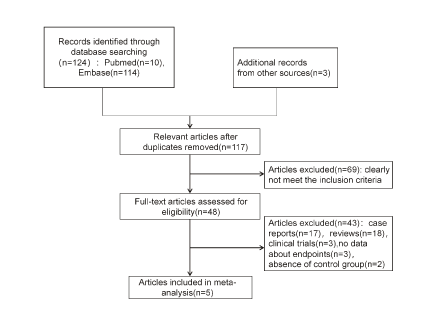
Figure 1: Flowchart diagram illustrating study selection methodology.


Bo Cao1# Xiaobo Hu1# Min Chen2 Mingfeng Shen1* Lan Xu1*
1Department of Clinical Pharmacy, Affiliated Ninth Hospital of Suzhou University, Suzhou, China*Corresponding author: Lan Xu, Department of Clinical Pharmacy, Affiliated Ninth Hospital of Suzhou University, Suzhou, China, E-mail: xulan848@126.com
Mingfeng Shen, Department of Clinical Pharmacy, Affiliated Ninth Hospital of Suzhou University, Suzhou, China, E-mail: 422644766@qq.com
Background: Evidence on the safety and effectiveness of Non-Vitamin K Antagonist Oral Anticoagulants (NOACs) in Atrial Fibrillation (AF) patients with cancer is rather limited, so we performed this meta-analysis to compare the efficacy and safety of NOACs with vitamin K antagonists (VKAs) in real-world patients with AF and cancer.
Methods: The PubMed and Embase databases were searched up to June 2020 for eligible studies. Outputs were presented as Risk Ratios (RRs) and corresponding 95% Confidence Intervals (CIs) using a random-effects model.
Results: A total of five observational studies involving 232, 234 cancer patients with AF were included. Compared with VKAs, use of NOACs was associated with decreased risks of stroke or systemic embolism (RR, 0.79; 95% CI 0.69-0.90), ischaemic stroke (RR, 0.82; 95% CI, 0.72-0.93), Venous Thromboembolism (VTE) (RR, 0.28; 95% CI 0.14-0.53), all-cause death (RR, 0.57; 95% CI 0.50-0.64), major bleeding (RR, 0.60; 95% CI 0.51-0.72) and intracranial or gastrointestinal bleeding (RR, 0.61; 95% CI, 0.51-0.73). In subgroup analysis, all NOACs showed similar rates of stroke or systemic embolism, ischaemic stroke but reduced rates of all-cause death, major bleeding and intracranial or gastrointestinal bleeding compared to VKAs.
Conclusions: In this combined analysis of real-world observational studies, NOACs showed lower risks of stroke or systemic embolism, ischaemic stroke, VTE, all-cause death and reduced rates of major bleeding and intracranial or gastrointestinal bleeding compared to VKAs in patients with AF and cancer
Atrial fibrillation; Cancer; Oral anticoagulants; Real-world; Meta-analysis
Atrial Fibrillation (AF), inducing a five-fold increase in thromboembolic risk [1], is the most common cardiac arrhythmia. AF commonly occurs in cancer patients; approximately 2.5% with preexisting AF [2] and 30% will have new-onset AF [3]. The possible mechanism for higher prevalence of AF in cancer patients may be due to a hypercoagulable state [4,5], anticancer drugs or chest surgery [6,7]. Cancer patients are at high risk of morbidity and mortality due to thrombosis and bleeding [8,9]. The risks of both thromboembolic and bleeding incidents are higher in AF patients with cancer compared to those free of cancer [10-14]. Therefore, the optimization of anticoagulation therapy is particularly important in reducing the risk of both thrombotic and bleeding complications in AF patients with cancer.
Warfarin is traditionally prescribed anticoagulant in prevention of thrombosis among AF patients. However, warfarin therapy remains challenging in AF patients with cancer because of the metabolic interactions with chemotherapy, chemotherapy-induced thrombocytopenia, and the frequent need for surgical or invasive procedures [15]. Non-vitamin K Antagonist Oral Anticoagulants (NOACs) are an alternative approach for stroke prevention in NonValvular AF (NVAF) patients. There are currently four NOACs available: factor Xa inhibitors (edoxaban, apixaban, and rivaroxaban) and direct thrombin inhibitor (dabigatran), which are considered as effective and safe as VKAs [16-18].
Although the efficacy and safety of NOACs in patients with venous thromboembolism and cancer has been well presented [19,20], data of NOACs therapy in AF patients with cancer is extremely limited. There is no established guidance for anticoagulation management of AF patients with active cancer, and little controlled data on the use of NOACs of these population is found because the active cancer is usually an exclusion criterion in randomized NOAC trials [21,22].
Therefore, data comparing the efficacy and safety of NOACs to VKAs is post hoc analysis of Randomized Clinical Trials (RCTs) [23- 25]. There were three meta-analyses assessing the performance of NOACs versus warfarin with the post hoc analysis data of RCTs and observational studies [26-28]. However, clinical trial populations are selected due to overly restrictive eligibility criteria and do not reflect all demographic features. Therefore, it’s imperative to assess the efficacy and safety profiles of NOACs in real-world settings from observational studies. In this context, we performed an updated meta-analysis in observational studies of real-world representative populations to compare the efficacy and safety of NOACs with VKAs in NVAF patients with cancer.
The current analysis was performed according to the Meta-Analysis of Observational Studies in Epidemiology (MOOSE) [29]. There was no need to provide the ethical approval because all studies included for meta-analysis have been published already
We performed a systematic search in PubMed and Embase electronic database until June 2020 for relevant studies comparing the effect of any NOAC (apixaban, dabigatran, edoxaban or rivaroxaban) versus VKA in AF patients with cancer. The searching strategy was conducted by combining four kinds of search terms-1) “Atrial fibrillation” or “AF” and 2) “cancer” or “carcinoma” and 3) vitamin K antagonists or warfarin and 4) “new oral anticoagulants” or “non vitamin k antagonist oral anticoagulants” or “rivaroxaban” or “apixaban” or “edoxaban” or “dabigatran” or oral factor “Xa inhibitors” or “oral factor IIa inhibitors”. We did not exclude any study due to linguistic restrictions and the references of retrieved studies were screened to identify additional reports which may have been neglected. The electronic search strategies are provided in Supplementary (Table S1).
| Databases | Queries |
| PubMed | |
| #1 | Atrial fibrillation [MeSH Terms] OR AF [Title/Abstract] |
| #2 | Cancer [MeSH Terms] OR carcinoma [Title/Abstract] |
| #3 | new oral anticoagulants [Title/Abstract] OR non-vitamin K antagonist oral anticoagulants [Title/Abstract] OR rivaroxaban [Title/Abstract] OR apixaban [Title/Abstract] OR edoxaban [Title/Abstract] OR oral factor Xa inhibitors [Title/Abstract] OR dabigatran [Title/Abstract] OR oral factor Ⅱa inhibitors [Title/Abstract] |
| #4 | Warfarin [MeSH Terms] OR vitamin K antagonists [Title/Abstract] |
| #5 | #1 and #2 and #3 and #4 |
| EMBASE | |
| #1 | ‘Atrial fibrillation’: ab, ti OR ‘AF': ab, ti |
| #2 | ‘cancer’: ab, ti OR ‘carcinoma’: ab, ti |
| #3 | ‘new oral anticoagulants’: ab, ti OR ‘non-vitamin k antagonist oral anticoagulants’: ab, ti OR rivaroxaban: ab, ti OR apixaban: ab, ti OR edoxaban: ab, ti OR ‘oral factor Xa inhibitors’: ab, ti OR dabigatran: ab, ti OR ‘oral factor ⅱ a inhibitors’: ab, ti |
| #4 | ‘warfarin’: ab, ti OR ‘vitamin K antagonists’: ab, ti |
| #5 | #1 and #2and #3 and #4 |
Table S1: Electronic search strategies determined on June, 2020.
Studies were considered to be eligible meeting the following criteria
1. Study design: observational studies.
2. Study population: AF patients with cancer.
3. Interventions: any NOAC (apixaban, dabigatran, edoxaban or rivaroxaban) compared to vitamin K antagonists or warfarin.
4. Outcomes: studies reported at least one of the efficacy or safety outcomes. Efficacy included Stroke or Systemic Embolism (SSE), Ischaemic Stroke (IS), Venous Thromboembolism (VTE) and all-cause death, and safety outcomes included major bleeding and intracranial or gastrointestinal bleeding.
The exclusion criteria were as follows
1) Articles such as systemic reviews, case reports and conference abstracts.
2) Clinical trials.
3) No data about endpoints and 4) absence of control groups.
To assess the efficacy and safety of NOACs versus VKA, we included the following clinical outcomes:
1) Efficacy outcomes, including Stroke or Systemic Embolism (SSE), Ischaemic Stroke (IS), Venous Thromboembolism (VTE) [30] and all-cause death.
2) Safety outcomes, including major bleeding, according to the definition of original research (e.g., International Society on Thrombosis and Hemostasis criteria) [31], and intracranial or gastrointestinal bleeding.
All of the retrieved articles were assessed by two reviewers (Bo Cao and Xiaobo Hu) independently. Titles and abstracts were screened for eligibility and then the full texts were reviewed in detail. The final selection of studies was performed by consensus. The extracted data included the first author’s name, the year of publication, design, time period, patient characteristics, population size, treatment, control, type of cancer and follow-up duration. The Newcastle-Ottawa Scale (NOS) was used to assess the quality of the included observational studies, which includes three components: selection (0-4 points), comparability (0-2 points) and the assessment of the outcome (0-3 points). The specific information is presented in Supplementary (Table S2).
| Included studies | Selection (0-4 points) | Comparability (0-2 points) | Outcome (0-3 points) | Total points | ||||||
| Selectionn of Non- Exposed Cohort | Ascertainment of Exposure | Demonstration That Outcome of Interest Was Not Present at Start of Study |
Adjust for the important Risk factors |
Adjust for other risk factors |
Assessment of outcome |
Follow- up length | Loss to follow- up rate | |||
| Kim K, et al. 2018 [32] | * | * | * | * | * | * | * | * | 8 | |
| Ording AG, et al. 2017 [33] | * | * | * | * | * | * | * | * | 8 | |
| Sawant AC, et al. 2019 [34] | * | * | * | * | * | * | 6 | |||
| Shah S, et al. 2018 [35] | * | * | * | * | * | * | * | * | 8 | |
| Yasui T, et al. 2019 [36] | * | * | * | * | * | * | * | * | * | 9 |
Table S2: NOS for assessment of quality of included studies.
The Newcastle-Ottawa Scale (NOS) items, with a total score of 9 points, were used to evaluate the quality of the observational studies, which involve the selection of cohorts (0-4 points), the comparability of cohorts (0-2 points), and the assessment of the outcome (0-3 points).
All of the statistical analyses were performed by using the Review Manager 5.3 software (the Nordic Cochrane Center, Rigshospitalet, Denmark) and Stata software (version 14.0, Stata Corp LP, College Station, TX). The Risk Ratio (RR) with 95% Confidence Interval (CI) was calculated for each included study, and then pooled by a randomeffects model using the Mantel-Haenszel method. The Cochrane Q test and I2 statistic were the most commonly used statistical methods to evaluate heterogeneity, where P<0.1 and I2 >50% indicated a substantial heterogeneity, respectively. In the sensitivity analysis, method of exclusion of one study at a time was used. We also performed the subgroup analysis based on the type of NOAC (apixaban, dabigatran or rivaroxaban). A value of P<0.05 was considered statistically significant.
Database search generated 127 related articles and 5 studies [32-36] were identified for analysis after exclusion according to the eligibility criteria (Figure 1). All studies were retrospective and included a total of 232,234 patients, 44,739 were treated with NOACs and the remaining 187,495 with VKAs. The baseline characteristics of the included studies are shown in (Table 1). All included studies had an acceptable quality with an (NOS score ≥ 6). Definitions of safety and efficacy endpoints in the 5 included studies were presented in Supplementary (Table S3).

Figure 1: Flowchart diagram illustrating study selection methodology.
| Study | Study design | Period |
Clinical Characteristics |
Population | Age (years) | Female (%) | ChA2DS2- VASc | Treatment | Control | Type of cancer | Follow- up(years) |
| Ording AG, et al. 2017 [33] | Retrospective cohort |
07/2004- 12/2013 |
AF, cancer | 11855 | 77 | 45.8 | 3.3 | NOAC | VKA | Gastrointestinal (12.0%), Cancer of the lung or pleura (3.9%), Breast (11.9%), Urological (14.5%) Intracranial (0.1%), Hematological (3.4%), All other cancer sites (54.2%) |
1 |
|
Kim K, et al. 2018 [32] |
Retrospective cohort |
11/2005- 06/2015 |
non-valvular AF, newly diagnosed cancer | 776 | 72.4 | 31.4 | 3.6 | apixaban, rivaroxaban, dabigatran | warfarin | Stomach (20.6%), colorectal (14.9%), thyroid (10.8%), prostate (9.3%), lung (12.2%), Melanoma (5.9%), biliary tract (5.4%), urinary tract (6.1%), genitourinary (12.2%), head and neck (4.1%), hepatocellular carcinoma (3.0%), breast (2.4%), ovary and endometrial (2.6%), renal cell carcinoma (3.1%), hematologic malignancy (2.2%), others (3.2%) |
1.8 |
|
Shah S, et al. 2018 [35] |
Retrospective cohort |
01/2010- 12/2014 |
non-valvular AF, cancer | 22862 | 74.9 | 40 | 4.4 | apixaban, rivaroxaban, dabigatran | warfarin | Breast (19.2%), gastrointestinal (12.7%), lung (12.3%), genitourinary (29.2%), gyneco- oncological (2.4%), hematological (9.8%), Others (14.4%) |
1 |
|
Yasui T, et al. 2019 [36] |
Retrospective cohort |
01/2014- 12/2016 |
AF, active cancer | 224 | 72.7 | 12.5 | 3.1 | apixaban, rivaroxaban, dabigatran | warfarin | Gastrointestinal (44.2%) , Lung (24.1%), Genitourinary (11.2%) , Head and neck (9.8%), Breast (4.0%), Hematological (3.1%), Other (3.6%) |
1 |
| Sawant AC, et al. 2019 [32] |
Retrospective cohort |
01/2010- 12/2015 |
AF, active cancer | 196517 | 76 | 1.9 | - | apixaban, rivaroxaban, dabigatran | warfarin | - | - |
Table 1: Baseline characteristics of included studies.
| References | Stroke or systemic embolism | Ischaemic stroke | VTE | All-cause death | Major bleeding | Intracranial or gastrointestinal bleeding |
| Kim K, et al. 2018 [32] | Ischaemic stroke or systemic thromboembolism | Ischaemic stroke | NA | All-cause death | ISTH | Intracranial or gastrointestinal bleeding |
| Ording AG, et al. 2017 [33] | Ischaemic stroke, hemorrhagic stroke, other arterial embolism | Ischaemic stroke | VTE | NA | hemorrhagic stroke or gastrointestinal, lung, or urinary hemorrhage | hemorrhagic stroke or gastrointestinal hemorrhage |
| Sawant AC, et al. 2019 [34] | Ischaemic stroke | Ischaemic stroke | NA | All-cause mortality | hemorrhagic stroke | hemorrhagic stroke |
| Shah S, et al. 2018 [35] | Ischaemic stroke | Ischaemic stroke | Venous thromboembolism | NA | Intracranial hemorrhage or gastrointestinal bleeding | Intracranial hemorrhage or gastrointestinal bleeding |
| Yasui T, et al. 2019 [36] | Stroke or systemic embolism | Ischaemic stroke | NA | NA | ISTH | Intracranial or gastrointestinal bleeding |
Table S3: Definitions of safety and efficacy endpoints in the 5 included studies.
Abbreviations: VTE: Venous Thromboembolism; ISTH: International Society on Thrombosis and Haemostasis; NA: Not Available
As presented in figure 2 and figure S1, the rates of stroke or systemic embolism were 10.7% in patients treated with NOACs versus 12.9% in those on VKAs, showing the decreased risk of SSE for NOACs users compared with VKAs (RR, 0.79; 95% CI, 0.69-0.90; I2= 86%). The incidence of ischaemic stroke was significantly different between NOACs and VKAs (10.1% vs. 11.82%; RR, 0.82; 95% CI, 0.72-0.93; I2= 85%). In two studies reporting venous thromboembolism, the use of NOACs was associated with reduced risk of VTE (2.4% vs. 5.9%, RR, 0.28; 95% CI, 0.14-0.53; I2= 92%) compared with VKAs. All-cause death was reported in two studies, and the incidence was significantly reduced in the NOACs group (25.9% vs. 44.8% in the VKAs group; RR, 0.57; 95% CI, 0.50-0.64; I2= 95%).
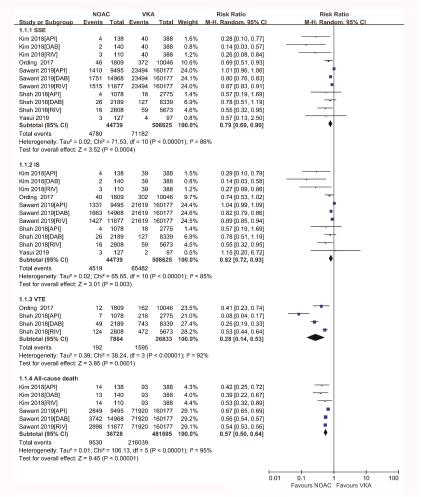
Figure 2: Forest plot comparing NOACs vs VKAs regarding the efficacy outcomes in real-world patients with AF and cancer. Atrial Fibrillation (AF); Stroke or Systemic Embolism (SSE); Ischaemic Stroke (IS); Venous Thromboembolism (VTE); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
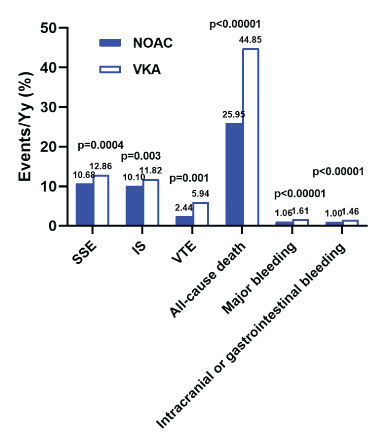
Figure S1: Incidence rates of clinical events in real-world AF patients with cancer receiving NOACs vs VKAs. Atrial Fibrillation (AF); Stroke or Systemic Embolism (SSE); Ischaemic Stroke (IS); Venous Thromboembolism (VTE); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs)
Major bleeding was reported in all included studies as shown in figure 3 and figure S1, the rate of major bleeding was significant lower in people treated with NOACs compared to VKAs (1.06% vs. 1.61%, RR, 0.60; 95% CI, 0.51-0.72; I2= 59%). Intracranial or gastrointestinal bleeding occurred in 1.0% of patients receiving NOACs and in 1.46% of those on VKAs (RR, 0.61; 95% CI, 0.51-0.73; I2= 59%).
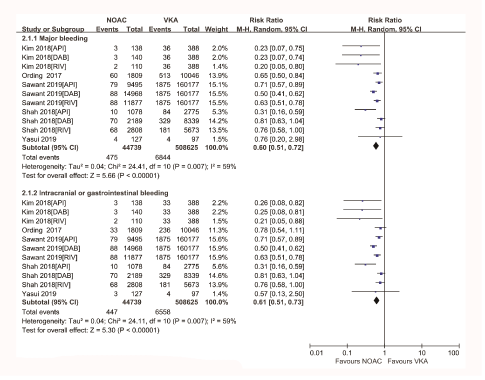
Figure 3: Forest plot comparing NOACs vs VKAs regarding the safety outcomes in real-world patients with AF and cancer. Atrial Fibrillation (AF); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
The sensitivity analysis was performed in analyzing the risk of stroke or systemic and major bleeding in NOACs versus VKAs, and the pooled effect results did not change when any individual included study was deleted at a time (Figure S2), which indicated reliable results.
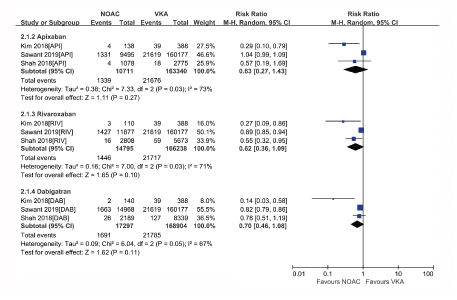
Figure S2: Forest plot comparing individual NOAC vs VKA regarding SSE in real-world patients with AF and cancer. Stroke or Systemic Embolism (SSE); Atrial Fibrillation (AF); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB)
The subgroup analysis was performed based on NOAC type, showing that apixaban, dabigatran or rivaroxaban had similar risks of SSE (Figure S3) or IS (Figure S4), but lower rates of all-cause death (Figure S5) compared with VKAs. In the safety outcome comparison of NOACs versus VKAs, all NOAC was associated with reduced risks of major bleeding (Figure S6) and intracranial or gastrointestinal bleeding (Figure S7) irrespective of NOAC type. It is difficult to evaluate publication bias with a funnel plot in analyses that include <10 studies, so we did not perform the publication bias analysis.
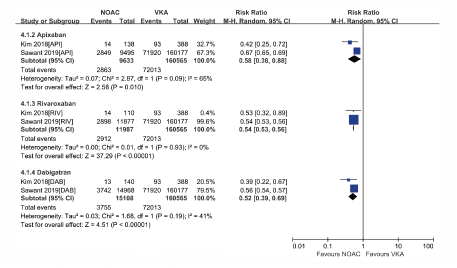
Figure S3: Forest plot comparing individual NOAC vs VKA regarding IS in real-world patients with AF and cancer. Ischaemic Stroke (IS); Atrial Fibrillation (AF); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
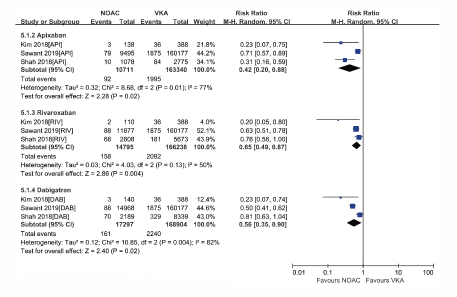
Figure S4: Forest plot comparing individual NOAC vs VKA regarding all-cause death in real-world patients with AF and cancer. Atrial Fibrillation (AF); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
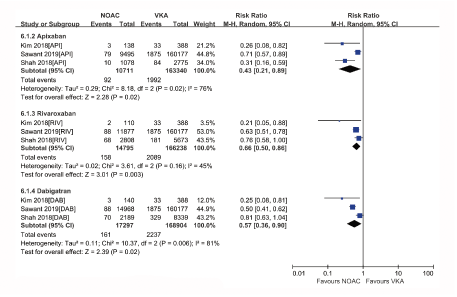
Figure S5: Forest plot comparing individual NOAC vs VKA regarding major bleeding in real-world patients with AF and cancer. Atrial Fibrillation (AF); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
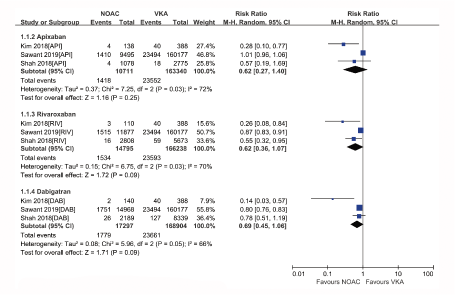
Figure S6: Forest plot comparing individual NOAC vs VKA regarding intracranial or gastrointestinal bleeding in real-world patients with AF and cancer. Atrial Fibrillation (AF); Non-Vitamin K Antagonist Oral Anticoagulants (NOACs); Vitamin K Antagonists (VKAs); Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
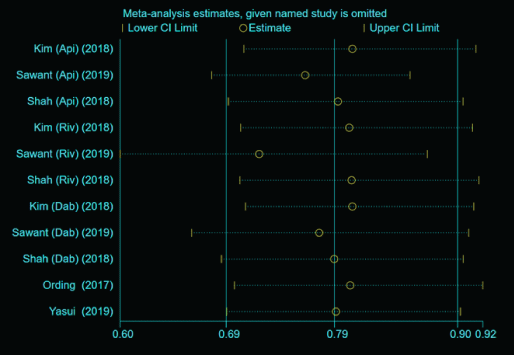
Figure S7: The leave-out-one sensitivity analysis results of the included studies. Apixaban (API); Rivaroxaban (RIV); Dabigatran (DAB).
In this meta-analysis of real-world observational studies, use of NOACs was associated with significant lower risks of stroke or systemic embolism, ischaemic stroke, VTE, all-cause death, major bleeding and intracranial or gastrointestinal bleeding compared to VKAs in AF patients with cancer. In subgroup analysis based on the type of NOACs, individual NOAC (apixaban, rivaroxaban or dabigatran) all showed similar rates of stroke or systemic embolism and ischaemic stroke, but lower rates of all-cause death, major bleeding and intracranial or gastrointestinal bleeding compared to VKAs.
The results are discordant from previous meta-analyses [26-28], which showed similar or lower rates of thromboembolic and bleeding events for NOACs users versus VKAs in AF patients with cancer. The results of Deng Y, et al. [28] showed the noninferior efficacy and safety of NOACs compared with VKAs but with wide confidence intervals and only borderline significant reductions in ischaemic stroke and major bleeding. Moreover, in Cavallari’s meta-analysis [27], there were no significant reductions in thromboembolic events or major bleeding. Three studies of post hoc analysis of the NOAC seminal trials, i.e., ARISTOTLE [37], ROCKETAF [23], ENGAGE AF-TIMI48 [38] were all included and pooled separately [27] or together [26,28] with observational studies in these meta-analyses. However, participants in randomized clinical trials do not always represent the patients in real-world settings. Therefore, in our meta-analysis, only the realworld observational studies were included and we have extended the research period to June 2020, with much larger number of patients (232,234) in real-life settings.
Compared with pooled analysis of three NOAC seminal trials [28], the rates of stroke or systemic embolism, ischaemic stroke, VTE and all-cause death were much higher while the risks of major bleeding and intracranial or gastrointestinal bleeding were much lower (Figure S1) in our combined analysis of real-world observational studies. The pooled analysis of three randomized trials showed similar risks of stroke/systemic embolism, ischaemic stroke, VTE and all-cause death with NOACs compared to VKAs, and only the rate of intracranial bleeding reduced in NOACs versus VKAs group [27]. However, in our analysis of real-world data, use of NOACs decreased the risks of all thromboembolic events compared to VKAs, as well as bleeding events. The difference could be explained by the more restricted clinical trial populations and the more controlled clinical environment in the followup daily practice in the clinical trials compared with real-life settings. However, observational studies always reflect more about practical use of these drugs in clinical anticoagulation therapy. Actually, practical factors like cancer types, stages, antineoplastic drugs, comorbidities, patient preferences, potential drug-drug interactions all can affect real-world anticoagulation outcomes [39]. For example, pancreas, stomach, gynecological, brain, lung, hematological, and metastatic cancers are reported to have the higher propensity of thromboembolic risks [40,41]. In our analysis of included observational studies, proportion of these cancer types was obviously higher than that in three randomized trials (Table 1). Indeed, gastrointestinal malignancy was also associated with higher risk of gastrointestinal hemorrhages. The proportion of these patients, up to 44.2% in Yasui T, et al. study [36] and 20.6% in Kim’s study [32], was also much higher than that in three RCTs (average 8.5%) [28]. Different from the similar results of NOACs versus VKAs regarding thromboembolic and bleeding events in RCTs [27], the incidences of both thromboembolic and bleeding events were significantly lower in our analysis, indicating a better efficacy and safety profile of NOACs compared to VKAs in AF patients with cancer in real-world settings.
In the aging population, the incidence and prevalence of AF, VTE and cancer is increasing and evidence shows that there is higher risk of VTE and AF in cancer patients [42,43]. Indeed, thrombotic events are the second leading cause of mortality in patients with malignancy as approximately 20% of all VTE cases occur in patients with cancer [44,45]. Possible mechanism for higher prevalence of AF in cancer patients is autoimmune paraneoplastic syndrome, autonomic nervous dysfunction, systemic inflammation and a hypercoagulable state promoted by releasing fibrinolytic and procoagulant agents in this patient population [46]. Thus, the risk of thromboembolism in patients with AF and cancer would be higher and the optimized anticoagulant therapy should be explored. The SSC ISTH guidance from August 2019 recommends the use of NOACs over a VKA in patients on chemotherapy with newly diagnosed AF and individualized anticoagulation is recommended based wherever possible on the risk of stroke, bleeding, and patient values. However, the effect of NOACs might be interfered by the anticancer drugs in patients undergoing chemotherapy as these agents are inhibitors or inducers of P-glycoprotein or CYP3A4 [47]. Therefore, the drug-todrug interactions should be taken into consideration when NOACs are used in these patients.
We noticed that the statistical heterogeneity was significant across the included cohorts in some comparisons, however, the pooled results did not change in the sensitivity analysis, indicating that our results were reliable. Nevertheless, confouners do exist due to the nature of real-world data. First, the population across the included studies was heterogeneous, that two studies were Asians, two were Americans and one study was European patients. As have been reported, racial differences in baseline characteristics (e.g. age, body weight, comorbidities) may influence clinical outcomes [48]. Notably, Asian patients with AF have higher incidences of stroke and bleeding (especially intracranial bleeding) than non-Asians [48-50]. Moreover, reduced-dose NOACs therapy is common in Asia as many physicians have a tendency to prescribe a lower dose of NOACs to avoid bleeding complications [51]. Take dabigatran as an example, 88% patients were prescribed with 110 mg and only 12% received 150 mg in Asia [52]. As a result, the heterogeneous population and different regional anticoagulation therapy could lead to uncontrolled confounding. Despite the mainly same basic characteristics, patients in Sawant AC, et al. [34] were mainly males (98.1%) as its population was selected from the veteran’s affairs database and there’s also low proportion of female (12.5%) in Yasui T, et al. [36], which were different from three other cohorts with a mainly female proportion of 30% to 40%. As female sex is an independent risk factor for AF-related stroke [53], and evidence shows that women are associated with an increased risk of thromboembolism [54,55]. Therefore, the extremely lower proportion of female in Sawant AC, et al. [34] and Yasui T, et al. [36] studies may lead to lower incidence of thromboembolism than in real world, which could result in the heterogeneity as well. Second, the type and dose of NOACs were different among these studies. The Danish cohort study [33] did not define which type of NOACs or VKAs was used, and patients in Kim k, et al. [32] study used both full and reduceddose of NOACs compared to warfarin. In addition, type of cancer and stage, the anticancer medication and type of chemotherapy drugs were also different across included studies, which may also count for the heterogeneity. Therefore, the effect of NOACs versus VKAs in AF and cancer should be further investigated for the optimal anticoagulation strategies taken the heterogeneity into consideration.
The subgroup analysis based on the NOAC type found that apixaban, rivaroxaban, or dabigatran had similar risks of stroke or systemic embolism, ischaemic stroke but reduced risks of all-cause death, major bleeding and intracranial or gastrointestinal bleeding compared with VKAs. However, it was notable that the risks of stroke or systemic embolism and ischaemic stroke were reduced when the data was pooled compared with VKAs. It might be explained by the limited number of studies, and even fewer studies analyzed in the subgroup analysis. Thus, further well-controlled large-scale studies would be needed to confirm the corresponding results. Overall, this meta-analysis of observational studies with more than 230,000 AF patients with cancer in real-world data showed better efficacy and safety profiles of NOACs compared to VKAs.
Several limitations should be acknowledged in this meta-analysis. First, the heterogenicity of cancer, i.e., the cancer type, cancer staging and the chemotherapy drug, might result in bias. Second, the time in therapeutic range was not considered because only one included study [35] reported it in patients receiving VKAs. Third, residual confounders from unmeasured variables might exist due to the nature of real-world data. Fourth, two of the included cohorts had a relatively small number of study populations, which may limit the analytical power. Finally, the included observational studies were all retrospective, and high-quality prospective cohort’s studies maybe required to confirm our findings.
Based on published real-world studies, NOACs showed lower risks of stroke or systemic embolism, ischaemic stroke, VTE, allcause death and reduced rates of major bleeding and intracranial or gastrointestinal bleeding compared to VKAs irrespective of NOAC type in patients with AF and cancer.
Bo Cao and Xiaobo Hu contributed equally to this paper.
Mingfeng Shen and Lan Xu.
Literature search, study selection and data extraction, quality assessment, and statistical analysis: Bo Cao and Xiaobo Hu.
Bo Cao and Lan Xu.
Min Chen and Mingfeng Shen.
All authors approved the final version of the manuscript.
The authors declare that they have no conflict of interest.
Not required.
No external funding was available for this research.
Download Provisional PDF Here
Article Type: RESEARCH ARTICLE
Citation: Cao B, Hu X, Chen M, Shen M, Xu L (2021) Efficacy and Safety of Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation and Cancer in Real-World: Meta-Analysis of Retrospective Observational Studies. J Drug Res Dev 7(1): dx.doi.org/10.16966/2470-1009.162
Copyright: © 2021 Cao B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access