Figure 1: Designation of respondents.

Bisi Bright1* Wael Ali1 Niyi Fajimi1 Toyin Adesope1 Suzanne Tairu1 Patrick Sobande2 Ewaoche Sunday Itodo3
1LiveWell Initiative LWI, Nigeria/USA*Corresponding author: Bisi Bright, 1st Vice Chairman/CEO, LiveWell Initiative LWI, Nigeria/USA, Tel: 234 1 280 9337, 234 7018001787; Fax: 234 1 280 9330; E-mail: bisibright@livewellng.org
Background: LiveWell Initiative LWI is a self-funded nonprofit social enterprise which thrives on innovation.
The organization has, for 5 years, supervised MPH and Dr. PH Practicums for the Harvard T. H. Chan School of Public Health, Harvard University, Boston, USA. It also supervises PhD thesis at University of Helsinki, Finland. At the inception of COVID-19, LWI designed and compiled three (3) sets of study protocols in response to the COVID-19 response in Africa with a goal to arriving at a practical and affordable solution to the pandemic using 4-Aminoquinolines.
Method: A concurrent cohort/descriptive study of observation in patients exposed to HCQ/CQ prophylaxis and treatment with HCQ/CQ and Quinine with categorization as specified as not critically ill, critically ill but not on a ventilator and critically ill on a ventilator. One hundred and twenty-three (123) subjects were categorized into shelter-in-place, self-quarantine and self-isolation (Not critically ill, critically ill but not on a ventilator and critically ill on a ventilator). The 4-Aminoquinolines were administered with macrolide and zinc sulphate as appropriate for a defined duration and doses.
Results: The preliminary study of the 123 subjects covering all categories listed above resulted in 100% positive outcomes; nil death, nil relapse in symptomatic persons and total recovery with no relapse after 6 weeks lockdown, and asymptomatic persons post-prophylaxis, after 6 weeks lockdown. The results have been gathered principally from clinician reported outcomes with a few patient reported outcomes.
Conclusion: These repurposed drugs with unique strengths- Chloroquine and its analogue Hydroxychloroquine as well Quinine, hold sway in the treatment of COVID-19.
4-Aminoquinolines; Zinc sulphate; COVID-19; Ventilator; Pandemic; Repurposing; Ionophore
Perlman S [1] stated Coronavirus to be an envelope, positive single-strand RNA virus which belongs to the Orthocoronavirinae subfamily, as the name, it possesses the characteristic “crown-like” spikes on their surfaces. Bai Y, et al. [2] stated that alongside SARSCoV, bat SARS-like CoV and others, it also falls into the genus betacoronavirus.
COVID-19 enters the cell by using the S spike on its surface to bind onto an ACE2 receptor through a process of glycolysis. Once in the cell, the virus RNA uses the host cell ribosome to synthesize more RNA and protein. The protein is used to form the virus the capsule that protects the genetic material RNA. After making a million copies of itself using the host ribosome, it then assembles new viral particles and leaves the cell to find a new host.
The immune system which is a complex interconnection of molecular interactions across the cell, nucleus and cytoplasm, sometimes overreacts because of its weakened and dysfunctional state during the process of infection, releasing cytokines than required resulting in a cytokine storm. As of present, there is yet to be an ideal treatment for COVID-19, the treatment is mainly supportive.
The 3 sets of protocols designed by LiveWell Initiative have undergone professional debates among physicians, researchers and pharmacists for Hypothesis Testing over 5 weeks. Thereafter the team presented a global webinar on April 15th 2020 at which the panelists were the country rep WHO Nigeria Dr. Fiona Braka, The FIP Vice President, EMROPHARM Secretary-General, AFRO Lead at FIP, UN Ambassador of Goodwill Dr. Corinne Shenouda and a Clinician at Mayo Clinic, Dr. Michael Leise. The webinar series was thereafter taken along with the Nigeria National COVID-19 THINK TANK and a third webinar was held with the medical community in Nigeria, hosted by GSK.
The study protocols are currently undergoing random physicianpatient trials at the discretion of prescribing clinicians and clinical researchers; they are as recommended in a compilation of recent findings by LiveWell Initiative LWI on COVID-19. LiveWell Initiative LWI, a nonprofit organization, takes no liability for damage from the use of the above-suggested study protocols for COVID-19 response in Africa. It is a study protocol designed to ‘evolve’ as an African Solution to COVID-19 response.
This document is not intended for non-physicians, non-researchers or non-pharmacists. It is strictly meant for research, as we look towards a cure for the pandemic. The outcomes of the empirical data are encouraging, as there has been 100% recovery from COVID-19 on 11 patients at an Isolation Center in Oyo State Nigeria; and a patient in faraway Canada recovered under the ventilator, with the protocols. The study is to be scaled through prospective partnership or collaboration. This is also an open call for partnerships to scathe COVID-19 response in Africa and the Diaspora.
- Asymptomatic healthcare workers working in non-COVID hospitals/non-COVID areas
- Asymptomatic healthcare workers involved in containment and treatment of COVID-19
- Asymptomatic frontline workers
- Asymptomatic household contacts of laboratory-confirmed cases
- Symptomatic self-isolated persons
- Symptomatic hospital isolated persons
- Symptomatic critically ill persons
- Retinopathy
- Hypersensitivity to HCQ or 4-aminoquinoline compounds
- G6PD deficiency
- Pre-existing cardiomyopathy and cardiac rhythm disorders
Study design and COVID-19 testing: Most patients were not tested for COVID-19 because this was principally a prophylaxis trial, and the preliminary studies took place during the early phase of COVID-19 in the country, between late April and July during which time there were no accredited testing laboratories and government was still in the process of building and expanding its molecular laboratories in collaboration with the national coalition on COVID-19, known as CA-COVID.
However, the 11 patients admitted at the OYO Isolation Center were tested (PCR-Positive). Same PCR Testing for the 2 other isolated patients who tested positive, the U7K Nurse and the patient on ICU admission, were all PCR positive tested at the pint of recruitment in the preliminary study.
For this reason, elimination and inclusion criteria exempted laboratory testing for the prophylaxis initially but not for treatment at the isolation center; as this was focused more on exposure, travel, movement and ‘zoning’ which categorized exposure in terms of the blue, orange and red zones in terms of movement. Whereas, blue zone depicted one’s home or within 100 meters movement, the orange zones depicted shops, restaurants, and red zones depicted all travels, places of worship, bars, and indoor venues.
Justification for testing: In Lagos, enrollees were recruited based on exposure; pre or post exposure; however our Research Center in Ibadan already had an ethical permit pending and the HREC while being awaited we were working with partner clinical researchers at the Ibadan Isolation Center so all patients were tested positive prior to recruitment, which afforded us the opportunity to test the 4-Aminoquinolines on positively tested clients.
Doses: Participants received either CQ or HCQ, not both.
The administered doses were:
- CQ 400 mg stat daily or, HCQ 500 mg stat daily and Azithromycin 250 mg stat daily for 3 days, PrEP; and
- CQ 400 mg stat daily or, HCQ 500 mg stat daily and Azithromycin 500 mg stat daily for 5 days, PEP
- CQ 400 mg stat or HCQ 500 mg stat once weekly, for postdischarge prophylaxis
I. Shelter-in-place: 1.5 G Chloroquine, 1.2 G Hydroxychloroquine and 0.7 G Azithromycin Maximum.
- Chloroquine 500 mg daily for three days
- Hydroxychloroquine 400 mg daily for three days
- Azithromycin 250 mg daily for three days
II. Self-Quarantine: 2.5 G Chloroquine, 2.0 G Hydroxychloroquine; and 1.5-2 G Azithromycin Maximum.
- Chloroquine 500 mg 12 hourly stat, then 500 mg daily for three days
- Hydroxychloroquine 400 mg 12 hourly stat, then 400 mg daily for seven days
- Azithromycin 250 mg daily for five to seven days
III. Self-Isolation: 1.8 G Quinine; 1.5-2 G Azithromycin Maximum.
(Not critically ill)
- Quinine 600 mg 8 hourly for five days
- Azithromycin 500 mg daily for seven days
- Zinc sulphate 220 mg daily for seven days (Critically ill)
- Quinine Intravenous with Dextrose saline 8 hourly for five days
- Azithromycin 500 mg intravenously
- Zinc sulphate 220 mg daily for seven days
- Generous fluid
IV. Treatment doses below 4G sustained due to the ionophoric effect of zinc on Chloroquine, which makes lower doses of Chloroquine more efficacious due to enhanced tissue binding affinity and uptake by the viral cell. Other mechanisms of action of HCQ/CQ offer addition a protection to the host. HCQ/CQ will alter the pH in the ACE2 Receptor thus making it difficult for the virus to penetrate the host cell. In addition, it breaks the polymerase chain, and, in advanced COVID-19 exhibits its Haemozoin inhibitor action, thus disabling the virus from engulfing food vacuoles of debris and dead blood cells. Quinine, highly soluble and crosses the blood brain barrier; penetrating in advanced COVID-19 patients into the alveoli and dislodging the viruses. Thus, the usefulness of the 4-Aminoquinolines end to end in COVID-19.
Use beyond 7 weeks: For its use beyond 7 weeks on weekly dosage with strict monitoring of clinical and ECG parameters which would also ensure that the therapy is given under supervision. This is ideally for IPT Intermittent Prophylactic Therapy post-recovery, as the virus is still shed through the bowels after recovery. For further research we recommend the bowel infiltrates of the virus for possible vaccine development as it is believed to be the attenuated virus.
Safety as a major concern: The safety of Chloroquine or Hydroxychloroquine is premised on the use of a dose below 4G cumulative total loading dose. This is carefully titrated at LWI. As long as HCQ/CQ is used at therapeutic concentrations it remains a very safe drug with a broad therapeutic margin. Even Quinine, used in advanced COVID-19, remains very safe with no requirements for TDM within the specified therapeutic concentrations where side effects are self limiting, and, reversible; namely reversible ototoxicity.
Affordable, replicable, scalable: The remedy is affordable, scalable and replicable for all low-income economies. It is hereby strongly recommended.
A preliminary study of 123 subjects covering all categories listed (Table 1) resulted in 100% positive outcomes, post-treatment; Nil death, Nil relapse in symptomatic persons and total recovery with no relapse after 6 weeks lockdown, and asymptomatic persons. Outcomes are mostly clinician reported outcomes, post-treatment; with a few patient reported outcomes, namely FHCW (Frontline Healthcare Workers) and HCW (Healthcare Workers). Oyo State Isolation Center, where 11 positively tested persons were treated with the 4-Aminoquinolines, also recorded 100% positive outcomes with at least 2 negative PCR tests after treatment.
| Resource | Classification | PrEP | PEP | Age | Gender | Outpatient | Inpatient |
Critical Care |
Enrolment Criteria |
Lab. Test for COVID - PCR Test |
|
|
|||||||||
| F.A. | HCW | 20 | 8 | 18-35 (22) | 24 M | - | - | - | Asympt (28) | Nil Tested |
| 36-55 (6) | 4 F | |||||||||
| K.O. | HCW | - | 1 | 36-55 (1) | 1 F | - | - | - | Sympt (1) | Nil Tested |
| F.O. | FHCW | 20 | 2 | 18-35 (7) | 22 M | - | - | - | Asympt (20) | Nil Tested |
| 36-55 (15) | *Security men | Sympt (2) | ||||||||
| J.M. | FHCW | 5 | 15 | 18-35 (12) | 14 M | - | - | - | Asympt (20) | Nil Tested |
| 36-55 (8) | 6 F | |||||||||
| R.B. | >55 yrs | 2 | >55 yrs (2) | 1M | - | - | Asympt (2) | Nil Tested | ||
| 1F | ||||||||||
| A.P. | FHCW | 22 | 2 | 18-35 (14) | 13 M | - | - | - | Asympt (22) | Nil Tested |
| 36-55 (10) | 11 F | Sympt (2) | ||||||||
| U.U. | HCW | 1 | 36-55 (1) | - | - | - | Asympt (1) | Nil Tested | ||
| B.B. | HCW | 4 | 2 | 18-35 (2) | 2 M | - | - | - | Asympt (4) | Nil Tested |
| 36-55 (4) | 4 F | Sympt (2) | ||||||||
| B.A. | HCW | 1 | 36-55 (1) | 1F | Asympt (1) | Nil Tested | ||||
| S.S (proxy) |
HCW | - | 1 | >55 yrs (1) | 1 F | - | - | - | Sympt (1) | Nil Tested |
| JESS | Youth | - | 1* | 18-35 (1) | 1F | Sympt (1) | Online Tested | |||
| Y.A. | >55 yrs | 1 | 2* | 18-35 (2) | 3F | - | - | - | Asympt (1) | Online Tested |
| 36-55 (1) | Sympt (2) | |||||||||
| Nurse, UK | FHCW | 36-55 (1) | 1F | - | - | Sympt (1) | Lab. Tested +ve |
|||
| OYO State Isolation Center |
C-19 Patients/ FHCW |
18-35 (7) | - | 11 | - | Sympt (11) |
Lab. Tested +ve |
|||
| 36-55 (2) | ||||||||||
| >55 yrs (2) | ||||||||||
| Ventilator - Patient, Canada |
Patient’s Family |
>55 yrs (1) | 1F | - | - | 1 | Sympt (1) | LAB. TESTED +ve |
||
| Total | 76 | 34 | 123 | 1 | 11 | 1 | ||||
| Grand Total | 123 |
Table 1: Baseline preliminary study.
Oyo Isolation Center Ibadan-preliminary results of 11 Patients (see ‘missing’ ages here (Table 2); but this data was released long after we sent to press, in early September). No patient was symptomatic after treatment or prophylaxis, the enrolment criteria column speaks to the result after treatment. Those tested post-treatment were negative. Our conclusion on 100% recovery of all participants is hereby upheld. Even as at today, which is several months down the line, none of our clients has had a relapse. So the prophylaxis has worked well. Moving forward with the research, we do recommend repeat prophylaxis once in every 6-8 weeks for frontline workers and frontline healthcare workers and travellers; however this is for further research studies, when our randomized clinical trials approval, still pending, are obtained.
| S.No. | Age | Sex | DOA | Baseline | CT Value | Date | 1st Repeat | CT Value | Date | 2nd Repeat | CT Value |
Date | 3rd Repeat |
| 1 | 56 yrs | M | 27-04-2020 | Positive | 25.86, 29.2 | 27th April | Positive | 39.46 | 1/5/2020 | Negative | ˃40 | 12/5/2020 | Negative |
| 2 | 45 yrs | M | 27-04-2020 | Positive | 29.02 | 16th April | Negative | ˃40 | 1/5/2020 | Negative | ˃40 | 4/5/2020 | Negative |
| 3 | 22 yrs | M | 27-04-2020 | Positive | 33.26, 37.3 | 16th April | Negative | ˃40 | 1/5/2020 | Negative | ˃40 | 4/5/2020 | ------ |
| 4 | 26 yrs | F | 30-04-2020 | Positive | Not supplied from Lagos | * | Positive | * | 4/5/2020 | Positive | Negative | ||
| 5 | 30 yrs | F | 1/5/2020 | Positive | 37.14 | 27-04-2020 | Positive | 33.3 | 1/5/2020 | Negative | ˃40 | 13-05-2020 | Negative |
| 6 | 57 yrs | M | 1/5/2020 | Positive | 35.85 | 23-04-2020 | Negative | ˃40 | 4/5/2020 | Negative | ˃40 | 13-05-2020 | ------ |
| 7 | 52 yrs | F | 1/5/2020 | Positive | 24.69, 33.63 | 23-04-2020 | Negative | ˃40 | 4/5/2020 | Negative | ˃40 | 13-May | ------ |
| 8 | 20 yrs | F | 1/1/2020 | Positive | 33.27 | 24-04-2020 | Negative | ˃40 | 4/5/2020 | Negative | ˃40 | 13-05-2020 | ------ |
| 9 | 26 yrs | M | 1/1/2020 | Positive | 33.1 | 24-04-2020 | Negative | ˃40 | 4/5/2020 | Negative | ˃40 | 12/5/2020 | ------ |
| 10 | 20 yrs | M | 1/1/2020 | Positive | 30.95 | 24-04-2020 | Negative | ˃40 | 4/5/2020 | Negative | ˃40 | 13-05-2020 | ------ |
| 11 | 30 yrs | M | 1/5/2020 | Positive | 33.18 | 24-04-2020 | Negative | ˃40 | 4/5/2020 | Negative | ˃40 | 13-05-2020 | ------ |
Table 2: Oyo Isolation Center Ibadan-Preliminary results of 11 patients.
| PrEP | PEP |
| 20 | 8 |
| - | 1 |
| 20 | 2 |
| 5 | 15 |
| 2 | - |
| 22 | 2 |
| 1 | - |
| 4 | 2 |
| 1 | - |
| - | 1 |
| - | 1 |
| 1 | 2 |
| 76 | 34 |
Table 3: A preliminary study on Chloroquine and Hydroxychloroquine pre- and post- exposure prophylaxis.
| Classification | Laboratory Tested (n) | Non-Tested (n) | Online Tested - Awaiting Laboratory Testing | Total |
| PrEP | NIL | 75 | 1 | 76 |
| PEP | NIL | 31 | 3 | 34 |
Table 4: Laboratory testing for COVID-19.
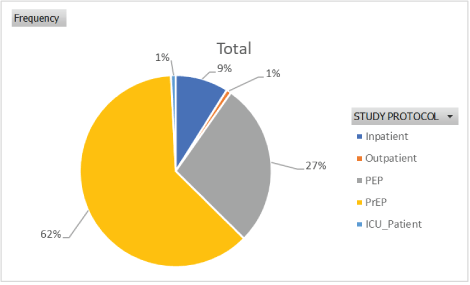
As seen in table 5, 62% of respondents took the 4-Aminoquinolines prophylactically and 27% were administered the drugs because of their contact with positive COVID-19 patients (Figure 1). From table 6, 52.85% of the respondents were adults while 2.4% were above 55 years (Figure 2). Table 7 showed that 69% were males while 31% were females (Figures 3-7 and Tables 8-11).

Figure 1: Designation of respondents.
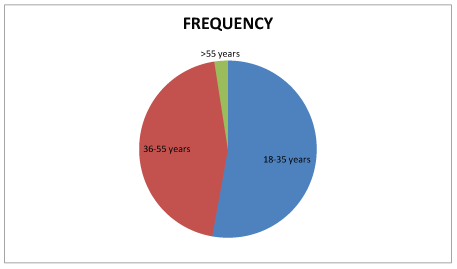
Figure 2: Age of respondents.
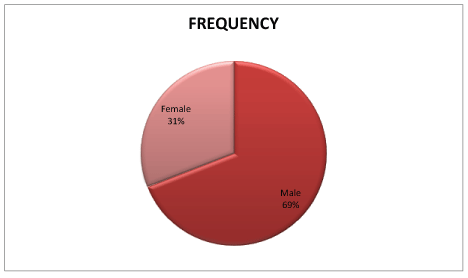
Figure 3: Gender of respondents.
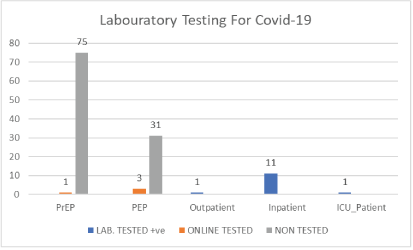
Figure 4: Laboratory testing for COVID-19.
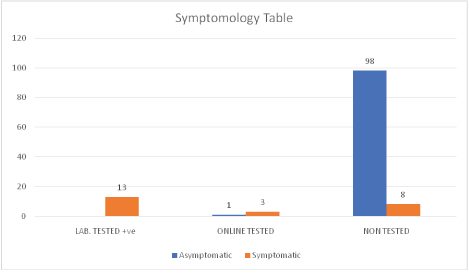
Figure 5: Symptomology table.
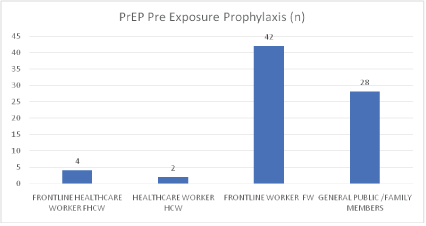
Figure 6: PrEP pre exposure prophylaxis.
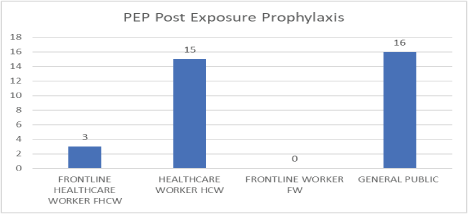
Figure 7: PEP post exposure prophylaxis.
| Study Protocol | Frequency |
| Inpatient | 11 |
| Outpatient | 1 |
| PEP | 34 |
| PrEP | 76 |
| ICU_Patient (Treated on Quinine I.V.) | 1 |
| Grand Total | 123 |
Table 5: Designation of respondents.
| Age (Years) | Frequency |
| 18-35 | 65 |
| 36-55 | 55 |
| >55 | 3 |
| Grand Total | 123 |
Table 6: Age of respondents.
| Gender | Frequency |
| Male | 76 |
| Female | 34 |
| Grand Total | 123 |
Table 7: Gender of respondents.
| Classification | Laboratory Tested (n) | Non- Tested (n) | Online Tested - Awaiting Laboratory Testing (2) |
Total |
| PrEP | Nil | 75 | 1 | 76 |
| PEP | Nil | 31 | 3 | 34 |
| Outpatient (U.K. Nurse) | 1 | - | - | 1 |
| Inpatient/Isolation Center (Oyo State Isolation Center) | 11 | - | - | 11 |
| Critical Care/Ventilator Patient (Canada) *treated on Quinine Injection I.V. |
1 | - | - | 1 |
| Total | 13 | 106 | 4 | 123 |
Table 8: Laboratory testing for COVID-19.
| Symptoms & Testing Stratification | Number (n) | Comments-After |
| Symptomatic, online tested, Awaiting laboratory test | 3 | General public-awaiting laboratory test for 3 weeks but now symptom-free |
| Symptomatic, Not laboratory tested | 8 | 4 frontline workers, 4 frontline healthcare workers, No symptoms |
| Symptomatic, Laboratory tested positive | 13 | 1 Inpatient in Canada, 1 Self Quarantined HCW in the UK, 11 Isolation Center inpatients in Nigeria; No Symptoms |
| Total | 24 | COVID-19 Free, 100% NIL mortality and NIL morbidity |
Table 9: Symptom outcomes assessment before and after use of Chloroquine and Hydroxychloroquine for COVID-19 prophylaxis.
| Category | PrEP Pre Exposure Prophylaxis (n) | Post-Lockdown/6 weeks after | Comments |
| Frontline Healthcare Worker (FHCW) | 4 | Nil Symptoms | COVID-19 Free |
| Healthcare Worker (HCW) | 2 | Nil Symptoms | COVID-19 Free |
| Frontline Worker (FW) | 42 | Nil Symptoms | COVID-19 Free (22- man Cohort of Security men and 20 Bankers) |
| General Public/Family Members | 28 | Nil Symptoms | COVID-19 Free |
| Total on PrEP | 76 | Post-PrEP post-lockdown symptom-free after 6 weeks | |
Table 10: Outcomes of pre-exposure prophylaxis using Chloroquine/Hydroxychloroquine for COVID-19 pre emptive-therapy.
| Category | PEP Post Exposure Prophylaxis | Post-Lockdown/6 weeks after | Comments |
| Frontline Healthcare Worker (FHCW) | 3 | Nil Symptoms | COVID-19 Free (3 Isolation Center Staffers) |
| Healthcare Worker (HCW) | 15 | Nil Symptoms | COVID-19 Free (15 Community Pharmacists) |
| Frontline Worker (FW) | - | - | - |
| General public | 16 | Nil Symptoms | COVID-19 Free *2 persons awaited Laboratory Testing after symptoms but are now symptom-free |
| Total on PEP | 34 | Post-PEP post-lockdown symptom-free after 6 weeks | |
Table 11: Outcomes of post exposure prophylaxis using Chloroquine/Hydroxychloroquine for COVID-19 prophylaxis.
As of present, there is yet to be a validated and consensus treatment for COVID-19, the treatment is mainly supportive. However, the repurposed drugs with unique strengths-Chloroquine and its analogue Hydroxychloroquine as well as Quinine, from time immemorial have been largely touted as possessing an anti-inflammatory and immunomodulatory properties, hence its use in the treatment of Systemic Lupus Erythematosus (SLE) and other rheumatism [3]. As the mechanisms of action of the 4-Aminoquinolines are been further elucidated, it is been believed to distort the ACE2 receptor by altering the glycan attachment thereby preventing the attachment of SARS-CoV2. It is also believed to decrease the pH of the cell thereby inhibiting viral replication, altering the viral particle assemblage and stopping cytokine storm [4-6]. Researchers have even reported both prophylactic and therapeutic advantages of CQ for SARSCoV infection [7]. However, other researchers have reported in a retrospective study with no defined administration time that no evidence in the use of Hydroxychloroquine either with or without Azithromycin reduced the risk of a mechanical ventilator in patients hospitalized with COVID-19 [8,9] in an open-label non-randomized clinical trial concluded that Hydroxychloroquine showed a significant association with viral load reduction/disappearance in COVID-19 patients and its effect is reinforced by Azithromycin. In a randomized controlled trial in china of 62 patients who were positive for COVID-19 and hospitalized, Chen Z, et al. reported that 31 patients received Hydroxychloroquine 400 mg per day and the remaining 32 received a placebo, Chen Z, et al. concluded pneumonia improved in 81% compared to 55% control [10].
The LWI study protocols have undergone hypothesis testing among physicians, researchers, pharmacists and clinicians, with online debates on several professional health platforms.
The results in this preliminary study are based on preliminary data gathered from physician-patient recommendations of prophylaxis using the 4-Aminoquinolines in COVID-19 treatment and prophylaxis. It also recognizes some selfmedicating individuals who took advantage of the nonprescription remedy.
The LWI Study Protocols are currently being used in Kaduna State, Bauchi state, and some other states in Nigeria. The unique thing about the study protocols, the 4-Aminoquinolines offer an end to end care in COVID-19, from CQ/HCQ in pre and post exposure to mild and moderate COVID-19 and escalating into quinine I.V. for critical care in COVID-19.
CQ and HCQ Prophylaxis works as none of the 110 clients placed on prophylaxis has progressed into COVID-19 in 6 weeks postlockdown; none of them is symptomatic.
CQ/HCQ is relevant for ambulatory care as the laboratory tested positive healthcare worker on self quarantine who was treated with CQ is fully recovered, up to 6 weeks post-lockdown with no relapse, and having tested negative twice post-treatment.
CQ/HCQ is also relevant for inpatient care as the 11 laboratory tested positive patients placed on admission at the COVID-19 Isolation Center who were treated with CQ are all fully recovered, up to 6 weeks post-lockdown with no relapse, and having tested negative twice post-treatment.
Of note is the fact that CQ (400 mg) or HCQ (500 mg) exercised the same level of efficacy in prophylaxis and are recommended as an ‘either or’ prophylactic.
Quinine works in advanced COVID-19 as the single laboratory tested positive client on the ventilator, has fully recovered after treatment with I.V. Quinine and is still symptom-free 6 weeks postlockdown.
The preliminary study resulted in 100% positive outcomes, as all subjects recovered fully with no relapse. This was significantly measurable as symptomatic subjects at the entry level criteria were all asymptomatic at the end of the preliminary study.
The preliminary study concluded 100% recovery and all participants were asymptomatic after 6 weeks.
This preliminary study has resulted in very positive outcomes with CQ/HCQ in COVID-19 Response. However we know that Remdesivir was approved for compassionate use by the FDA in the USA when it was found to show shorter duration of hospitalization [1] and improved clinical outcomes among inpatients who received the drug [2]. From our findings, CQ/HCQ has shown great promise for PrEP against COVID-19 from some studies apart from our preliminary study; including an observational study pre-print among healthcare workers [3], but was reported ineffective for PEP in this population in another study [4]. Since the observational study recruited subjects online, this could be arguably faulted. However, other studies, including from China [5] have found only marginal benefits in Remdesivir use.
Another interesting observation is that those studies which applied CQ/HCQ without combining with Zinc had poorer outcomes with CQ/HCQ [6]. Some other studies with poor outcomes using CQ/HCQ were found to have administered very high doses of CQ/HCQ to elderly COVID-19 patients [7], some of whom were the oldest and sickest patients [7]; thus leaving questions on toxicity due to overdosage, and questioning the inclusion criteria.
Our carefully defined inclusion and exclusion criteria also helped us in obtaining positive outcomes from this preliminary study.
In applying for our Clinical Trial Approvals, we have proposed to double blind our CQ/HCQ trials against Ivermectin. These and other possibilities may be explored as we progress into clinical trials and will extend the scope of study to accommodate more positive outcomes.
Thank you.
Download Provisional PDF Here
Article Type: RESEARCH ARTICLE
Citation: Bright B, Ali W, Fajimi N, Adesope T, Tairu S, et al. (2021) A Preliminary Study on Various Types of 4-Aminoquinolines for Pre- or PostExposure Prophylaxis and for Treatment in Severe COVID-19. Clin Res Open Access 7(1): dx.doi.org/10.16966/2469-6714.165
Copyright: © 2021 Bright B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access