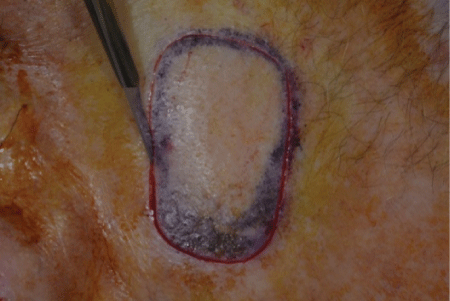
Figure 1: Score technique


Prativa SA Jayasekera1* Thomas Oliphant2 James AA Langtry2
1Royal Victoria Infirmary, Newcastle upon Tyne BM Medicine MRCP DERM, England, UK*Corresponding author: Prativa SA Jayasekera, Royal Victoria Infirmary, Newcastle upon Tyne BM Medicine MRCP DERM, England, UK, Tel: 0191 233 616; E-mail: prativaj@googlemail.com
We describe a technique of freehand scalpel harvesting whereby the split thickness skin graft to be harvested is outlined and the margin scored with a scalpel prior to freehand scalpel harvesting. We have experience of this technique with 11 cases and all of them had a good cosmetic outcome. We term this ‘Score and Shave’ and describe the technique with illustrative cases in the reconstruction of surgical wounds following Mohs Micrographic surgery (MMS) and standard excisional surgery. This technique is time efficient and relatively simple and does not require expensive equipment and therefore could be utilised as an alternative to standard methods of harvesting split thickness skin grafts.
Split thickness skin grafts; Mohs micrographic surgery
Techniques for harvesting split thickness skin grafts (STSG) include hand operated dermatomes, electric dermatomes and freehand scalpel harvesting. These may be used in conjunction with biological dressing or tumescent anaesthesia [1].There are a number of electric dermatomes available as well as hand operated dermatomes such as the Weck knife and the Humby knife. Other accepted partial thickness skin grafting techniques include pinch grafting and strip grafting with a sharp, disposable curette [2,3]. STSG consist of the epidermis and a small amount of the underlying dermis and they are classified according to their thickness; thin 0.005- 0.012 inches (0.2-0.3 mm), medium 0.012-0.018 inches (0.3-0.45 mm) and thick 0.018-0.28 inches (0.45-0.75 mm).
Snow et al. [4] described free hand scalpel harvesting in 1991, using a #15 scalpel blade in a bevelled or vertical incision to enter the dermis and slicing the upper dermis when at the desired level of the dermis to harvest the graft.
We describe a technique of freehand scalpel harvesting, whereby the STSG to be harvested is outlined and the margin scored with a scalpel prior to freehand scalpel harvesting [5]. We term this ‘score and shave’ and describe the technique and application in auricular reconstruction following Mohs micrographic surgery (MMS) [6]. Our technique differs from that of Snow et al for a number of reasons; firstly we perform a vertical score to outline the size and shape of STSG required which allows access to the superficial to mid dermis. Shave excision of the graft is undertaken with the blade held parallel to the skin surface contour.
The score and shave technique involves marking the donor site to the size of the STSG required with a surgical marker pen. The donor site is then injected with local anaesthetic. A #15 blade on a scalpel handle is used to gently score the perimeter of the graft to an appropriate level of the dermis, at a right angle to the skin surface (Figure 1). The #15 blade is then used to harvest the STSG by laying it flat against the dermis (Figure 2) and making a smooth and gentle horizontal cutting motion parallel to the skin surface at the same depth throughout the graft until the STSG is released (Figures 3 and 4). The intention is to maintain the blade at a constant level of the dermis throughout harvesting. The donor site will show pinpoint capillary bleeding points which may be cauterised with aluminium chloride hexahydrate 20% solution and a TegadermTM dressing is applied. The STSG harvested is then sutured to the recipient wound along with basting sutures figures 5 and 6 is a long term photograph.

Figure 1: Score technique
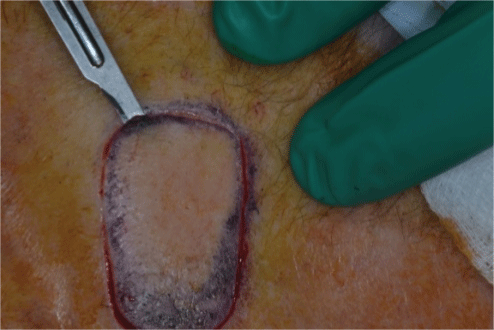
Figure 2: Shave technique
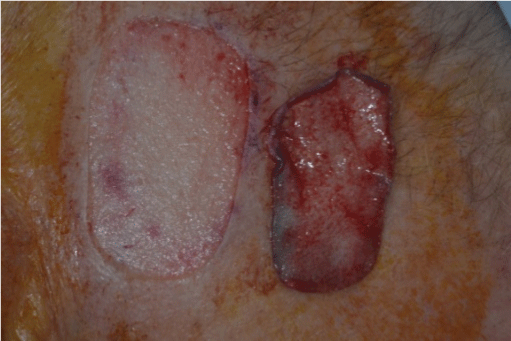
Figure 3: A Split Thickness Skin Graft (STSG) is produced

Figure 4: Post Mohs micrographic surgery defect
We report our experience with score and shave STSG with eleven patients (six females and 5 males) with an average age of 75 years. There were eight auricular defects, two toes and one finger. The average defect size was 21 × 15 mm. The majority of the donor sites were the post auricular ear where the ear was reconstructed and the thigh or neck for toe/finger reconstructions. All cosmetics outcomes were good. 10 of the defects resulted after Mohs micrographic surgery and one after standard excision. Figures 1-3 illustrate the score and shave technique, figure 4 shows the auricular MMS defect, figure 5 shows the graft sutured to the defect and figure 6 is a five month post operative photo.
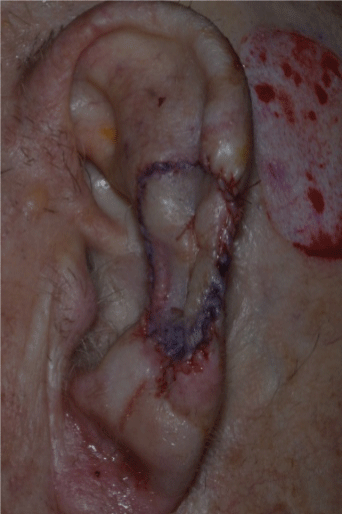
Figure 5: Graft has been sutured to the Mohs defect
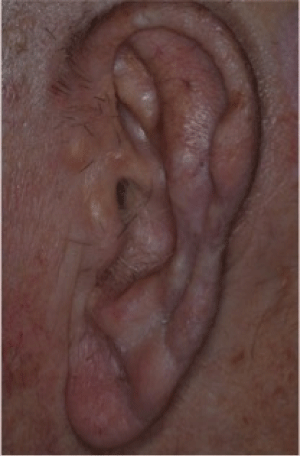
Figure 6: 5 month post operative photo
All the STSGs and donor sites healed well with good cosmesis achieved. One STSG had partial necrosis, this had been sutured directly to auricular cartilage where there was subtotal loss of the perichondrium.
The head and neck accounts for 85% of Basal Cell Carcinomas (BCC), high risk sites include the nose, ear, eyelid, eyebrow and temple [7]. A longitudinal study in Queensland looking at the anatomical location of BCCs found the following incidence rates per 1,000: nose 5.2, forehead/ temple 3.0, cheek/perioral 2.8, ears 1.8, neck 1.7, eyes 1.6, chin/jaw 0.6 and scalp 0.2 [8].
Skin cancers located on the anti-helix, crura of the anti-helix, scaphoid fossa, triangular fossa, anti-tragus and the posterior surface of the ear are known collectively as the ‘non helical rim’ ear [9]. Non helical rim wounds may be reconstructed with a Full Thickness Skin Graft (FTSG) or STSG, flap or secondary intent healing. Helical rim reconstructive options include; advancement flap, transposition flap or FTSG. Defects affecting the anti-helix and conchal bowl may be allowed to heal by secondary intent. If more rapid wound healing is desirable a STSG may be used to cover the thin skin of the anti-helix and conchal bowl. The two stage post auricular interpolation flap (PAIF) may be used for large anti-helical defects with conchal bowl defects [10].
Three types of flaps are commonly used in auricular reconstruction: advancement, transposition and interpolation. Chondrocutaneous advancement flap can be used to reconstruct helical rim defects [11]. The Banner transposition flap may be useful in superior helical rim defects however it is considered to be less useful nearer the root of the helix [12].
Moderately sized defects of the mid and inferior helical rim may be closed by a Helical Advancement Flap (HAF). HAF may be unilateral or may be bilateral for larger and more superiorly located defects. A superior helical rim wound may be repaired by a V-Y advancement from the anterior helix. A wedge closure should be reserved for small wounds located on the mid or inferior helix with full thickness cartilage loss [10].
Transposition flaps are used to repair anterior or superior helical defects, they are based at the anterior root of the helix and use either preauricular or post-auricular donor sites [10]. The bilobed flap has been used to transpose skin from the post auricular surface to cover a full thickness wound defect of the helical rim and posterior ear [13].A PAIF is used for helical rim reconstruction particularly for broad defects where HAF cannot be used. This two stage flap is well vascularised and therefore can be thinned considerably to match the recipient skin [10].
FTSG consist of the epidermis and dermis. They are considered to be easy to harvest and secure to the recipient site. FTSG have a higher metabolic demand and are more prone to necrosis than STSG but contract less. FTSGs are useful for repairs involving the ear, nasal tip, dorsum, ala and sidewall and periocular skin. Common donor sites for facial wounds include pre-auricular, post-auricular, supraclavicular and clavicular areas. When choosing a donor site it is important to take into consideration texture, thickness, colour and actinic damage of the recipient skin in choosing the donor site [14]. Placing a FTSG on cartilage without perichondrium may result in graft failure, however fenestrating the cartilage may improve graft survival. Leibovitch et al. [15] described short-term recipient site complications in FTSGs and found that the complication rate for the auricle was 5% compared to 11.8% for the nose and 15.6% for the periocular area.
A FTSG may be too thick for the thin skin of the non helical rim and a STSG may be preferable. Hendi et al. [9] used a Weck blade to produce an STSG harvested from the mastoid area in order to cover a defect on the anti-helix with excellent cosmesis.
Hochwalt et al. [16] compared outcomes of 18 patients who had FTSG and 29 patients who had secondary intent healing on Mohs defects of the helix. The primary aim was to compare outcomes of FTSG vs secondary intent wound healing assessed by a Visual Analog Score (VAS). The VAS was higher in the secondary intent healing group (82.1) compared to the FTSG group (75.2) but this was not statistically significantly different. Patient reported outcomes were not significantly different between the groups. The complication rates were also similar and although reported pain seemed to be equal between the two groups there did seem to be a trend for increased pain in the secondary intent healing group. Time to complete healing was similar between the two groups, as was the cosmetic outcome scored between 1 to 10 by the patients. Secondary intent healing is commonly employed for defects involving the conchal bowl and antihelix but not so often for the helical rim due to the risk of a depressed scar. In this study the authors found that the cosmetic outcome for secondary intent healing was equivalent to FTSG.
STSG has advantages compared to FTSG which includes plentiful donor site availability and therefore can provide wound coverage to large wounds. STSGs can also be meshed to enable greater surface coverage and allow the drainage of blood and exudates underneath the graft. STSGs have a reduced metabolic demand and therefore have an increased chance of graft take which may be important with regards to ear cartilage particularly where perichondrium has been partially excised and at sites which have a less robust vascular base. There are also disadvantages to using a STSG, which includes graft contraction, hyper or hypo pigmentation of either the donor site, recipient site or both as well as donor site morbidity [14].
The smallest STSG is the pinch graft, which requires forceps, a skin hook, a needle tip and a scalpel blade or double edged razor blade. Other freehand STSG harvesting methods include a disposable curette and scalpel. Hand operated dermatomes for STSG harvesting include the Weck knife, for smaller STSG, and the Humby knife for larger STSGs. These are largely operator and experience dependent. Electric dermatomes such as Brown, Zimmer and Padgett may be easier to operate and may be easier to produce uniform thickness STSG [17].
The ‘score and shave’ method of harvesting STSGs is a freehand scalpel harvesting technique. This technique has a number of benefits compared to other STSG harvesting techniques. These include a shorter operating time and smaller donor site wound, which matches the defect size exactly. Furthermore there is no requirement for specialist equipment. Snow et al. [4] described this technique to harvest skin for 65 patients with facial defects affecting the nose, ear, forehead, 19 patients had ear defects. The authors found that the skin from the pre-auricular region provided the best match for skin on the ear, nose and forehead due to the similarity in texture and sebaceous content. The authors suggested that the preauricular area is a favourable site to harvest a STSG because it can be done in a time efficient manner, is easy to shape, provides a good cosmetic match and has good success rates [4]. Hexsel et al. [18] found that the post-auricular region was also a good donor site. The authors reported 39 patients with 41 STSG and donor sites, the grafts were harvested by using a PersonnaTM Double Edge flexible blade taken from the post auricular area. All the skin grafts were successful on the head, neck and upper chest and the cosmesis of the post-auricular donor sites were acceptable as illustrated by the range of Vancouver scar scales being between 1.6 to 3.
Traditional use of STSG harvesting with a dermatome may take as long as FTSG harvesting, whereas score and shave harvesting of STSG is more time efficient and convenient for the patient. The technique allows for production of a STSG of the exact size, a well defined donor site of even depth, rapid healing of a small donor site, and there is no requirement for specialist equipment. We have had favourable results with 11 patients, all of who had a good cosmetic outcome. This technique is less invasive than harvesting a traditional STSG and results in a better graft take than FTSG, where cartilage is exposed and perichondrium may not be intact.
Download Provisional PDF Here
Aritcle Type: Research Article
Citation: Jayasekera PSA, Oliphant T, Langtry JAA (2017) Score and Shave Scalpel Harvesting of Split Thickness Skin Grafts in Reconstruction of the Ear, Finger and Toe. J Clin Cosmet Dermatol 2(1): http://dx.doi.org/10.16966/2576-2826.122
Copyright: © 2017 Jayasekera PSA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access