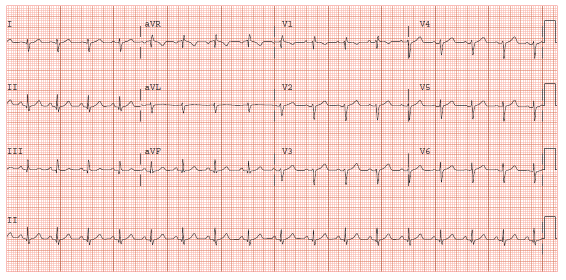
Figure 1: EKG demonstrating sinus tachycardia, right axis deviation, slow R wave progression in precordial leads, and relative low voltage in precordial leads


Gregory S Harris1 Irfan A Siddiqui2 Assad Movahed1* Ramesh Daggubati1
1Department of Cardiovascular Sciences, East Carolina Brody School of Medicine, Greenville, North Carolina, USA*Corresponding author: Assad Movahed, Department of Cardiovascular Sciences, East Carolina Brody School of Medicine, Greenville, North Carolina, USA, Tel: (252) 744-4561; E-mail: movaheda@ecu.edu
Though malignancy remains the most common cause of cardiac tamponade, large mediastinal T cell lymphoma mimicking as tamponade on transthoracic echocardiogram is rare. In this case report, we describe a 21-year-old otherwise healthy university student with a brief history of dyspnea and flu like constitutional symptoms. He was found to have left sided pleural effusion with pneumothorax on chest radiography with pulsus paradoxus on physical examination. An electrocardiogram also demonstrated low voltage. Chest tube placement and fluid analysis revealed exudate per Lyte’s criteria and chest computed tomography revealed a large anterior mediastinal mass with small to moderate pericardial effusion. Transthoracic echocardiogram revealed moderate pericardial effusion and tamponade physiology with right atrial and right ventricular collapse during both systole and diastole. Pericardiocentesis was deferred, as RV diastolic collapse was secondary to large anterior mediastinal mass as opposed to significant pericardial effusion. The patient was started on fractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone chemotherapy regimen as well as intrathecal methotrexate followed 14 days later by repeat transthoracic echocardiogram. This demonstrated resolution of his pericardial effusion, reduction in mass compression, and proper right ventricular filling.
Mediastinal T cell lymphoma; Cardiac tamponade; Pleural effusion; Mediastinal tumor; Cardiac compression
T cell acute lymphoblastic lymphomas (T-ALL) are aggressive neoplasms of T cell lineage that generally result in lymphadenopathy or bulky mediastinal masses which have been associated with superior vena cava syndrome, tracheal obstruction and/or pericardial effusions. These neoplasms account for approximately 2% of all non-Hodgkin’s lymphomas with a predominance of 2:1 males to females [1]. Nearly 10% of all cancer patients develop tamponade usually as a result of direct implantation of tumor cells. Here we describe a case of a 21 year old male who presented with a large mediastinal mass causing severe, direct compression of the right atrium and right ventricle who had pulsus paradoxus but did not have complete hemodynamic collapse. There have been no previous reports of large mediastinal mass secondary to T-ALL resulting clinical manifestations of cardiac tamponade.
A 21 year old male presented to ED with a 1-2 week history of flu like illness including non-productive cough, mild weight loss, and dyspnea during exercise. He denied hemoptysis, night sweats, fever, chills, nausea, vomiting or diarrhea. He was found to have a small left sided pneumothorax and moderately large left pleural effusion. An EKG revealed low voltage and sinus tachycardia (Figure1). A 20 French chest tube was placed and 1 liter of serous fluid was drained after which a repeat chest X-ray demonstrated reduction in the pleural effusion and suspected moderate left pneumothorax (Figure 2A). CT scan of the chest was performed demonstrating a large anterior mediastinal mass measuring approximately 8.6x 14.6 cm in the greatest transverse diameter extending from the thoracic inlet to the diaphragm, enlarged left supraclavicular and axillary lymph nodes, as well as a moderate pericardial effusion (Figure2B).

Figure 1: EKG demonstrating sinus tachycardia, right axis deviation, slow R wave progression in precordial leads, and relative low voltage in precordial leads
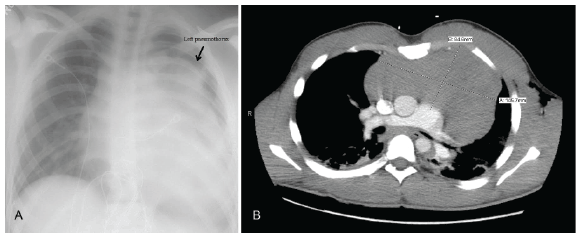
Figure 2: A. Chest x-ray demonstrating left pleural effusion and
moderate left pneumothorax
B. Computed tomography of chest demonstrating large anterior
mediastinal mass
Physical exam revealed BP 127/86, HR 87 beats per minute, Temp 36.8°C, Respirations 18, SpO2 92-94% on room air. He was in no acute distress and was able to converse with us during our exam without difficulty or broken sentences. Heart sounds were distant, but there was no rub, murmur, or gallop. Pulsus paradoxus was present with a 14 mmHg decrease in his blood pressure with inspiration without Kussmaul’s sign [2].
Ultrasound guided biopsy of the anterior mediastinal mass as well as bone marrow biopsy was performed that confirmed T-lymphoblastic leukemia/ lymphoma. The histologic sections of bone marrow demonstrated a markedly hypercellular marrow with an estimated cellularity of 100%. There was a diffuse infiltrate of blasts (80%) with intermediate to small size with round to irregular nuclei. The sample from the mediastinal mass was CD1a, CD4, CD7, CD8, and CD10 positive. This type of lymphoma was amenable to chemotherapy and reduction of tumor size.
Echocardiogram prior to starting chemotherapy with fractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone (HyperCVAD) demonstrated a normal left ventricle with EF of 60-65% without wall motion abnormalities. The left atrium (LA) was normal in size; however, the right atrium (RA) was noted to be small in size and compressed by the tumor. The right ventricle (RV) and RV outflow tract was severely compressed by the mediastinal mass resulting in trace to mild tricuspid regurgitation (Figures 3). The inferior vena cava (IVC) was dilated with abnormal collapsibility suggesting increased right atrial pressures. A 1-2 cm pericardial effusion was noted with echocardiographic evidence of cardiac tamponade [3]. The heart was noted to be freely swinging in the pericardial fluid with 25-30% respiratory variation in the mitral valve E wave velocity.
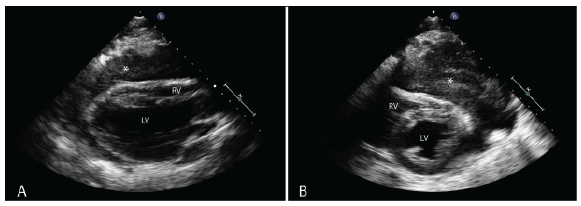
Figure 3: A. Echocardiogram demonstrating parasternal long axis view with compression of the right ventricular outflow tract. Star (* ) indicates tumor B. Short axis view demonstrating compression of the right ventricle by tumor. Star (* ) indicates tumor
The patient was started on hyper-CVAD 1A as well as intrathecal methotrexate. Two weeks after the initiation of treatment, repeat CXR and ECHO were performed to evaluate the effects of treatment on cardiac performance. The studies demonstrated no compression of the RV and normal left ventricular (LV) function. Chest X-ray demonstrated large reduction in the size of the mediastinal tumor.
Here we present a case right ventricular compression as a result of extrinsic tumor burden and moderate pericardial effusion. The patient was relatively stable hemodynamically (ie. Normal blood pressure) despite having pulsus paradoxus on presentation. Hemodynamic compromise in cardiac tamponade is greatly influenced by the rate at which fluid accumulation occurs. Reddy et al characterized the changes in hemodynamics as a result of cardiac tamponade into three phases [4].In the first phase there is a gradual increase in pericardial fluid accumulation that results in increased intrapericardial pressure; RV and LV filling pressures are increased with RV filling pressure being equal to intrapericardial pressure. RA and intrapericardial pressures continue to increase during phase two until they equal LA pressure. During phase two, pulsus paradoxus becomes apparent with decreased arterial pressures during inspiration reaching 10 mmHg. In phase 3, the pressure in the RA, LA and pericardium equalizes resulting in even greater decreases in cardiac output. It is also during this phase total circulatory collapse occurs culminating in cardiogenic shock [4].
In the case of our patient, he presented with normal blood pressure, distant heart sounds, jugular venous distension (JVD), and pulsus paradoxus thus meeting two out of three of Beck’s Triad [5]. Regardless, clinically he did not appear to be in acute danger. Echocardiogram was performed for baseline measures before starting chemotherapy and it was at that point right atrial and right ventricular compression, moderate pericardial effusion, “swinging heart” and mitral inflow velocity variation was made apparent [6,7]. Though these echo findings would suggest cardiac tamponade, much of the compression of the RA and RV was realized to be secondary to his mediastinal tumor directly impeding RV expansion, and thus, he would be classified in phase 2 as described above. Patients in this category are of great concern as they can quickly degenerate to phase 3 resulting in collapse. The fact that he did not progress to phase 3 is likely a result of his history of physical training and young age.
T-cell acute lymphoblastic lymphomas, as demonstrated in our patient, have a higher rate of mediastinal tumors than those of B cell origin and are likely to present at Stage IV (80%) and with B symptoms (50%). Bone marrow is usually spared in these patients at initial presentation, however approximately 60% will have infiltration followed by a leukemic phase. In these patient’s lumbar puncture should be performed to rule out CNS involvement. Patients are more likely to be male with a 2:1 predominance and more likely to be in their teens to twenties. Though this neoplasm is highly aggressive it has a good prognosis with current therapy. Seventy-three to ninety percent of children and 62-66% of adults have a disease free survival [1].
Though our patient did have moderate pericardial effusion, his tumor burden was the primary reason for his RA and RV compression and phases 2 tamponade. Fortunately for this patient, his pericardial fluid accumulation likely occurred over a long period of time allowing his pericardial tissue to expand and compensate for the elevated pressures. His tumor was successfully reduced with treatment and follow-up ECHO demonstrated full expansion of his RA and RV.
None of the authors have any disclosures.
Download Provisional PDF Here
Article Type: Case Report
Citation: Harris GS, Siddiqui IA, Movahed A, Daggubati R (2016) “Tumor and Tamponade” Large Mediastinal Lymphoma Masquerading as Cardiac Tamponade on Transthoracic Echocardiogram. J Clin Case Stu 1(3): doi http://dx.doi.org/10.16966/2471-4925.121
Copyright: © 2016 Harris GS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access