Introduction
Pruritus (itch) is an unpleasant skin sensation, which induces an impulse to scratch [1,2]. Itch sensation is generated by the binding of itch inducing substances (pruritogens) to their cognate receptors (pruriceptors) on peripheral sensory afferents, especially C-fibers. The evoked action potential is transmitted through the ascending sensory pathway to the somatosensory cortex, resulting in the perception of itch (Figure 1). Because histamine is a well-known pruritogen, antihistamines (histamine H1 receptor antagonists) are the first line of treatment against itch. However, antihistamines are not fully effective in some dermatological conditions, such as xerosis, atopic dermatitis (AD) and psoriasis, and systemic diseases, such as chronic renal failure and chronic cholestasis, characterized by dry skin. Therefore dry skin is thought to be an important feature of antihistamine-resistant (histamine-independent) itch.
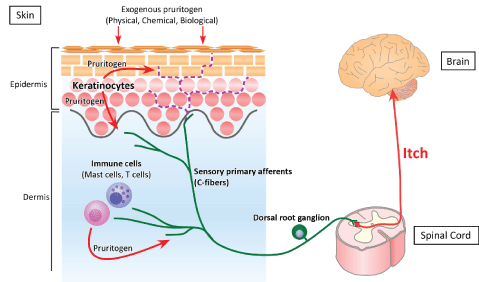
Figure 1: Pathway of itch. The perception of itch starts when itchinducing substances (pruritogens) bind to their receptors (pruriceptors) on peripheral sensory afferents. Exogenous physical, chemical and biological pruritogens can stimulate cutaneous nerve fibers, inducing itch. In addition, endogenous pruritogens may be produced by epidermal keratinocytes and dermal immune cells, such as mast cells and T cells. The signal following the binding of pruritogen to pruriceptor is transmitted through peripheral sensory afferents to the spinal cord and the somatosensory cortex, resulting in recognition of itch. Cutaneous nerve fibers usually terminate under the dermoepidermal junction (green line). Increased intraepidermal nerve density has been observed in the skin of patients with pruritic dermatologic diseases (purple dotted line).
The skin is the largest organ of the body, covering the body and providing the first physiological barrier to the external environment. Skin not only prevents damage from adverse external factors, acting as an outside to inside barrier, but loss of moisture and nutrients, acting as an inside to outside barrier. Therefore, loss of skin barrier integrity enables essential internal water to evaporate from the skin, leading to skin dryness. Skin barrier disruption by tape-stripping or treatment with acetone, diethyl ether and water (AEW) has been found to enhance dry skin characteristics, including increased transepidermal water loss (TEWL) and decreased hydration of the stratum corneum (SC) [3-5]. Following the induction of dry skin, various types of pruritogens and pruritogen-associated factors fluctuate in cutaneous cells [6-8]. Notably, in comparison with animal models involving acute barrier disruption by tape stripping or single acetone treatment, animals subjected to continuous AEW treatment showed increased induction of scratching behavior, with the latter depending on the number of treatments [4,9]. This AEW model is also characterized by abnormal itch sensations, called alloknesis (itch induced by non-itchy stimuli) and hyperknesis (enhanced itch induced by itchy stimuli) [9-11].
This review summarizes current knowledge regarding pruritogens, pruriceptors and modulatory mechanisms related to dry skin-induced itch. Further, it describes possible treatments based on the mechanisms of dry skin-induced itch. This information may enhance understanding of dry skin-induced itch.
Itch Inducing Substances and Receptors
Histamine: Histamine is a well-known pruritogen and a cause of urticaria, insect bite reactions, and nettle rash. Histamine is produced by cutaneous cells, including mast cells, basophils, and keratinocytes, and by neurons [12]. Histamine binds to four receptors, called the histamine H1 (H1 R), H2 (H2 R), H3 (H3 R) and H4 (H4 R) receptors, which mediate the effects of histamine, including inflammatory and immediate hypersensitivity responses [12,13]. At present H1 R and H4 R are considered therapeutic targets for pruritic diseases.
Low environmental humidity has been found to induce mast cell hypertrophy and degranulation, suggesting that histamine is associated, at least in part, to itch in winter xerosis [14]. Cytoplasmic granules of mast cells, however, contain not only histamine but other pruritogens, including serotonin, leukotriene B4 and several proteases, suggesting that histamine is not the only pruritogen involved in xerosis. Experimentally, repetitive application of AEW or the surfactant sodium dodecyl sulfate to mouse skin has been found to gradually induce skin dryness and increase scratching behavior [4,15]. Because the numbers of cutaneous mast cells were similar in AEW-treated and control mice, and because application of AEW or surfactant to the skin of mast cell deficient mice increased scratching behavior, mast-cell derived histamine likely did not play a key role in dry skin induced-itch. Histamine and L-histidine decarboxylase, a key enzyme involved in the endogenous production of histamine, were increased in the epidermis but not in the dermis of mice repeatedly treated with surfactant [16], suggesting that histamine derived from epidermal keratinocytes but not from dermal mast cells may have relevance for dry skin-induced itch.
Mas-related G protein-coupled receptor family: The Mas-related G protein-coupled receptor (Mrgpr) family in mice consists of more than 50 members, which can be grouped into several subfamilies: MrgprA, MrgprB, MrgprC, and MrgprD-G. MrgprX is a member of the Mrgpr family in humans [17]. Mice lacking a cluster of Mrgpr genes were found to display significant deficits in itch induced by chloroquine (CQ), a drug used in the management of malaria, but not by histamine, suggesting that this gene family is involved in histamine-independent itch [18]. MrgprA3, MrgprC11, and MrgprD in mice and MrgprX1, MrgprX2, and MrgprD in humans, which are expressed only on small-diameter sensory neurons in the DRG and trigeminal ganglia, were recently suggested to be involved in the transmission of itch [18-21].
The expression of mRNAs encoding MrgprA3 and MrgprC11 was found to be higher in AEW treated dry skin model mice than in water treated controls [22]. Moreover, the ablation of MrgprA3+ DRG neurons not only reduced acute itch induced by CQ injection but chronic itch induced by AEW treatment [23], suggesting that MrgprA3 and MrgprC11 are involved in dry skin-related itch. The increases in expression of MrgprA3 and MrgprC11 were inhibited in acid-sensing ion channel 3 (ASIC3) KO mice [22], suggesting that fluctuations in skin pH may be involved in dry skin-related itch.
Transient receptor potential family: Members of the transient receptor potential (TRP) family are known as polymodal cellular sensors. Mammalian TRP channels consist of 28 members and are categorized into six subfamilies: TRPC (canonical), TRPV (vanilloid), TRPM (melastatin), TRPP (polycystin), TRPML (mucolipin), and TRPA (ankyrin) [24]. Temperature-sensitive TRPs, especially TRPV1, and TRPA1, are among the most interesting itch-related receptors.
TRPV1 is expressed on cutaneous cells, including sensory nerve fibers, mast cells, epidermal keratinocytes, and dermal endothelial cells [25,26]. TRPV1 is activated by numerous stimuli and endogenous substances, including noxious heat (with a thermal threshold of >43°C), pH, bradykinin, ATP, lipoxygenase products, various neurotrophins, tumor necrosis factor (TNF)-α, and proinflammatory chemokines [24]. Recent studies have indicated that TRPV1+ neurons include itch-transmitting sub populations [27] and that histamine-induced inward currents in sensory neurons can be blocked by TRPV1 antagonists [28]. Therefore, TRPV1+ neurons are mainly involved in histamine-dependent itch.
TRPA1 is expressed on cutaneous nerve fibers, keratinocytes, and fibroblasts [29,30]. TRPA1 is highly expressed on a subset of TRPV1+ DRG neurons and is activated by environmental chemicals, such as allyl isothiocyanate, cinnamaldehyde, and allicin, and endogenous inflammatory agents. Sensory neurons from TRPA1-deficient mice displayed normal histamine-evoked responses but markedly diminished responses to CQ and bovine adrenal medulla peptide (BAM) 8-22, a MrgprC11 agonist [31]. These findings suggested that TRPA1 is a downstream target of both MrgprA3 and MrgprC11 and that TRPA1 at least partly played a key role in histamine-independent itch.
The levels of expression of TRPA1, MrgprA3 and MrgprC11 mRNAs were found to be higher in AEW- than in water-treated skin [22]. Moreover, TRPA1-deficient mice showed a marked reduction in scratching behavior induced by repetitive AEW treatment [32]. These findings suggested that TRPA1 may regulate itch in chronic dry skin.
Protease-activated receptors: To date, four protease-activated receptors (PARs) have been described, PAR-1, PAR-2, PAR-3, and PAR- 4, of which all except PAR-3 are expressed in cutaneous nerve fibers, keratinocytes, mast cells and macrophages and are considered involved in itch [33-35]. Endogenous activators of PARs include trypsin, papain, and kallikreins produced by keratinocytes, mast cells, macrophages, dendritic cells, B cells, T cells, and neutrophils, as well as by external sources, including mites, fungi, and plants [35]. Agents that activate PAR- 1, PAR-2, and PAR-4 (TFLLR, SLIGRL, and AYPGKF, respectively) were shown to increase intracellular calcium levels in DRG neurons [36]. Since antihistamines inhibit itch evoked by PAR-1 and PAR-4 agonist but not by PAR-2 agonist, PAR-2 is thought to be involved in histamine-independent itch [37]. On the other hand, trypsin induced scratching was suppressed by TRPV1 inhibition or in TRPV1-/- mice, which is involved in histaminedependent itch [38]. Recently, SLIGRL was reported to activate PAR-2 and MrgprC11, inducing scratching behavior in mice [39]. A shorter peptide, SLIGR, activated PAR-2 but not MrgprC11 and caused thermal pain hypersensitivity, suggesting that PAR-2 activation may be involved in neuronal sensitization rather than evocation of itch.
Spontaneous scratching in chronic dry skin model mice was significantly attenuated by an anti-PAR-2 antibody. Moreover, PAR-2 agonist enhanced scratching (hyperknesis) in AEW mice [9], suggesting that PAR-2 is at least partly involved in dry skin-related itch sensation.
Thymic stromal lymphopoietin (TSLP): Several cytokines also evoke itch sensation [40]. Thymic stromal lymphopoietin (TSLP) is produced by keratinocytes, mast cells, and dendritic cells [41,42]. The TSLP receptor (TSLPR) is a heterodimer that consists of the interleukin (IL)-7 receptor α chain and TSLPR [43]. Responsiveness to TSLP has been reported in dendritic cells, CD4+ and CD8+ T cells, B cells, mast cells, basophils, eosinophils, and natural killer T (NKT) cells [44]. The over expression of TSLP in keratinocytes in the epidermis of AD patients and in tapestripped barrier-disrupted skin suggested that TSLP may be a critical factor linking responses at interfaces between the body and the external environment [6,45,46]. Intradermal injection of TSLP into the cheeks of wild type mice evoked scratching bouts that were not observed following vehicle injection [46], suggesting that TSLP induces dry skin-induced itch. TSLP-evoked neuronal activation was attenuated in TRPA1-deficient but not TRPV1-deficient DRG neurons, suggesting that TRPA1 mediates TSLP-evoked neuronal excitability [46].
Acetylcholine receptor: Acetylcholine (ACh) is a neurotransmitter that is released from cholinergic nerves. Human keratinocytes and primary sensory neurons in the skin produce and secrete Ach [47-49]. Intradermal injection of cholinergic agonists in mice was reported to induce scratching behavior via M3 muscarinic ACh receptors [50]. Keratinocytes and DRG neurons express muscarinic M3 receptors and show increased intracellular calcium levels in response to cholinergic agonists [51,52]. ACh is generally considered an algesiogenic agent; intracutaneous injection of 500 mM ACh into healthy controls increased blood flow, and caused flares, wheals and short-lasting burning pain without any itching sensations [53,54]. In contrast, intradermal micro dialysis of 50 mM ACh induced burning pruritus [55]. Symptoms of dry skin in a mouse model of dextran sulfate sodium (DSS)-induced colitis were suppressed by the muscarinic cholinoceptor antagonist atropine, but not by the nicotinic cholinergic ganglion-blocking agent hexamethonium [56]. Although it is unclear whether dry skin in mice with DSS-induced colitis is accompanied by itch, muscarinic ACh receptor may be involved in dry skin-induced itch.
Toll-like receptors: Toll-like receptors (TLRs) are cellular sensors that recognize molecular danger signals associated with exogenous and endogenous threats. TLR3, TLR4 and TLR7 were shown to be expressed in primary sensory neurons, keratinocytes and mast cells, and are considered involved in itch [11,57-59]. Tlr3-/- mice showed markedly reductions in scratching behaviors induced by both histamine-dependent and -independent pruritogens [58]. In contrast, Tlr7-/- mice showed suppression of scratching behavior induced by histamine-independent, but not by histamine-dependent, pruritogens [57]. TLR3 partially colocalizes with TLR7, with both expressed in TRPV1 neurons. Recently, TLR4 was shown to be involved in itch associated with experimental dry skin. Tlr4-/- mice exhibited normal acute itch induced by both histaminedependent and -independent pruritogens, but not chronic itch induced by AEW treatment [11]. Interestingly, AEW treatment increased TLR4 expression in glial fibrillary acidic protein (GFAP)-expressing astrocytes in the spinal dorsal horn. As mentioned below, glial cells are involved in itch processing. Tlr4-/- mice and intrathecal administration of TLR4 antagonist (lipopolysaccharide R. sphaeroides) significantly reduced alloknesis induced by repetitive AEW treatment. Moreover, wearing of an Elizabethan collar that prevented scratching blocked astrogliosis in mice, which may express TLR4, suggesting that TLR4 may be involved in alloknesis under chronic dry skin conditions.
Opioid systems: The three subtypes of opioid receptors, µ, κ, and δ, as well as opioid receptor like-1 (ORL1) receptor, are located in both the peripheral and central nervous systems. Morphine, a μ-opioid receptor (MOR) agonist, has been used as an analgesic for centuries. Generalized itch may accompany systemic administration of morphine. Previous studies have shown that activation of the MOR and ORL1 receptors causes pruritus, whereas activation of the κ-opioid receptor (KOR) suppresses itch [1,60].
Spontaneous scratching after AEW treatment was significantly suppressed by subcutaneous injection of MOR antagonists, such as naloxone and naltrexone [4], and the KOR agonist nalfurafine [61]. The latter agent also inhibited alloknesis in AEW mice [62].
Distribution of Cutaneous Nerve Fibers
In healthy skin, most cutaneous nerve fibers terminate under dermoepidermal junctions. Increased intraepidermal nerve density has been observed in the skin of patients with pruritic dermatologic diseases, such as senile xerosis, AD and psoriasis [63-66], as well as dry skin model mice [5,67] (Figure 1). The controlling mechanism of cutaneous nerve density is thought to involve the expression balance of axonal guidance molecules by epidermal keratinocytes [7] (Figure 2). Increased nerve density is induced by dominant expression of nerve elongation factors (NEF; nerve growth factor [NGF] etc.). Conversely, dominant expression of nerve repulsion factors (NRF; semaphorin 3A [Sema3A] etc.) reduce the nerve density. Compared with the expression balance in healthy controls, the dominant expression in NEF is observed in barrier disrupted skin such as dry skin and AD [5,67-69]. Therefore the imbalance of axonal guidance molecules and increased intraepidermal nerve density may be a physiological marker of the disruption of barriers required to maintain homeostasis.
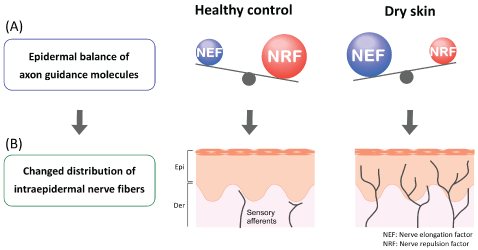
Figure 2: (A) Epidermal balance of the expression of axon guidance molecules. In healthy skin, nerve repulsion factor (NRF) is dominant over nerve elongation factor (NEF). In dry skin, however, NEF is dominant. (B) Changed distribution of intraepidermal nerve fibers. In healthy skin, with NRF dominant, cutaneous nerve fibers remain under the dermo epidermal junction. In dry skin, with NEF dominant, cutaneous nerve fibers elongate into the epidermis. Epi; epidermis, Der; dermis
In contrast, decreased nerve density has been observed in patients with AD and prurigo nodularis [70,71], returning to normal levels during healing of patients with prurigo nodularis [72]. Intraepidermal nerve fiber density is also reduced in patients with small fiber neuropathy, with 60% of these patients having pruritus [73,74]. Although the mechanisms linking changes in intraepidermal nerve density with itch remain unknown, altered nerve distribution may be an important factor in the development and/or sensitization of itch.
Transmission and Modulation of Itch
Itch intensity is influenced not only by the concentrations of pruritogens and pruriceptors but by the functions of nerve fibers and glial cells in the ascending sensory pathway, similar to glial modulation of pain processing [1, 75, 76]. Increased expression of NGF not only induces nerve elongation but alters neural populations and receptor properties [77].
Pruritogen-evoked alloknesis has been reported following the intradermal injection of histamine, serotonin, BAM8-22 and PAR-4 agonist, but not after the injection of CQ and PAR-2 [62]. Alloknesis scores and spontaneous scratching bouts were higher in mice receiving repeat AEW treatment than in control mice [11,62]. Moreover following AEW treatments, the number of scratching bouts was significantly increased by intradermal injection of CQ and serotonin, but not histamine [9,66]. These pruritogens enhanced the activity of DRG neurons from these AEW mice, suggesting that this model also represents hyperknesis [9]. Abnormal itch sensations such as alloknesis and hyperknesis may result from neural sensitization (i.e., lowering of neural thresholds for their stimuli), but these abnormal sensations do not necessarily require neural sensitization under inflammatory conditions. Ongoing activation of C-fibers lowers the thresholds of secondary neurons in the spinal cord [1]. Therefore, although the leading cause of this phenomenon remains unclear, continuous AEW treatments may induce repetitive activation of C-fibers, inducing neural sensitization. In contrast, the spontaneous activity of DRG neurons from AEW- and water-treated mice did not differ significantly [66]. In this study, AEW treatment increased Ret-positive but not calcitonin gene-related peptide (CGRP)- or glial cell line-derived neurotrophic factor family receptor (GFR) α3-positive fibers, suggesting that hyperinnervation of nonpeptidergic fibers may contribute to dry skin-induced itch. Taken together, these findings indicate that dry skininduced itch may be caused not only by the concentrations of pruritogens and pruriceptors and the distribution of neurons, but also by functional changes in neurons of the ascending pathway.
Treatment of Dry Skin-Related Itch
Moisturizers that enhance skin barrier function: Moisturizers improve barrier function, by preventing excess TEWL and the entry of external pruritogens, which may help alleviate pruritus. Moisturizer application was found to effectively suppress the increased epidermal NGF expression observed after acetone-induced skin barrier disruption, with suppressed NGF expression correlating with a reduction in intraepidermal nerve density [67]. These effects depended on the time between skin barrier disruption and the application of emollient, suggesting a role for moisturizers in the rapid and temporary relief of barrier disruption. In daily usage, the preventive application of emollients (e.g., soon after showering, bathing or hand-washing) seems effective in maintaining healthy skin barrier function.
As the composition of moisturizers varies, moisturizers should be selected based on symptoms. Topically applied ointments mainly composed of lipids act as occlusive membranes, reducing the loss of water from the skin [78]. Products containing some water, such as emollients and creams, immediately hydrate the SC through absorption into the skin. Although water is important in maintaining skin flexibility, excess water may result in the deterioration of skin barrier function. Therefore adequate water supply for evaporation from the skin and occlusive for temporary lack of SC would likely be effective.
Antihistamines: Histamine induces not only pruritus but skin barrier impairment. Histamine application to skin culture inhibited terminal differentiation of keratinocytes, including loss of granular layer and reduced tight junction proteins [79,80]. Therefore, although antihistamines are insufficient for use against intractable itch, they may improve skin barrier function.
Local anesthetics: Local anesthetics, such as lidocaine, have been reported to have antipruritic effects, inhibiting the action potential of afferent nerves [81,82]. Therefore local anesthetics may be effective in the rapid disruption of the itch-scratch cycle. However, these effects are limited to the local area of anesthetic treatment and are reversible. To date, the effects of local anesthetics on epidermal imbalance of axon guidance molecules have not been determined. If these agents induce cutaneous hyperinnervation and inflammation, additional treatments may be needed to normalize the imbalance in expression of axon guidance molecules (e.g., emollients).
Opioids: Dry skin-related itch in animal models was suppressed by μ-opioid receptor antagonists and κ-opioid receptor agonists [4,61]. Clinically also, μ-opioid receptor antagonists and κ-opioid receptor agonists were found to inhibit itch in different dry skin-related systemic diseases [83,84].
Phototherapy: Phototherapies, such as psoralen-UVA (PUVA), UVA1, narrow band (NB)-UVB, and excimer lasers or lamps, have been reported effective in patients with intractable pruritus, such as AD [85]. UV-based therapies are thought to suppress inflammation [86,88], following the reduction of pruritogen production. Although their detailed mechanisms of action remain unknown, PUVA and NB-UVB normalize epidermal expression of axonal guidance molecules, such as NGF and Sema3A [89,90], as well as reducing the densities of epidermal and dermal nerve fibers [91]. Moreover, excimer lamp treatment has been shown to directly induce nerve fiber degeneration, suggesting that the excimer lamp has a direct effect on cutaneous nerve fibers [90].
Conclusion
A decline in skin barrier function is thought to be the primary cause of dry skin induced-itch. During the process of skin barrier recovery, however, fluctuating conditions in the skin may result in the continuous activation of C-fibers, leading to neural sensitization and the itch-scratch cycle. Treatment should therefore include aggressive prevention by emollients, or quick disruption of the itch-scratch cycle in the chronic state.
Conflicts of Interest
The authors declare that they have no competing interests.
Acknowledgements
This work was supported by JSPS KAKENHI (Grant numbers 15K19193) and Strategic Research Foundation Grant-aided Project for Private Universities from MEXT (Grant number S1311011).



