
Figure 1: Image showing centrofacial pattern of melasma involvement


Dinko Kaliterna1* Kristina Zuzul2 Irena Kovacevic3
1Polyclinic Poliderma, dermatovenerolog, Vukovar Street (Zagreb), Croatia*Corresponding author: Dinko Kaliterna, dermatovenerolog, Polyclinic Poliderma, Vukovar Street (Zagreb), 284, 10000, Croatia, Tel: 4828-961; E-mail: poliderma@poliderma.hr
Melasma is one of the most common pigmentary disorders characterized by symmetrical pigmented macules on the face. It most commonly affects women of reproductive age with darker complexions, and has a considerable impact on quality of life. The pathogenesis of melasma is complex and involves various risk factors such as genetic predisposition, UV exposure, darker phototype and hormonal influence. Because of its chronic and relapsing course, as well as the fact that despite numerous treatment modalities available, no single treatment option has proven to be efficient for all patients with melasma, the treatment still remains a challenge. This paper provides a review of the current literature on melasma treatment and presents the results of a study assessing a new promising resorcinol-based topical formulation with better efficacy and safety profile than other currently available treatment options for melasma.
Melasma; Resorcinol; Pigmentary Disorders; Melasma Treatment
Melasma (from the Greek word “melas” meaning black) [1] is an acquired hyperpigmentation of the skin, typically affecting the sunexposed areas of the face, occasionally occurring on the neck, and rarely on the forearms. The term, “chloasma” (from the Greek word “chloazein” meaning green) is used to describe melasma developing during pregnancy (but since the pigmentation is never green, the term, “melasma” is preferred) [2].
It presents clinically as symmetrical hyperpigmented macules on the face (the color varies from light brown to bluish gray depending on the depth of melanin deposition). There are usually three patterns of involvement: centrofacial (involves the cheeks, forehead, upper lip, nose, chin (Figure 1)), malar (involves the cheeks and nose), and mandibular (involves the mandible) [3].

Figure 1: Image showing centrofacial pattern of melasma involvement
It is a common disorder affecting millions of people worldwide [4], with the exact prevalence varying between 1.5% and 33.3%, depending on the population. Melasma, most commonly affects women of reproductive age with darker complexions (Fitzpatrick skin type IV-VI), although it can also be seen in men. Among pregnant women, prevalence can be up to 70% [5]. Melasma of the pregnancy usually improves a year after delivery, but areas of hyper pigmentation may never completely resolve [6]. Because of its facial involvement, melasma has a considerable impact on the quality of life of the affected individuals.
The pathogenesis of melasma is complex and still not well understood. Known risk factors include genetic predisposition, ultraviolet (UV) radiation exposure, darker phototype, hormonal influence (pregnancy, oral contraceptives, increased levels of luteinizing hormone, thyroid disease), and certain medications such as phenytoin [5,7]. It is known that UV radiation induces melanocyte proliferation. Although the number of melanocytes is similar in lesional and perilesional skin, melasma may be caused by biologically more active melanocytes in the affected skin [8].
In melasma lesions, there are increased stem cell factor from fibroblast and tyrosine kinase receptor c-kit [9], as well as the expression of vascular endothelial growth factor (VEGF). Direct role of VEGF in pathogenesis of melasma is supported by the observation of increased number and size of blood vessels in melasma lesions (Figure 2), as well as by the finding that human melanocytes in vitro express VEGF receptors [10,11]. This warrants investigation of new treatment options to target the vascular component of melasma [5].
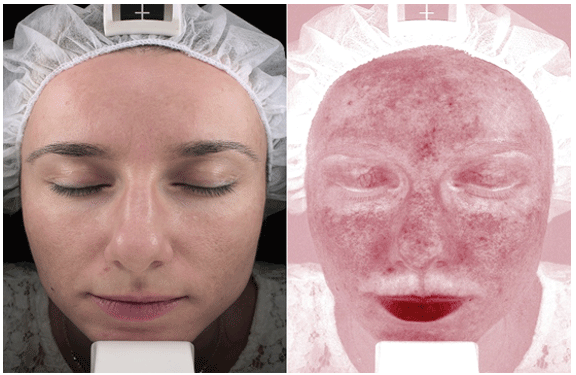
Figure 2: Image using VISIA analysis-in the areas of hyper pigmentations, there is increased number and size of blood vessels
Histopathology of melasma lesion shows an increased melanin deposition in all layers of epidermis, with elastosis and mast cells more pronounced than in normal skin [12]. The most affected biological process is lipid metabolism [8] there is down regulation of various lipid metabolism genes caused by chronic UV exposure.
Melasma is traditionally classified according to the depth of distribution of melanin pigment in three types [5]:
Examination with a Wood’s lamp can be used to determine the location of the pigment, but is limited in detecting dermal melanin composition [4]. The severity of melasma may be assessed by using melasma area and severity index (MASI) or Melasma Severity Index (MSI) [13].
A study was conducted in a Clinic in Zagreb according to the protocol and in accordance with ethical guidelines. The study included 30 female volunteers of 25 to 45 years of age, diagnosed with melasma. The volunteers were either treatment naïve or had not used any treatment modality for melasma for at least 6 months. The exclusion criteria included any treatment for melasma within past 6 months, hypersensitivity to resorcinol or any other ingredient of the formulation, pregnancy, and any type of facial skin damage, disease, or infection.
The volunteers were given a cream (Name of the cream: My skin by Dr. Kaliterna-White skin) based on resorcinol derivative at a baseline visit, and were instructed to apply a small amount of cream once daily on a clean face at bedtime, during a period of 30 days. They were also instructed to avoid sun exposure and apply sunscreens.
The efficacy of the treatment was done using the VISIA image analysis. The images were taken before the treatment, and again after 30 days.
A total of 30 volunteers were included in a study. No subjects were lost to follow-up, and all of them reported compliance to the protocol.
27 out of 30 subjects reported satisfaction with the results. Of those, 20 were extremely satisfied with results, and 7 were very satisfied. Aside from significant reduction in hyper pigmentation, they reported improved skin quality and texture, as well as reduction in skin pores. 2 out of 30 patients reported moderate satisfaction with results, while 1 patient reported that she is not satisfied.
No severe side effects were reported. Only 2 volunteers reported irritation or burning sensation during a few days after cream application, but spontaneous resolution of symptoms after a few minutes. Most of the clients reported first visible change and improvement between days 7 and 10. (Figures 3A and 3B).
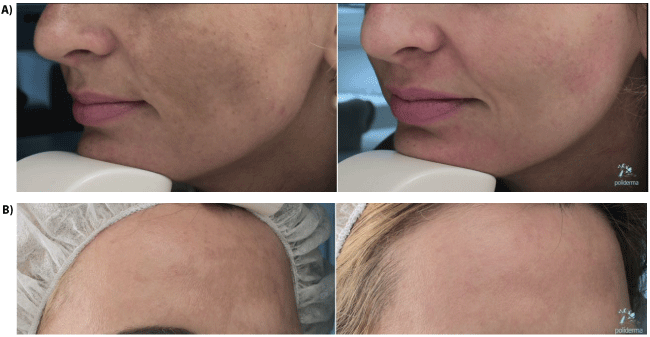
Figure 3: After 30 days before and after only with white skin 1 cream
Because of the chronic and relapsing nature of melasma, as well as the fact that no single treatment option has proven to be efficient for all patients with melasma, the treatment may be challenging. Before choosing the treatment modality, analysis of the characteristics of melasma lesion is recommended. This can be performed by VISIA or other similar systems. The area most responsive to treatment is the forehead, while other locations and older lesions seem to be more refractory. Current treatment options include the following:
4-n-Butylresorcinol is a resorcinol derivative that inhibits both tyrosinase and tyrosinase-related protein-1 (TRP-1) [24]. Its hypopigmenting action was first reported in 1995, and many following studies have documented its efficacy [25] and safety in melasma treatment with the 0.1% cream, but there is paucity of clinical studies that used the 0.3% cream. The biochemical assay on inhibition of human tyrosinase activity has revealed the superiority of 4-n-butylresorcinol over other hypopigmenting agents [26]. Many studies show good efficacy and safety in treating melasma [26,27].
Despite numerous treatment modalities available, due to its chronic and relapsing course, treating melasma still remains a challenge. Although hydroquinone and triple combination creams remain the gold standard of treatment, long term safety concerns and possible side effects warrant investigation of new, more efficacious and safer treatment modalities.
The investigated resorcinol derivative-based cream has shown the fastest onset of action (as measured by first reported improvements after 7-10 days) among all currently available topical preparations. Unlike other available topical preparations, this cream can be used throughout the year (including summer), the period of application is not limited (can be used continuously), and there are no pronounced side-effects (such as erythema or desquamation). In majority of cases, its effectiveness is better, and the results seen faster than with fractional laser. Its usage during pregnancy is not recommended, since no safety studies in this population were made.
Aside for its main purpose-the reduction in hyper pigmentation, it has shown a strong rejuvenation effect. Accordingly, other indications for using this cream include premature signs of aging and sun damage, such as excessive redness, large pores, or uneven texture.
Because of its fast onset of action, once-a-day application, and a wide range of indications, this promising product may be a revolution both in treatment of melasma and in skin rejuvenation.
Download Provisional PDF Here
Aritcle Type: Research Article
Citation: Kaliterna D, Kristina Zuzul, Kovacevic I (2017) Melasma-Review of Current Treatment Modalities and Efficacy Assessment of a New Resorcinol-Based Topical Formulation. J Clin Cosmet Dermatol 1(3): doi http://dx.doi.org/10.16966/2576-2826.116
Copyright: © 2017 Kaliterna D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access