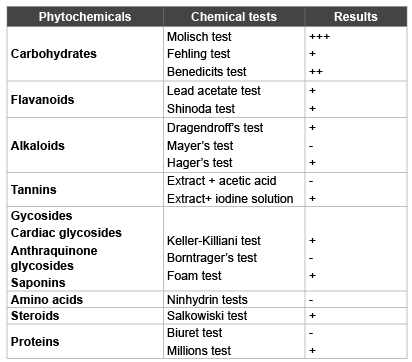
Table 1: Preliminary phytochemical screening of AMHE

Sunil Kumar1 Manjusha Choudhary1* Priya Yadav1 Nitesh2 Vikaas Budhwar3
1Institute of Pharmaceutical Sciences, Kurukshetra University, Kurukshetra-136119, Haryana, India*Corresponding author: Manjusha Choudhary, Institute of Pharmaceutical Sciences, Kurukshetra University, Kurukshetra-136119, Haryana, India, E-mail: manjushachoudhary@gmail.com
Objective: The present study was carried out to investigate the antidiabetic activity of hydro alcoholic extract of Acacia melanoxylon (AMHE) seeds.
Methods: AMHE was administered orally at 100, 200 and 400 mg/kg doses to normal and streptozotocin (STZ) induced type-2 diabetic rats. Oral glucose tolerance test was performed by inducing hyperglycemic state via administration of glucose (2 g/kg) in water. Fasting blood glucose level, biochemical parameters like serum cholesterol, creatinine, triglycerides, HDL cholesterol, urea, total protein and physical parameters like change in body weight was performed for evaluation of hypoglycemic effects.
Results: The extract was found safe upto dose of 2000 mg/kg body weight when administered orally. A significant decrease in blood was observed within 90 minutes in glucose tolerance testing among STZ-induced diabetic rats with a high dose of AMHE(400 mg/kg). Daily oral treatment with the extract for 21 days significantly (P<0.01) reduced blood glucose in STZ induced type-2 diabetic rats. The altered levels of biochemical parameters in diabetic animals as compared to normal indicating impaired metabolic functions were also significantly improved by oral administration of AMHE.
Conclusion: The results suggest that hydro alcoholic extract of A. melanoxylon seeds revealed significant anti-diabetic activity.
Hypoglycaemic activity; Acacia melanoxylon; Streptozotocin
Diabetes mellitus is a chronic metabolic disorder characterized by hyperglycemia is a major cause of morbidity and mortality. Recently, it was estimated that over 150 million people throughout the world had diabetes which is likely to be increased to double by year 2025. India leads the world today with highest number of diabetes and is declared “Diabetic Capital of World” by International Diabetes Federation [1].
Diabetic patient can be clinically categorized as Insulin Dependent Diabetes Mellitus (IDDM) type-1 diabetes a T-cell mediated autoimmune disease in which there is β-cell destruction in pancreatic islet or NonInsulin Dependent Diabetes Mellitus (NIDDM) type-2 diabetes that involves combination of resistance to insulin by both muscle and liver with improper function of pancreatic β-cell. World Health Organization (WHO) and American Diabetes Association (ADA) subdivide this disease in autoimmune 1A and idiopathic 1B on the basis of presence and absence of β-cell antibody respectively. Genetic and environmental factor affects the risk of developing either type-1or type-2. Some ethnic groups like African-American and Hispanics have higher incidence of type-2 diabetes. Marked hyperglycemia leads to number of microvascular (nephropathy, neuropathy and retinopathy) and macrovasclar (coronary disease, cerebral disease and peripheral disease) complications [2,3]. Traditional medicinal plants are considered as good source for a new drug or a lead to make new drug as they have advantage of having no or little side effect [4].
Acacia is a genus of trees and shrubs belonging to subfamily Mimosoideae of the family Fabaceae. Acacia trees are considered as valuable forest species and are useful for various purposes including furniture, woodcraft products, charcoal, firewood and gum extraction [5]. Acacia melanoxylon Linn. commonly known as Australian blackwood is a shrub or small to large trees 35-40 meter in length. The leaves in young plant are bipinnately compound but soon replaced by narrowly elliptical to lanceolate and usually curved pyllodes with reticulate veins. Seeds are glossy black, 3-5 mm long, broadly ellipsoid and compressed [6]. It is found in Kerala, Manipur, Meghalya, Tamil Nadu, West Bengal and Sikkim [7]. The constituents reported in this plant are quercetin, melacacidin [8], leucoanthocyanidins [9] and acamelin [10]. The plant has been reported to possess anti-helmintic [11] and antioxidant [12] activites. The lectin of seeds of plant exhibits a strong cytotoxic effect in a brine shrimp and insecticidal activities on mosquito larva [13]. Therefore; the present study is an attempt to investigate the anti-diabetic activity of hydroalcoholic extract of Acacia melanoxylon seeds.
Acacia melanoxylon seeds were collected from Chhajed Garden, Pune, Maharashtra, India in the month of December, 2013. The seeds were authenticated by Dr. B.D Vashistha, Chairman, Department of Botany, Kurukshetra University and were kept in the herbarium of Department of Botany, Kurukshetra University for further future reference (KUK/BOT/ IPS-16).
Seeds were washed and dried under shade at room temperature for one month. The dried seeds were powdered and stored in air tight container. One kg powder was extracted with hydro-alcohol in a ratio of 30:70 by soxhlet extraction at temperature of 65-75°C until the siphoning tube liquid become colorless. The remaining solvent was removed at 40-50°C in rotatory evaporator under reduced pressure to give solid extract which was then weighed to calculate percentage yield. Percentage yield of seed extract was 12.7%. The dried extract was stored in air tight container at 4-8°C for further investigation.
Phytochemical screening of AMHE was conducted using 5% solution of all the samples prepared in distilled water and filtered. Filtrate was subjected to phytochemical screening using the following reagent and chemicals:alkaloids with dragendroff ’s reagent, flavonoids with the use of Mg and HCl, amino acids with Millon’s reagent, tannins with ferric chloride and saponins with the ability to produce stable foam. Characteristic colour changes were identified using standard operating procedures [14].
Healthy Wister rats of either sex (150-200 g) were obtained from disease free animal house of National Institute of Pharmaceutical Sciences and Research (NIPER), Mohali, India. The animals were acclimatized for 7 days in the Animal House, Institute of Pharmaceutical Sciences, Kurukshetra University, Kurukshetra. They were housed in polypropylene cages under standard environmental conditions i.e ambient temperature of 22 ± 2°C at 45-55% relative humidity and 12/12 h light/dark cycle. The rats were allowed to take standard laboratory feed and water ad libitum. The study was approved by Institutional Animal Ethics Committee (Reg. No. 562/GO/02/a/CPCSEA).
Toxicity study was performed as per internationally accepted Organization for Economic Co-operation and Development (OECD)-423 guidelines. Three male Wistar rats were used for each dose. The dose levels of 50, 500, 1000, 2000, and 5000 mg/kg/body weight were selected [15]. The extract was administered to fasted rats. The animals were observed individually for sign of acute toxicity, such as behavioural changes, locomotion, convulsions, and mortality for 72 hours.
The effect of AMHE was evaluated on glucose loaded animals. Overnight fasted rats were separated into six groups of five animals each. Group I served as normal control. Group II received glucose solution (2 g/ kg). Group III, IV and V were given orally glucose (2 g/kg) with the AMHE at doses of 100, 200 and 400 mg/kg b.w. respectively. Group VI received glucose (2 g/kg) and drug glibenclamide. Blood samples were collected from the animals at 0, 30, 60 and 90 minutes after the administration of glucose and glucose level were analysed [16].
Diabetes was induced in fasted animals by a single intraperitonial dose of 60 mg/kg of Streptozotocin (STZ) dissolved in 0.1 M fresh cold citrate buffer (pH 4.5).On the third day, blood samples were taken from retro orbital plexus of the animals for estimation of blood glucose levels by using the auto analyzer. Animals having hyperglycemia (i.e. with blood glucose of 185 to 460 mg/dl) were selected for the experiment [17].
Experimental procedure: Animals were fasted overnight and were divided into six groups of seven animals in each group. Group I served as vehicle control. Group II served as diabetic rats received STZ (60 mg/ kg) only. Group III, IV and V received AMHE at doses of 100, 200 and 400 mg/kg b.w. respectively. Group VI received glibenclamide (10 mg/kg). The effect of extract was studied in all the groups, for 21 days and blood samples were collected by retro-orbital plexus on 0, 5th, 10th, 15th and 21stday after oral administration.
Blood sample were centrifuged at 3000 rpm for 30 min. Serum was separated and stored at temperature of -20°C. Serum samples were analyzed for serum cholesterol, serum triglycerides, HDL cholesterol, LDL, total protein, urea creatinine, AST and ALT by using various kit methods.
On 21st day, animals were sacrificed and the pancreas of animals were excised and kept in 10% formalin solution after washing with normal saline. Histopathological parameters were analysed at Mittal Path Lab, Kurukshetra.
The data obtained was represented as Mean ± SD. The statistical significance was computed using One Way ANOVA followed by Dunnet’s multiple comparison test and compared with diabetic control group where the n=6 animals in each group were used. P<0.01 was considered statistically significant.
Preliminary phytochemical screening revealed the presence of carbohydrates, glycosides, flavonoids, alkaloids and steroids as shown in Table 1.

Table 1: Preliminary phytochemical screening of AMHE
In AMHE treated animal, there were no discernible behavioural changes seen during observation period and no mortality was observed up to72 h at the tested dose.
In oral glucose tolerance test, the blood glucose levels were estimated before and after drug treatment at different time intervals (Table 2). In the vehicle control group the blood glucose was found to increase from 74 ± 1.14 to 128.4 ± 1.1 in first 30 minutes. After 60 minutes of glucose loading, the blood glucose level was increased to 130 ± 1.70 and then slight decrease from 130 ± 1.70 to 120.5 ± 1.00 was observed at 90 minutes. In AMHE treated animals, only a little elevation in blood glucose was seen at the dose of 100 and 400 mg/kg but continuous increase in blood glucose was recorded at dose 200 mg/kg. Maximum glucose tolerance was seen at 90 minutes with AMHE at dose 400 mg/kg. A consistent and significant fall in blood glucose level was observed in rats treated with glibenclamide (10 mg/kg) at 30, 60 and 90 min after glucose administration.
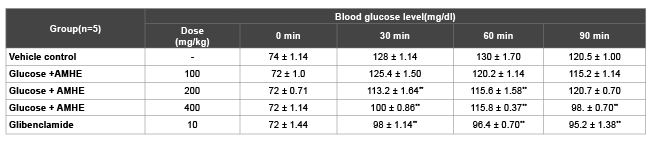
Table 2: Effect of AMHE on blood glucose level by using OGTT in normal rats
The values are mean ± SEM, n=number of animals used;
**P<0.01 vs vehicle control (One way ANOVA followed by Dunnett’s, Multiple comparison test)
Administration of STZ leads to elevation in blood glucose level in rats. In STZ induced diabetic rats the fasting blood glucose level was found to increase from 298 to 312 mg/dl. Oral administration of AMHE 100 mg/kg showed a significant (P<0.01) decrease in blood glucose level from 302.12 ± 3.23 to 225.34 ± 1.41 as compared with diabetic control. The daily treatment of rats with AMHE leads to dose dependent fall in blood glucose level (Table 3). AMHE at all doses showed significant (P<0.01) decrease in blood glucose level but effect at 400 mg/kg was superior. In the standard drug treated group, blood glucose was found to decrease throughout the study.
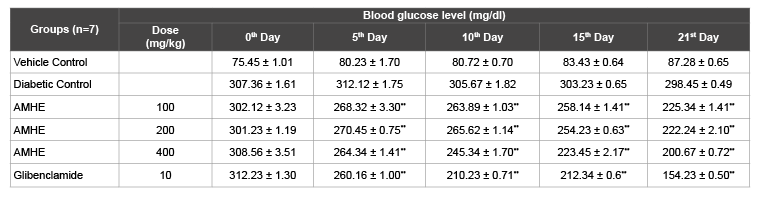
Table 3: Effect of AMHE on blood glucose level in STZ induced diabetic rats
The values are mean ± SEM, n=number of animals used;
**P< 0.01 vs diabetic control (One way ANOVA followed by Dunnett’s, Multiple comparison test)
Table 4 shows the effect of AMHE and glibenclamide on body weight of STZ-induced diabetic rats. Diabetic rats showed constant reduction in body weight during the study. The body weight of vehicle control group was increased from 217 to 227 in 21 days study. However in diabetic control group, weight was found to decrease from 228 to 212 g. AMHE treatment reversed the reduction in body weight and showed improvement in body weight gain continuously.
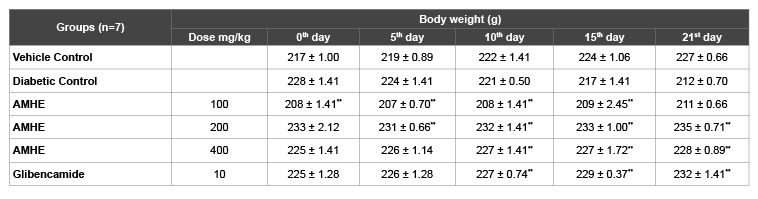
Table 4: Effect of AMHE on body weight in STZ induced diabetic rats
The values are mean ± SEM, n=number of animals used;
**P< 0.01 vs diabetic control (One way ANOVA followed by Dunnett’s, Multiple comparison test)
Serum cholesterol, LDL and TG levels were increased in STZ-induced diabetes. Doses 100, 200, 400 mg/kg of AMHE showed a dose related reduction in the concentrations of serum cholesterol, triglycerides, LDL cholesterol (Table 5). In diabetic rats, HDL cholesterol and total protein were decreased. There was significant (P<0.01) increase in HDL cholesterol at all doses whereas in total protein significant (P<0.01) increase was observed only at 100 and 400 mg/kg of extract (Table 6).
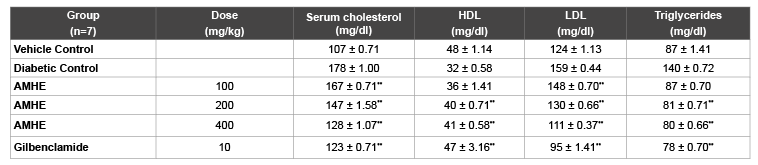
Table 5: Effect of AMHE on Lipid profile in STZ induced diabetic rats
The values are mean ± SEM, n=number of animals used;
**P< 0.01 vs diabetic control (One way ANOVA followed by Dunnett’s, Multiple comparison test).
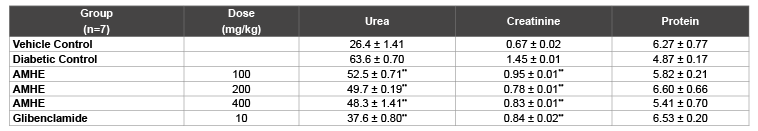
Table 6: Effect of AMHE on kidney function
The values are mean ± SEM, n=number of animals used;
**P< 0.01 vs diabetic control (One way ANOVA followed by Dunnett’s, Multiple comparison test)
Diabetes is a major health problem affecting major population throughout the world. Epidemiological studies and clinical trials strongly supports the notion that hyperglycemia is a main cause of complications associated with diabetes. Thus, effective blood glucose control can decrease the risk of developing micro vascular complications and most likely reduce the risk of macro vascular complications [18].
Excessive amount of blood glucose induces insulin secretion. This secreted insulin will stimulate peripheral glucose consumption and controls the production of glucose through different mechanisms [19]. The effect of standard drug glibenclamide used in this study on glucose tolerance has been attributed to secretion of larger amounts of insulin due to enhanced activity of beta cells of the pancreas. So the mechanism behind this anti-hyperglycemic activity of plant involves an insulinlike effect, probably, through peripheral consumption of glucose or enhancing the beta cells sensitivity to glucose, resulting in increased insulin release [20].
The hypoglycemic activity of AMHE was evaluated in STZ induced diabetic rats. STZ is a naturally occurring nitrosourea product of Streptomyces achromogenes used widely to induce diabetes in experimental animals [21]. It acts by selectively damaging β-cells of pancreatic islets. This damage to β-cell leads to the liberation of stored insulin that inhibits the insulin synthesis resulting in persistent diabetic state [22]. Administration of AMHE to STZ-induced diabetic rats showed significant and consistent decrease in blood glucose level throughout the period of study indicating its anti-diabetic activity.
Loss in body weight is observed in STZ-induced diabetic rats. This might be due to unavailability of carbohydrates for utilization as an energy source which leads to protein wasting [23]. The AMHE treated group enhanced glucose metabolism and thus, results in improved body weight in STZ diabetic rats.
The diabetic hyperglycemia induced by STZ produces elevation in urea and creatinine plasma level which are considered as significant markers of renal dysfunction [24]. While, after treatment of STZ-induced diabetic rats with AMHE, the level of urea and creatinine were significantly decreased which further indicate the utility of this plant in diabetes associated complications.
Hyperlipidemia is a recognized complication of diabetes mellitus demonstrated by elevated levels of phospholipids, tissue cholesterol and free fatty acids. The abnormal high concentration of serum lipids is regarded as a consequence of uninhibited action of lipolytic hormone on fat depots mainly due to action of insulin. Under normal conditions, enzyme lipoprotein lipase activated by insulin, hydrolyses the triglycerides. However in diabetic state, due to insulin deficiency lipoprotein lipase is not activated resulting in hypertriglyceridaemia [25]. In the present study, STZ-induced diabetic rats treated with AMHE significantly decreases serum cholesterol, triglycerides and LDL level.
Several authors have reported increase in AST and ALT activities in diabetic patients. Moreover, AST (a non-specific marker of hepatic injury) and ALT (a specific marker of hepatic injury) were used to evaluate the extent of hepatic damage in STZ-induced diabetic rats. Increase in activities of these enzymes might be due to the leakage of these enzymes from the tissue and migrating into the circulation [26]. Animals treated with STZ developed hepatic damage which was evident from increase in enzyme activities. The treatment of diabetic rats with AMHE was able to increase the level of these enzymes, demonstrating its hepatoprotective effect also.
AMHE exhibited significant hypoglycemic activities in STZ induced diabetic mice. The extract showed improvement in various serum and body parameters as well as regeneration of β cells of pancreas might be of value in diabetes. From the present study, we concluded the anti-diabetic activity of hydroalcoholic extract of Acacia melanoxylon. However, further investigation of this plant is necessary to isolate the active constituent and to elucidate its mechanism of action.
We declare that we have no conflict of interest.
The authors are thankful to Institute of Pharmaceutical Sciences, KUK, for providing essential facilities for the research work.
Download Provisional PDF Here
Article Type: Research Article
Citation: Kumar S, Choudhary M, Yadav P, Nitesh, Budhwar V (2016) Anti-Diabetic Activity of Hydroalcoholic Extract of Acacia melanoxylon Linn. Seeds in Streptozotocin Induced Diabetic Rats. J Dia Res Ther 2(3): doi http://dx.doi.org/10.16966/2380- 5544.120
Copyright: © 2016 Kumar S, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access