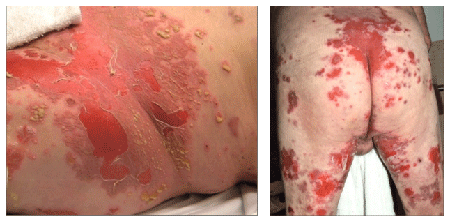
Figure 1: Multiple pustules and erosions of the groin, buttock and thighs.


IgG/IgA Pemphigus with Autoantibodies to Desmoglein 1 and Desmocollin 3, and Associated Lung Cancer
Bunimovich YL1* Teye K2 Ishii N2 Ho J1 Hashimoto T2 Patton TJ1
1Department of Dermatology, University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania, USA*Corresponding author: Yuri Bunimovich, MD, PhD, Department of Dermatology, University of Pittsburgh Medical Center, 3708 Fifth Avenue, Pittsburgh, PA 15213, USA, Tel: 412-647-4200; Fax: 412-864-3734; E-mail: bunimovichyl@upmc.edu
IgG/IgA pemphigus is a rare atypical immunobullous disease which may be a unique form of pemphigus. We describe a case of IgG/IgA pemphigus with IgG antibodies to desmoglein1and desmocollin 3, IgA antibodies to desmoglein 1, and associated metastaticnon-small cell lung cancer. Pemphigus was treated successfully with prednisone and dapsone. IgG/IgA pemphigus may follow an aggressive course and, based on a review of the literature, has a high prevalence of associated malignancy, necessitating early accurate diagnosis and treatment.
IgG/IgA pemphigus; Immunobullous; Paraneoplastic pemphigus; Desmoglein; Desmocollin
Pemphigus is an autoimmune bullous disorder characterized by autoantibodies to keratinocyte cell surface components. The two major types of pemphigus, pemphigus vulgaris (PV) and pemphigus foliaceus (PF), demonstrate IgG autoantibodies to desmoglein (Dsg) 3 and Dsg1, respectively. Other, less common forms of pemphigus, such as pemphigus herpetiformis (PH), IgA pemphigus and paraneoplastic pemphigus (PNP), are diagnosed based on their unique clinical, histopathological and immunological features [1].
IgG/IgA pemphigus is the name given to an atypical form of pemphigus characterized by cell surface deposits of IgG and IgA and acantholysis [1]. Based on approximately twenty reports of IgG/IgA pemphigus, the following conclusions can be drawn regarding this atypical form of pemphigus. The clinical features of the reported cases are quite heterogeneous, resembling either one of the established forms of pemphigus (PV, PF, PH, IgA pemphigus) or demonstrating atypical, mixed clinical findings [1]. While most of the reported cases describe vesicular and/or pustular cutaneous eruption, only four cases had mucosal involvement [1]. The histopathology of IgG/IgA pemphigus is also variable, and may exhibit acantholysis, subcorneal and intraepidermal blisters, and neutrophilic or eosinophilic infiltrate. The keratinocyte cell surface autoantigens of circulating IgG and IgA antibodies are primarily Dsg1 and Dsg3, but desmocollins (Dscs) have also been implicated in a minority of cases. Interestingly, in almost all cases of IgG/IgA pemphigus the autoantigen of IgG and IgA antibodies is the same. Approximately thirty percent of reported cases of IgG/IgA pemphigus were associated with malignancy such as lung cancer [2,3], ovarian [4] and gall bladder carcinoma [5], endometrial [6] and pancreatic adenocarcinoma [7].
A 65-year-old male with history of chronic obstructive pulmonary disease and emphysema presented after a recent hospitalization for an unspecified immunobullous disease. The patient initially noted several small pustules on the lower back. His condition progressed over two weeks to multiple pustules and erosions of the intertriginous areas, lower back and thighs (Figure 1). While hospitalized, he was treated with prednisone 1 mg/kg/day with significant but incomplete improvement.

Figure 1: Multiple pustules and erosions of the groin, buttock and thighs.
Histopathological analysis of a biopsy specimen of the skin lesion revealed subcorneal pustule, intraepidermal acantholysis and a mixed perivascular infiltrate in the superficial dermis (Figure 2 top). Direct immunofluorescence detected keratinocyte cell surface deposits of IgG and IgA. Enzyme-linked immunosorbent assays (ELISAs) performed with commercially available kits detected anti-Dsg1 antibodies (ELISA index value 38, cut-off<14) but not anti-Dsg3 antibodies, anti-BP230 antibodies or anti-BP180 antibodies.
The patient was diagnosed with IgG/IgA pemphigus and was initially treated with dapsone 75 mg/day with complete resolution of all skin lesions. One year later he began developing an increased number of blisters and erosions. Increasing dapsone to 200 mg/day only proved marginally effective, and prednisone 60 mg/day was added.
Additional serum analysis was performed at that time. Indirect immunofluorescence of normal human skin sections detected IgG and IgA anti-keratinocyte cell surface antibodies (Figure 2 bottom). Immunoblotting of patient’s serum IgG and IgA using normal human epidermal extract revealed no reactivity to PV and PNP antigens. By ELISAs using eukaryotic recombinant human Dsg1 and Dsg3, IgG antibodies reacted with Dsg1 (index 38, cut-off>20) but not Dsg3, and IgA antibodies reacted with Dsg1 (OD 0.227, cut-off>0.15) but not Dsg3. In addition, novel ELISAs for recombinant human Dsc1-3 revealed reactivity of IgG antibodies with Dsc3 (OD 0.974, cut-off<0.12) but not Dsc1 or Dsc2. Patient’s IgA antibodies did not react withDsc1, Dsc2 or Dsc3.
One year after the initial presentation the patient was diagnosed with poorly differentiated squamous cell carcinoma of the lung with metastases to the mediastinum, abdominal lymph nodes and the liver. He received three cycles of gemcitabine and carboplatin, while being maintained on 200 mg/day of dapsone and 20 mg/day of prednisone with absence of skin lesions.
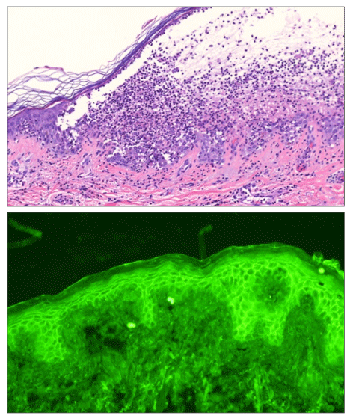
Figure 2: (Top) The biopsy showing subcorneal pustule, intraepidermal acantholysis and mixed perivascular infiltrate beneath the blister (H&E, x20). (Bottom) Indirect immunofluorescence of normal human skin sections demonstrating cell surface pattern of patient’s serum IgA antibodies.
We describe a case of atypical pemphigus, with overlapping clinical features of IgA pemphigus and PF. This case fits under the definition of IgG/IgA pemphigus as it demonstrated epidermal acantholysis and cell surface deposition of both IgG and IgA. We further detected IgG antibodies reactive with Dsg1 and Dsc3, and IgA antibodies reactive with Dsg1. The definition of IgG/IgA pemphigus is, however, somewhat general, and controversy exists as to whether it represents a separate disease entity [1]. The coexistence of both IgG and IgA autoantibodies to keratinocyte cell surface antigens does not appear to be unique to IgG/IgA pemphigus, as a study by Mentink et al. [8] retrospectively demonstrated the existence of IgA autoantibodies to Dsg1 and Dsg3 in patients previously diagnosed with PF, PV and PNP who also had IgG autoantibodies. Interestingly, the authors also observed that in the majority of cases IgG and IgA antibodies reacted with the same autoantigen [8]. The overlapping clinical features described in cases of IgG/IgA pemphigus suggest the existence of a spectrum of pemphigus diseases, from purely IgG-mediated to purely IgA-mediated as either extreme of such a spectrum.
Most of the cases of IgG/IgA pemphigus, including the present case, demonstrated IgG and IgA antibody reactivity with the same autoantigen. It is not clear whether both Ig isotypes recognize the same epitope on the desmogleins, and whether they are produced by the same plasma cell clone following an isotype switch. Such clonogenic rearrangement of the Ig heavy chain has been described for multiple myeloma, and may result in the emergence of an IgA clone from an IgG clone, and vice versa [9]. Such investigations have not, to our knowledge, been undertaken in the field of immunobullous diseases. Characterizing the mechanism which leads to the emergence of several auto-Ig isotypes in pemphigus may help better classify and diagnose this class of diseases.
Approximately one-third of IgG/IgA pemphigus cases described in the literature, as well as the present case, have been associated with malignancy [2-7]. This is in contrast to “pure” IgA pemphigus, in which paraproteinemias have been reported in up to 20% of cases, but lymphoproliferative and solid tumor malignancies are rare [10]. The association of IgG/IgA pemphigus with malignancy suggests that a comprehensive history should be obtained in all cases and cancer screening should be considered where appropriate, especially in cases with recalcitrant and progressive skin disease. In the above case, it is possible that the lung carcinoma was present at the initial presentation, but was only diagnosed at an advanced stage one year later.
The significance of the antibodies to two autoantigens (Dsg1 and Dsc3) and the pathogenic role of anti-Dsc3 antibodies are not well understood. Recent reports established that Dsc3 is a tumor suppressor and a marker of squamous carcinoma of the lungs [11,12]. Reduced expression of Dsc3 correlated with poor clinical outcome in patients with lung cancer [13]. The presence of anti-Dsc3 antibodies in the above case of IgG/IgA pemphigus with poorly differentiated squamous cell carcinoma of the lungs raises questions about a possible pathogenic role of anti-Dsc3 antibodies. Do autoantibodies to Dsc3 contribute to carcinogenesis or metastasis? Are anti-Dsc3 antibodies a marker of internal malignancy? Additional studies are required to better characterize atypical pemphigus cases with IgG and IgA autoantibodies and their associations with malignancy.
Download Provisional PDF Here
Aritcle Type: Case Report
Citation: Bunimovich YL, Teye K, Ishii N, Ho J, Hashimoto T, et al. (2016) IgG/IgA Pemphigus with Autoantibodies to Desmoglein 1 and Desmocollin 3, and Associated Lung Cancer. J Clin Cosmet Dermatol 1(1): doi http://dx.doi.org/10.16966/2576-2826.102
Copyright: © 2016 Bunimovich YL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access