
Following angiographic embolization of the allograft, the patient
underwent an uneventful nephrectomy and completed chemotherapy
with cytarabine and daunorubicin in accordance with the HematologyOncology
recommendations. A bone marrow biopsy and repeat PET
scanper formed five months following the initial diagnosis did not show
any evidence of disease. He resumed hemodialysis and remained in
remission until his death secondary to a cardiovascular event13 months
following nephrectomy.
Recipient 2: A 77 year old Caucasian female with a history of ESRD
secondary to interstitial nephritis was on hemodialysis for 2 years and
had a history of a prior failed renal transplant (at a different institution)
secondary to renal artery thrombosis resulting in immediate allograft
nephrectomy. She underwent uncomplicated kidney retransplantation
and received alemtuzumab induction in combination with tacrolimus and
mycophenolate. The recipient and donor were a three-HLA mismatch.
She experienced immediate graft function and serum creatinine levels
stabilized in the 1.4-1.7 mg/dl range. A three week allograft surveillance
biopsy demonstrated recovered acute tubular injury and donor transmitted
nodular diabetic glomerulosclerosis and hyalinosis. Four months
following transplant, she was admitted to another facility for a urinary
tract infection and acute kidney injury with a serum creatinine level of >4.0
mg/dl. A renal biopsy performed at the other institution showed acute and
chronic thrombotic microangiopathy, although on further review, atypical
cells were noted in the biopsy. At this point in time, the patient who had
received the mate kidney from this donor had been already diagnosed
with myeloid sarcoma (recipient 1). Consequently, we advised this patient
to undergo evaluation and allograft nephrectomy. Following admission to
our facility, the patient’s laboratory analysis revealed a serum creatinine
of 4.3 mg/dl, hemoglobin of 8.3 g/dl and platelet count of 109,000/μl. The
patient refused a bone marrow biopsy and no blasts were noted in her
peripheral blood smear. Additionally, imaging with PET scan did not
reveal any uptake of fluorodeoxyglucose (FDG) in locations other than
the renal allograft. Following angiographic embolization of the allograft,
the patient underwent nephrectomy and subsequent pathologic analysis
of the specimen showed a monotonous population of myeloid blasts
that were morphologically identical to the pathology noted in the first
transplant recipient.Further molecular genotyping analysis performed
on the renal allograft established myeloid sarcoma of donor origin and
identical haplotypes. The patient did not opt for systemic chemotherapy,
but remained in remission and on hemodialysis until her death secondary
to a cardiovascular event 18 months following nephrectomy.
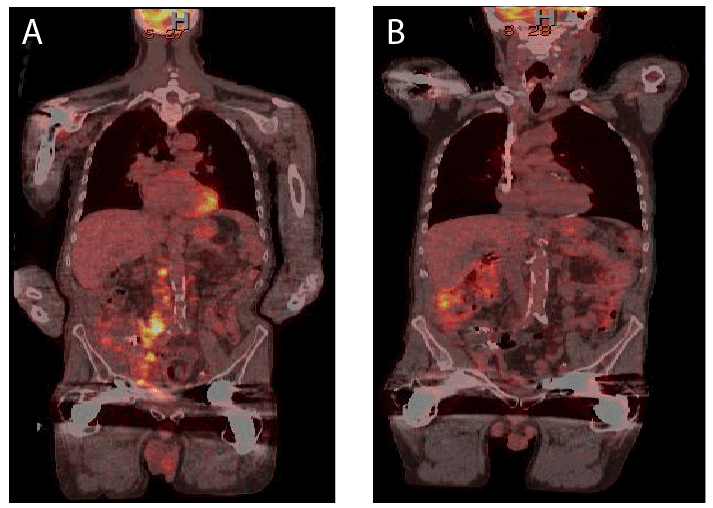
Figure 3: Transitional Cell Carcinoma (TCC) in Transplant Kidney
Figure 3: Case 3: PET scan (A) demonstrated metastatic disease. Patient underwent radical nephrectomy; intraoperatively, cancer was encasing
iliac vessels and margins of resection were positive for tumor. Following cessation of immunosuppression, the patient received chemotherapy. At 3
year follow-up, patient remained cancer free (B).
Case 6
A 74 year old African American female with a history of end-stage
renal disease secondary to glomerulonephritis underwent a5-HLA
mismatch kidney transplant from a 50 year old male DCD donor in July
2011. She had a history of a prior right laparoscopic nephrectomy for RCC
in her native kidney in 2010. She received alemtuzumab induction in
combination with tacrolimus and mycophenolate and initially experienced
delayed graft function. She subsequently did well with a serum creatinine
level nadir of 2.1 mg/dl. Fourteen months following transplantation, she
presented to the Emergency Department in September 2012 with nausea
and vomiting and had a CT scan of the abdomen and pelvis, which
showed lymphadenopathy and a 7 × 4 cm lobular soft tissue mass along
the right pelvic side wall in close proximity to an enlarged, indistinct
kidney transplant (Figure 4A). A CT scan of the chest showed multiple
pulmonary nodules bilaterally and an enlarged left lower clavicular lymph
node measuring 1.1 x 1.6 cm in size.
A biopsy of the mass was performed, which showed high grade RCC.
Following angiographic embolization of the allograft, she underwent
uncomplicated transplant nephrectomy for metastatic RCC with
immediate cessation of immunosuppression. At this point, it was unclear
whether the RCC was residual from her native kidney or if the cancer was
donor-transmitted or de novo in the allograft. Follow-up pathology of the
allograft revealed Fuhrman Grade 4 RCC withtubule-cystic features and
focal clear cell change consistent with a primary renal cancer given the
patient’s history of RCC in the native right kidney in 2010.The pathology
report concluded that the tumor was unifocal in nature with a size of 5.0
cm in the largest dimension and extension into perinephric tissue and
renal pelvic fat most likely representing a metastasis from the primary
RCC. Additionally, the margins were positive with intraluminal and soft
tissue deposits identified at the vascular margins. Following nephrectomy,
the patient resumed hemodialysis and underwent treatment with
temsirolimus. She initially did well but was never disease-free and
eventually died in hospice care 22 months later in July, 2014 secondary to
metastatic RCC.
Case 7
A 67 year old Caucasian female was on hemodialysis following bilateral
native nephrectomies in 2004 for malignancy (right kidney oncocytoma
and left kidney RCC). She underwent one HLA-match expanded criteria
(61 year old male donor) kidney transplantation in April 2005, and
experienced immediate graft function with a serum creatinine level
stabilizing in the 2.3-2.6 mg/dl range. She received rATG induction in
combination with tacrolimus, mycophenolate, and prednisone. She did
well for approximately 8.5 years until September2013, when she presented
with deteriorating renal function with a serum creatinine level of 3.9 mg/
dl noted on routine follow-up. Renal ultrasonography revealed moderate
transplant hydronephrosis and an abdominal and pelvic CT scan
confirmed transplant hydronephrosis with high density material in the
dependent renal collecting system and proximal ureter (Figure 2B). She
subsequently underwent nephrostomy tube placement for a ureteropelvic
junction (UPJ) obstruction and further imaging with a fluoroscopic
nephrostogram revealed a large, irregularly contoured filling defect in the
renal pelvis with extension into multiple infundibula and the proximal
ureter, suggesting a high likelihood of a neoplasm of urothelial origin.
Subsequently, a biopsy of the mass noted in the renal pelvis collecting
system revealed invasive urothelial malignancy consistent with transitional
cell carcinoma (TCC). The patient underwent a metastatic work-up and
cystoscopy, which did not show any evidence of disease in the bladder
or her native ureteral remnants. Subsequently, the patient underwent
angiographic embolization of the allograft followed byuneventful radical
allograft nephro-ureterectomy in October 2013. Pathology of the removed
specimen revealed invasive papillary urothelial high grade carcinoma, 3.0
cm in size in the largest dimension. The neoplasm involved the inferior
calyx and renal pelvis with focal extension into the proximal ureter, and
the margins were free of neoplasia and no lymphovascular invasion was
identified. Immunosuppression was discontinued immediately except for
prednisone. She resumed hemodialysis through her previous fistula. A
routine surveillance CT scan performed in January, 2014, did not reveal
any evidence of residual disease or recurrent tumor in the resection bed.
Her most recent imaging study in March2015 (17 months following
nephrectomy) showed no evidence of metastatic disease. Currently, she is
alive and doing well on hemodialysis.
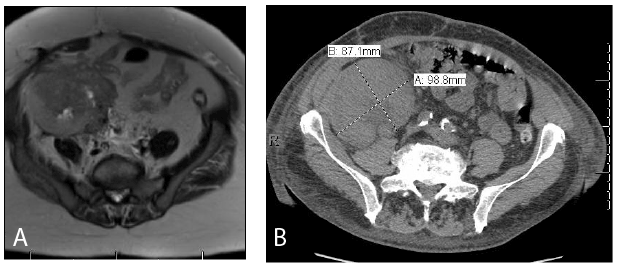
Figure 4: Large Masses in Kidney Allograft
Figure 4A: (Case 6): 74 year old female presented with elevated creatinine and found to have a enlarged allograft. Subsequent MRI demonstrated
lesion concerning for RCC (arrow). Final pathology demonstrated RCC, Fuhrman grade 4, with positive margins and extension into the surrounding
tissue.
Figure 4B: (Case 4): 72 year old male presented with elevated creatinine and had an ultrasound and biopsy. Biopsy was concerning for myeloid
sarcoma. CT scan demonstrated a large mass in the transplant kidney (arrow).
Discussion
Patients on renal replacement therapy have a higher risk of malignancy
compared to age- and gender-matched control patients in the general
population. The magnitude of the increased risk varies with the modality
of renal replacement therapy and the type of malignancy, with kidney
transplantation conferring a much greater risk of cancer compared to
patients on dialysis [8,10-13,16-18]. Overall risk of malignancy may
be as high as 15-20% at 10 years following kidney transplantation.
Certain cancers that develop in patients on dialysis or following kidney
transplantation share similar risk factors to patients in the general
population [10-12,14,15]. Alternatively, however, different rates and
patterns of site-specific cancers are observed in patients on renal
replacement therapies that are related in part to the severity and duration
of renal failure as well as the burden of immunosuppression and type of
organ transplant.
It is well established that the requisite post-transplant
immunosuppression in kidney transplant recipients contributes to their
heightened cancer risk. In particular, the suppression of CD4+ and
CD8+ T-cells, responsible for detecting and killing tumor cells and the
susceptibility to tumorigenic viral infections are hypothesized as the
main mechanisms driving malignancy following renal transplant. Kidney
transplant recipients are at risk for 3 types of malignancies; pre-existing
or recurrent tumors, de novo tumors occurring following transplantation,
and donor-derived or transmitted tumors. In one study, the average
time to cancer development following transplantation was 9.4 years,
and all-cancer rates continued to rise with increasing time following
transplantation. Conversely, in the case of occult or known donor-derived
malignancy, average time to cancer discovery was 2 months (range 2 days
to 38 months post-transplant) [12,13].
The above seven case studies are representative of the spectrum
of malignant disorders affecting the renal allograft that may result in
nephrectomy. For example, the first two case studies involve patients that
developed incidental de novo RCCs in failed living donor renal allografts
that functioned for greater than10 years. In case 1, the localized RCC was
detected during retransplant evaluation, was managed by nephrectomy
alone, and did not preclude successful living donor kidney retransplantation
performed 2 months later. In case 2, a localized RCC was detected 20 years
following primary kidney transplant and 9 years following simultaneous
kidney-pancreas transplant during work-up of a ventral incisional hernia.
Once again, the lesion was treated by nephrectomy alone although a
second localized malignancy was identified on the explant specimen. In
both of these cases, because the lesions were localized and thought to be
de novo in origin, no changes were made in immunosuppression and
both patients continue to do well with excellent allograft (retransplant)
function and exhibit no evidence of disease on surveillance imaging 4-5
years following nephrectomy of the primary transplant.
In comparison, cases 3-5 involve examples of donor-derived
malignancies. Case 3 chronicles an unusual case of high grade urothelial
neoplasia in a dual kidney transplant recipient diagnosed 9 months
following the index transplant. This patient presented with localized signs
and symptoms and was subsequently found to have a large burden of locally
invasive and metastatic disease, which was not completely resectable. The
timing of diagnosis, the absence of disease in the native urothelium and
the history of unexplained weight loss in the donor all suggest a donor
etiology. Although histocompatibility typing of the tumor demonstrated
donor and recipient elements, the tumor responded more like a donorderived
malignancy as the patient is completely free of disease and is
doing well on dialysis at 3 years follow-up following dual nephrectomy,
cessation of immunosuppression, and 6 months of paclitaxel.
In the unfortunate pair of elderly mate kidney recipients reported in
cases 4 and 5; however, both patients developed biopsy-proven myeloid
sarcoma of the allograft within a few months of transplant, which is more
characteristic of donor-transmitted disease. Both patients presented with
acute kidney injury and the diagnosis of malignancy in the allograft was
serendipitous. Whereas one patient underwent bone marrow biopsy and
received subsequent chemotherapy, the other refused both a bone marrow
biopsy and post-nephrectomy chemotherapy. Molecular genotypic testing
in both cases confirmed acute myeloid leukemia of donor origin with
identical haplotypes. Both patients died more than one year following
allograft nephrectomy of cardiovascular events but were otherwise free
of disease.
Case 6 represents an example of recurrent RCC affecting the renal
allograft in a patient who had previously undergone laparoscopic right
native radical nephrectomy for a localized 1.4 cm, Fuhrman nuclear grade
3,acquired cystic disease-associated RCC 8 months prior to transplant.
A mandatory waiting period or disease-free interval was not deemed
necessary because of the favorable histopathology and localized nature
of this tumor. Unfortunately, the patient presented 9 months following
transplant with localized and metastatic RCC involving the renal allograft
that had been performed ipsilateral to her previous native nephrectomy.
Imaging did not show any evidence for suspicious lesions in her remaining
atrophic left native kidney. Although the patient survived 22 months
following allograft nephrectomy, she was never disease-free and died in
hospice care.
The final case is an example of probable de novo high grade urothelial
carcinoma presenting 8.5 years following transplantation. Similar to case
3, this patient presented with local signs and symptoms in conjunction
with acute kidney injury. Although initial imaging studies suggested
distant disease, the margins of resection were free of disease, lymph
nodes were negative, and the patient is currently doing well at 17 months
following nephrectomy.
Conclusion
Our case reports demonstrate the myriad and incidental presentations
of malignancy in functioning and failed renal allografts (including
localized and metastatic disease) and the unpredictable timeframe of their
presentation ranging from months to years following the renal transplant.
Furthermore, these cases illustrate the range of varied pathology ranging
from genitourinary malignancies such as RCC and TCC to blood/
mesenchymal derived malignancy (i.e., the cases of myeloid sarcoma).
Although most recent literature has emphasized the role of nephronsparing
procedures in the management of allograft malignancy, the
unique aspects of these cases in the setting of chronic immunosuppression
culminated in the decision to perform allograft nephrectomy. In our
thirteen year experience, approximately 9.5% of our patients (7 out of 74)
underwent allograft nephrectomy for a malignancy-related indication. Due
to the uncommon nature of malignancy occurring following transplant,
it is important acknowledge the idiosyncratic nature of malignancies
and their varying presentations, which must be dealt with on a case by
case basis. Likewise, it is also important to appreciate the complexity of
clinical decision-making and the importance of individualizing treatment
based on recipient, donor and tumor characteristics. Additionally, the
case series highlights the importance of comprehensive donor assessment
and recipient surveillance in light of expanding donor and recipient
acceptance criteria.
Conflict of Interest:
All authors declare that they have no conflict of interest.
(In case animals were involved) Ethical approval: This article does not
contain any studies with animals performed by any of the authors.
(And/or in case humans were involved) Ethical approval: This article
does not contain any studies with human participants performed by any
of the authors.
This article does not contain any studies with human participants or
animals performed by any of the authors.
The data reported in this study was generated in accordance with
local institutional review board guidelines and approval. Finally, we
declare that this study does not represent any conflict of interest for
any of the authors, and no intramural or extramural funding sources
were involved in this study.
Author Contributions
S H: Drafting article, data collection, data analysis/interpretation,
concept/design
K W: Data analysis/interpretation, drafting article, imaging
A C F: Approval, Data collection
J R: Approval, Data collection
G O: Approval, Data Collection, Concept/Design
R J S: Critical Revision, Concept/Design, Approval, Data analysis/
interpretation, drafting article