Abstract
Background: Oxidative stress and inflammatory cytokines play an important role in the development of hypertension and Atrial Fibrillation (AF). Clinical studies have shown that serum Uric Acid (UA) levels are positively associated with hypertension and AF. Xanthine Oxidase (XO) is a key enzyme in UA metabolism. It can be a source of oxidative stress in AF. In this study, we investigated the role of XO in oxidative stress and inflammatory cytokines in UA-induced AF.
Methods: According to the inclusion and exclusion criteria, 60 patients hospitalized in the Xinjiang Uygur Autonomous Region People’s Hospital from June 2017 to June 2018 were selected as the research group. Thirty healthy subjects who completed physical examination in the same period comprised the healthy control group. All subjects underwent echocardiography. Fasting blood samples were collected, and clinical data, such as sex, age, course of disease, BMI, smoking, drinking, dyslipidemia, diabetes mellitus, and diuretics, were recorded in detail. ELISA method was used to analyze the circulatory levels of UA, XO, nicotinamide adenine dinucleotide phosphate oxidase-4 (Nox-4; a biomarker of oxidative stress), and inflammatory cytokines (high-sensitivity C-reactive protein [Hs-CRP], interleukin-1β [IL-1β], interleukin-8 [IL-8], and TNF-α).
Results: According to the clinical manifestations, electrophysiological examination, and diagnostic criteria of hypertension, the patients were divided into hypertension with AF group (AF group), hypertension without AF group (non-AF group), and healthy control group. The average age, body mass index, course of hypertension, blood pressure, hyperlipidemia, and diabetes mellitus in the AF group were higher than in the non-AF and control groups (P<0.05). In comparison with the non-AF and control groups, the serum levels of high-density lipoprotein cholesterol, triglycerides, and creatinine in the AF group were significantly increased (P<0.05), whereas the estimated glomerular filtration rate was significantly reduced (P<0.05). In the AF group, the blood concentrations of UA, XO, and Nox-4 significantly increased compared with those in the non-AF and control groups (P<0.001). The circulatory levels of inflammatory cytokines (Hs-CRP, IL-1β, IL-8, and TNF-α) significantly increased in the AF group (P<0.001). Pearson correlation analysis showed that the serum levels of XO in the AF group have a significant positive correlation with the blood concentrations of Nox-4 and inflammatory cytokines (Hs-CRP, IL-1β, IL-8, and TNF-α) (P<0.01) with correlation coefficients of r=0.99, 0.91, 0.96, 0.95, 0.97.
Conclusions: XO showed remarkable elevated ROS accumulation and inflammatory cytokines, there by resulting in induced AF formation in patients with hypertension.
Keywords
Oxidative stress; Uric acid; Xanthine oxidase; Atrial fibrillation; Inflammatory cytokines
Background
Atrial Fibrillation (AF) is the most common persistent arrhythmia, and its incidence increases with age. Hypertension is a major risk factor of AF. The common arrhythmia in hypertensive patients is caused by the fact that AF and hypertension promote each other, thereby creating a vicious circle that accelerates the progression of hypertension [1-2]. The mechanism underlying their occurrence and development has not yet been clarified.
Serum Uric Acid (SUA) is an important plasma marker in patients with Coronary Heart Disease (CHD) and hypertension, which is closely related to the morbidity and mortality of Cardio Vascular Diseases (CVD) [3]. UA in the body is the final product of purine base metabolism. It is produced by hypoxanthine and xanthine via purine synthesis under the catalysis of Xanthine Oxidase (XO) [4]. The increased enzymatic activity of XO can lead to excessive accumulation of Reactive Oxygen Species (ROS), eventually causing atrial remodeling and promoting AF’s occurrence [5]. The increase of SUA level reflects the increase of XO activity in mammals and humans [6]. ROS contributes to the pathological consequences of AF and hypertension [7]. AF is also associated with excessive ROS generation. The high concentrations of Nicotinamide Adenine Dinucleotide Phosphate (NADPH) oxidase-4 (Nox-4) is markedly correlated with acute myocardial infarction and AF [8]. Nox-4 is frequently used as a biomarker of oxidative stress [9]. However, the role Nox-4 in the occurrence of AF and hypertension has not been fully investigated.
Inflammation plays an important role in the development of AF. Inflammation has been implicated in the pathophysiology of AF and modulates the cell signaling activation patterns associated with fibrosis, apoptosis, and visceral hyper sensation [10]. The serum levels of tumor necrosis factor-α (TNF-α) and interleukin-8 (IL-8), which are inflammatory mediators, remarkably increase in patients with AF [11]. Elevated serum levels of inflammatory biomarkers in subjects with AF and expression of inflammatory markers in cardiac tissues of patients with AF suggest the substantial role of inflammation in AF’s pathogenesis [12]. Inflammation is linked to various pathological processes, such as oxidative stress, apoptosis, and fibrosis, which promote AF formation. The frequencies of AF episodes are substantially reduced and the symptoms of AF are ameliorated in patients with concomitant Hyperuricamia (HUA) after treatment with UA inhibitors [13].
Therefore, the effects of UA, ROS, and inflammatory cytokines on the occurrence of AF need further study. Our study aims to examine the expressions of UA, XO, ROS marker (Nox-4), and inflammatory cytokines (High-sensitivity C-reactive protein [Hs-CRP], IL-8, IL1β, and TNF-α) in the serum of patients with hypertension and AF. This work also aims to explore the correlations of XO-induced ROS production and inflammation and determine its influences on the occurrence of AF.
Methods
Patient characteristics
Sixty patients admitted to the Xinjiang Uygur Autonomous Region People’s Hospital from June 2017 to June 2018 were selected as the study group. Healthy individuals (n=30) who do not have CVDs were selected as the control group. All patients in the study group met the typical symptoms and diagnostic criteria of hypertension. All subjects were required to sign informed consent documents. Pregnant and lactating women were not included in this study. Fasting blood samples were collected and clinical data, such as sex, age, course of disease, BMI, smoking and drinking, dyslipidemia, diabetes mellitus, and diuretics, were recorded in detail.
Inclusion criteria
Patients who satisfied the following criteria were included in the study: (1) patients who fit the standard of hypertension diagnosis (refer to the diagnostic standard of hypertension expert consensus of China in 2010); (2) patients who fits the diagnostic criteria of AF recorded by electrocardiogram, 24h dynamic electrocardiogram, and echocardiography; (3) patients who have no acute/chronic infective or autoimmune diseases; and (4) patients who are willing to sign an informed consent and can cooperate to complete the study.
Exclusion criteria
The exclusion criteria were as follows: (1) patients who do not fit the hypertension diagnostic criteria; (2) patients with gout, autoimmune, or inflammatory disease; (3) patients who have a history of heart surgery, respiratory diseases, thyroid diseases, and use of drugs that affect UA metabolism; and (4) unwillingness to participate.
Sample collection
All patients were asked to sign an informed consent form. In the morning, 5 ml of fasting blood was collected from patients in each group for biological analysis. The serum was centrifuged (15 min, 3,500 × g, 4°C) and stored in a refrigerator at -80°C until use.
ELISA assays
Plasma samples were obtained from all groups and processed as previously described [14-15]. The concentrations of SUA, XO, High-Density Lipoprotein-Cholesterol (HDL-C), Low Density Lipoprotein-Cholesterol (LDL-C), Total Cholesterol (T-cho), Triglyceride (TG), and blood glucose were determined with the respective sensitive ELISA kits (R&D Systems, Minneapolis, MN). The numbers of White Blood Cells (WBC), albumin, globulin, potassium, and creatinine were measured using Beckmann Kurt Uni Cel DxH 800 five-class hematology analyzer (purchased from Beckmann Kurt Commercial China Co., Ltd). The plasma levels of ROS biomarker (Nox-4) and inflammatory cytokines (Hs-CRP, IL1β, IL-8, and TNF-α) were determined using a competitive ELISA kit (R&D Systems, Minneapolis, MN) following the manufacturer’s instructions.
Echocardiography
Left Atrial Diameter (LAD), Interventricular Septal Thickness (IVST), Left Ventricular Posterior Wall Thickness (LVPWT), Left Ventricular End-Diastolic Dimension (LVEDD), and Left Ventricular Ejection Fraction (LVEF) were measured by DW-T6 trans thoracic doppler echocardiography (Beckmann Kurt Commercial China Co., Ltd).
Statistical analysis
All data were expressed as mean ± SD. Student’s t-test was performed to measure the differences between the groups. Fisher’s protected least significant differences (PLSD) test of ANOVA was performed to analyze the quantitative data collected from the groups. A p value of <0.05 was used to denote significance.
Results
Patient demographics
According to the clinical manifestations, electrocardiogram, dynamic electrocardiogram, electrophysiological examination, and diagnostic criteria of hypertension, the patients were divided into hypertension with AF group (AF group), hypertension without AF group (non-AF group), and healthy control group (Table 1). The average age, BMI, course of hypertension, blood pressure, hyperlipidemia, and diabetes mellitus in the AF group were higher than those in the nonAF and control groups (P<0.05; Table 1).
| Characteristic |
Control (n=30) |
non-AF (n=30) |
AF (n=30) |
P value |
| Male, n (%) |
13 (43.3%) |
11 (36.7%) |
10 (33.3%) |
0.04 |
| Age |
36.45 ± 6.34 |
42.33 ± 7.87 |
48.67 ± 9.34a |
0.956 |
| BMI (kg/m2) |
19.56 ± 1.32 |
22.32 ± 1.88 |
24.54 ± 1.63a |
0.048 |
| Smoking, n (%) |
6 (20.0%) |
10 (33.3%) |
11 (36.7%) |
0.076 |
| Drinking, n (%) |
4 (13.3%) |
6 (20.0%) |
7 (23.3%) |
0.647 |
| Course of hypertension (years) |
0 |
12.22 ± 9.35a |
16.45 ± 7.33a |
0.014 |
| SBP (mmHg) |
118.6 ± 10.26 |
141.4 ± 15.34a |
148.8 ± 18.65a |
0.023 |
| DBP (mmHg) |
77.63 ± 5.62 |
85.52 ± 7.82a |
94.72 ± 9.23a,b |
0.031 |
| With HLD, n (%) |
0 (0%) |
10 (33.3%)a |
17 (56.6%)a,b |
0.015 |
| With DM, n (%) |
0 (0%) |
9 (30.0%)a |
18 (60.0%)a,b |
0.011 |
Table 1: Comparison of the general characteristics of patients in each group.
Values are expressed in comparison with the control group. Data are presented as mean ± SD. Differences among the groups were analyzed by Student’s t-test. a P<0.05 versus control group; b P<0.05 versus non-AF group.
Abbreviations: BMI: Body Mass Index; BP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HLD: Hyperlipidemia; DM: Diabetic Mellitus
Comparison of serum biochemicals among groups
In comparison with the non-AF and control groups, the serum levels of HDL-c, TG, and creatinine in the AF group were significantly increased (P<0.05), whereas the estimated glomerular filtration rate (eGFR) was significantly reduced (P<0.05), as shown in table 2. Other blood biochemical parameters in the AF group showed altered tendencies, but no significant difference was observed among the different groups (P>0.05).
| Characteristic |
Control (n=30) |
non-AF (n=30) |
AF (n=30) |
P value |
| WBC (×109/L) |
5.84 ± 1.23 |
6.32 ± 1.36 |
6.54 ± 1.42 |
0.623 |
| Albumin (g/L) |
30.31 ± 2.76 |
33.63 ± 3.53 |
35.72 ± 4.45 |
0.867 |
| Globulin (g/L) |
24.65 ± 1.97 |
25.74 ± 2.87 |
26.32 ± 3.43 |
0.962 |
| Potassium (mmol/L) |
4.05 ± 0.64 |
4.14 ± 0.65 |
4.23 ± 0.55 |
0.762 |
| T-cho (mmol/L) |
4.02 ± 0.45 |
4.36 ± 0.71 |
4.51 ± 0.84 |
0.431 |
| HDL-c (mmol/L) |
1.09 ± 0.13 |
1.21 ± 0.21 |
1.25 ± 0.32a |
0.034 |
| LDL-c (mmol/L) |
2.38 ± 0.27 |
2.31 ± 0.33 |
2.28 ± 0.54 |
0.287 |
| TG (mmol/L) |
1.38 ± 0.35 |
1.45 ± 0.46 |
1.93 ± 0.62a,b |
0.037 |
| Glucose (mmol/L) |
5.23 ± 0.35 |
5.37 ± 0.61 |
5.45 ± 0.58 |
0.126 |
| Creatinine (μmol/L) |
87.36 ± 6.75 |
94 ± 7.82 |
120.34 ± 15.24a,b |
0.01 |
| eGFR (mL/min/1.73m2) |
125.34 ± 20.45 |
116.57 ± 13.92 |
102.56 ± 12.75a |
0.018 |
Table 2: Comparison of serum biochemicals in each group.
Values are expressed relative to the control group. Data are presented as mean ± SD. Differences between the two groups were analyzed by Student’s t-test. a P<0.05 versus control group; b P<0.05 versus non-AF group.
Abbreviations: WBC: White Blood Cells; T-cho: Total Cholesterol; HDL-c: High Density Lipoprotein-Cholesterol; LDL-c: Low Density LipoproteinCholesterol; TG: Triglycerides; eGFR: Estimated Glomerular Filtration Rate
Comparison of echocardiographic analysis among groups
As shown in table 3, the echocardiographic parameters, such as LAD, IVST, LVPWT, and LVEF, in the AF group were significantly altered compared with those in the non-AF and control groups (P<0.05). No statistical significance was found in the comparison of LVEDD among the groups (P>0.05). UA, XO, and Nox-4 expression was elevated in AF.
| Characteristic |
Control (n=30) |
non-AF (n=30) |
AF (n=30) |
P value |
| IVST (mm) |
9.42 ± 1.25 |
9.73 ± 1.33 |
11.53 ± 1.67a |
0.041 |
| LVPWT (mm) |
8.62 ± 1.26 |
9.02 ± 1.43 |
9.76 ± 1.65 |
0.047 |
| LAD (mm) |
32.47 ± 1.38 |
35.63 ± 1.45 |
40.72 ± 1.71a |
0.032 |
| LVEDD (mm) |
44.21 ± 1.18 |
45.94 ± 1.48 |
47.66 ± 1.54 |
0.478 |
| LVEF (%) |
68.56 ± 5.66 |
52.43 ± 5.42a |
40.85 ± 6.45a,b |
0.017 |
Table 3: Comparison of echocardiographic parameters in each group.
Values are expressed in comparison with the control group. Data are presented as mean ± SD. Differences between the two groups were analyzed by Student’s t-test. a P<0.05 versus control group; b P<0.05 versus non-AF group.
Abbreviations: LAD: Left Atrial Diameter; IVST: Inter Ventricular Septal Thickness; LVPWT: Left Ventricular Posterior Wall Thickness; LVEDD: Left Ventricular End-Diastolic Dimension; LVEF: Left Ventricular Ejection Fraction
We performed ELISA to examine the plasma levels of UA, XO, and Nox-4 among the groups. The ELISA results showed the blood concentrations of UA, XO, and Nox-4 in the AF group significantly increase compared with those in the non-AF and control group (P<0.001; Figures 1A, B, and C, respectively).
Inflammatory cytokines were increased in AF
ELISA results showed that the circulatory levels of inflammatory cytokines, such as Hs-CRP, IL-1β, IL-8, and TNF-α, significantly increased in the AF groups (Figure 2A-D) compared with the nonAF and control group, and the difference was statistically significant (P<0.001; Figures 2A, B, C, and D respectively).
Correlation between serum XO, Nox-4 and, inflammatory cytokines
Pearson correlation analysis showed that the serum levels of XO in AF group were positively correlated with the blood concentrations of Nox-4 and inflammatory cytokines (Hs-CRP, IL-1β, IL-8, and TNF-α) with statistical significance (P<0.01), and the respective correlation coefficients were r=0.99, 0.91, 0.96, 0.95, 0.97.
Taken together, our result suggests that blood levels of XO significantly elevated ROS accumulation and inflammatory cytokines, thereby inducing AF formation in patients with hypertension.
Discussion and Conclusion
Among the different types of cardiac arrhythmia, AF is of great concern, because it remarkably contributes to cardiac mortality and morbidity. Patients with AF are at high risk of stroke, heart failure, and dementia [16]. Atrial systolic function is lost when AF occurs, and the blood is easily congests in the atrium to form a thrombus. When the thrombus falls off, it follows the blood to all parts of the body, leading to cerebral or arterial embolism [17]. Aging, gender, rheumatic heart disease, hypertension, congestive heart failure, hyperthyroidism, chronic kidney disease, and diabetes mellitus have been recognized as AF risk factors.
HUA is associated with cardiovascular and renal diseases and an independent predictor of premature death [18]. The prevalence of comorbidities increases with gout duration, and gout is associated with all components of the metabolic syndrome [19]. Hypertension is the one of the most frequent comorbidities of gout, and the prevalence of hypertension is 69.1% and 30.3% in gouty and nongouty patients, respectively [20]. The prevalence of abdominal obesity is higher for gouty than non-gouty patients at 62.9% versus 35.3% [21]. Additionally, the risk of gout increases with obesity [22]. BMI and waist circumference are positively associated with uricaemia, and adiposity causes HUA [23]. A recent meta-analysis study showed that Chronic Kidney Disease (CKD) of stage 3 in gout was estimated at 24% [24]. Reduced kidney function reduces rate excretion in urine and increases gout risk. In a cohort study among CKD patients, gout prevalence increased from 16.0% to 35.6% for CKD patients along with declining estimated glomerular filtration rate [25]. Among patients with hypertension and AF, age, BMI, blood pressure (systolic and diastolic), hyperlipidemia, diabetes mellitus, creatinine, and eGFR were significantly altered compared with those in the non-AF and healthy control groups (Tables 1,2).
The presence of hyperuricemia increases AF risk, particularly in patients who have undergone cardiac surgery, such as a Coronary Artery Bypass Graft (CABG). Thus, SUA level can increase the sensitivity and specificity in AF prediction after CABG surgery [26]. Letsas et al. [27] found that SUA level is remarkably correlated with the new onset of AF in a concentration-dependent manner. A metaanalysis of six cross-sectional and three cohort studies confirmed the association of high SUA with AF [28]. Pinelli et al. [29] found that in patients with heart failure, serum UA levels are correlated with LVEF, which is independent from other clinical determinants, thereby indicating that the detection of progressive hyperuricemia in these patients may be an indicator of deteriorating cardiac function. Cicero et al. [30] reported that high SUA is a risk factor for cardiac dysfunction, which can be evaluated by LVEF and by plasma B-type Natriuretic Peptide (BNP). Owczarek et al. [31] demonstrated that SUA concentration variability is closely associated with BMI, eGFR values, serum NT-pro BNP levels, LVEF, and the use of thiazide diuretics. In the present study, LVEF values were remarkably reduced in hypertension and AF patients compared with non-AF and healthy controls (Table 3).
Although the pathophysiology underlying AF remains unclear, accumulated evidence suggests the involvement of inflammation and oxidative stress [32-33]. HUA is associated with the incidence of paroxysmal or persistent AF and the risk of AF in post-cardiovascular surgery patients [33]. To investigate the link between HUA and AF, we refer to HUA-induced atrial remodeling. Both ionic channel and structural remodeling caused by HUA might explain AF’s occurrence. Inhibition of XO and NADPH-oxidase or the use of antioxidants, along with the reduction in SUA levels prevent inflammation, might be useful. SUA is an independent predictor of CVD morbidity and mortality. The level of SUA is closely related to the severity of the disease and is an independent marker of adverse prognosis [34]. ROS plays an important role in AF and CHF occurrence and development. Oxidative stress during AF may play an important role in remodeling the atrial tissue [35]. Youn et al. [36] reported that the mechanisms of ROS production in human AF occurrence. They suggested that atrial oxidative stress during AF was caused by myocardial NADPH oxidase, nitric oxide synthase, and mitochondrial oxidases, which were associated with the electrical remodeling observed in AF. These findings suggest that oxidative stress may directly or indirectly regulate the metabolism of atrial muscle and functional properties of the atrium, thereby leading to atrial remodeling. Oxidative stress is closely associated with the pathogenesis of AF and hypertension results from overproduction of ROS. Long-term exposure to oxidative stress in hypertension induces chronic inflammation, vascular cell proliferation, and/or apoptosis in cardiovascular system, leading to formation and progression of disease states in this system. Indeed, markers for oxidative stress are over expressed in patients with hypertension and AF, suggesting that increased ROS may be primarily responsible for the development of AF. In the present study, serum levels of UA and XO in patients with hypertension and AF substantially elevated the expression levels of Nox-4 and accelerated AF progression (Figure 1).
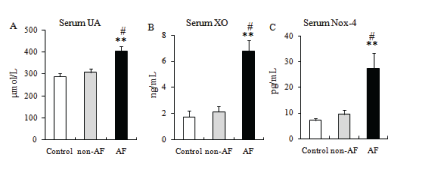
Figure 1: Uric acid, Xanthine Oxidase (XO), and NADPH oxidase-4 (Nox-4) expression was elevated in AF.
The blood levels of UA, XO, and Nox-4 were analyzed by ELISA. Values are expressed in comparison with the control group. Data are expressed as mean ± SD. The quantitative data were analyzed with one-way ANOVA, followed by Fisher’s PLSD test. The blood levels of UA (A), XO (B), and Nox4(C) among the groups. **P<0.001 versus control group, #P<0.01 versus non-AF group.
In the current study, direct measurement of ROS was not performed and this may consider a limitation associated with the study, which will need to be addressed in future work. Previously we reported that XO markedly induced the accumulations of ROS in adipose [9] and colon [14]. In the presents study we analyzed the expressions ROS marker (NADPH oxidase-4, Nox-4) using ELISA method. The results demonstrated that in AF patients high levels of XO significantly increased the circulatory levels of Nox-4. Therefore, we consume that XO significantly induced ROS production in AF patients.
The left appendage tissues obtained from AF patients showed more prominent presence of CD45-reactive cells, CD68-positive macrophages, and less CD3-positive T cells compared with tissues obtained from patients with a sinus rhythm [37]. In the atrium, inflammation induced its enlargement and the development of atrial fibrosis. Atrial fibrosis promoted AF by interrupting the continuity of fiber bundles, thereby causing atrial conduction disturbance [38]. Chao et al. [39] found that hs-CRP expression and insulin resistance were higher, and the LA diameter was large in patients with high SUA level. Increased inflammation and insulin resistance may be involved in the relationship between high SUA level and LA size. Mice treated with angiotensin-II showed an increase of neutrophil infiltration in their atrial tissue, which depends on CD11b and CD18 integrins, because angiotensin II stimulates the production of pro-inflammatory cytokines, such as IL-6, IL-8, and TNF-α, through the activation of angiotensin I receptors [40]. Although the relationship between UA and the RAAS system in cardiac atrial myocytes has never been tested, UA could stimulate the circulating and local RAAS system in the cardiovascular system. In accordance with these reports, we also demonstrated that the circulatory levels of inflammatory cytokines (Hs-CRP, IL-1β, IL-8, and TNF-α) were remarkably elevated in hypertension and AF patients, and SUA levels were closely associated with the levels of these cytokines (Figure 2).
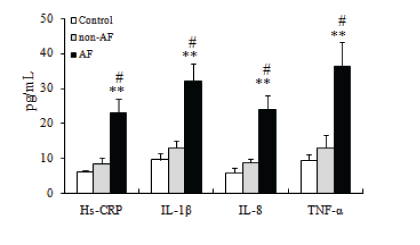
Figure 2: Inflammatory cytokines were increased in AF.
The plasma concentrations of inflammatory cytokines (Hs-CRP, IL-1β, IL-8, and TNF-α) were analyzed by ELISA. Values were expressed in comparison with the control group. Data were expressed as mean ± SD. The quantitative data were analyzed with one-way ANOVA followed by Fisher’s PLSD test. **P<0.001 versus control group, #P<0.01 versus non-AF group.
In conclusion, SUA is independently associated with the occurrence of AF. The accumulation of UA inside atrial cardiomyocytes might cause ionic and structural remodeling of the atria, which is referred to as atrial remodeling. In this study, the differences of LAD, IVST, LVPWT, and LVEF between the AF and non-AF group were significant (P<0.05) and may be related to the corresponding increase of UA and XO levels. The differences of age, BMI, and eGFR between the AF and non-AF group were statistically significant (P<0.05) and may be related to the increase of BMI and renal function. More importantly, XO activity in the AF group remarkably increased, thereby suggesting that SUA levels increased accordingly and further leading to the production of ROS (Nox-4), the activation of inflammatory cytokines (Hs-CRP, IL-1β, IL-8, and TNF-α), and the occurrence of AF. More studies are needed to obtain a more comprehensive understanding of AF pathophysiology and the measures to prevent it.
Acknowledgments
We thank Dr. Kyosuke Takeshita for carefully reading and applying changes to our manuscript.
Funding and Disclosure
The current study was supported by a grant from the Xinjiang Uygur Autonomous Region Natural Science Foundation Program (grant no. 2018D01C134). No duality of interest during the writing of this manuscript was declared by the authors.
Contribution Statement
MA, MY, and ZZZ contributed to the conceptualization and formulation of the experimental design and the interpretation of the experimental results. MA, AM, MY, WMZ, AA, YZ, TM, and XZ performed the experiments and analyzed the data.MA, MY, and WMZ wrote the manuscript. MA, MY, and ZZZ ensured the integrity of the entire work. This manuscript was revised by its authors. All authors decided to submit this version for publication.



