Article Information
Article Type: Case Report
Citation: Hyunsu Shin, Young Ri Kim, Jie Ae Kim, In Sun Chung (2017) Profound Bradycardia and Hypotension after Sugammadex Administration. J Clin Anesth Manag 2(1): doi http://dx.doi.org/10.16966/2470-9956.126
Copyright: © 2017 Hyunsu Shin et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
Received date: 31 Jul 2017
Accepted date: 16 Aug 2017
Published date: 21 Aug 2017
Abstract
We report a case of a 46-year-old female patient who underwent subtotal gastrectomy and developed profound bradycardia and hypotension after administration of sugammadex. She had no previous medical history and preoperative tests were normal, except for multiple atrial premature beats seen on an electrocardiography. Three minutes after sugammadex injection, she developed severe bradycardia and hypotension, which improved after administration of atropine. No rash or urticaria was observed, and airway pressure was not increased. In addition, because neuromuscular blockade was not reversed after sugammadex administration, pyridostigmine was administered to reverse it. After her muscular strength recovered completely, extubation was performed, and she was transferred to the postoperative care unit. In conclusion, the risk of severe adverse effects due to sugammadex should be considered, and close monitoring is recommended after sugammadex administration.
Keywords
Sugammadex; Bradycardia; Hypotension; Neuromuscular blockade
Introduction
Sugammadex was approved in the European Union in 2008, and received the US Food and Drug Administration approval in 2015 [1]. It is now widely used for reversal of the effects of the aminosteroidal neuromuscular blocking agents such as rocuronium and vecuronium [2]. Sugammadex encapsulates rocuronium molecules, preventing them from binding to nicotinic acetylcholine receptors at the neuromuscular junction [2]. It is thought to be relatively safe [3,4], but recently several cases of adverse effects including hypotension, arrhythmia, and anaphylactic reactions were reported [5-8]. Here, we describe a case of a patient who developed severe bradycardia and hypotension after administration of sugammadex.
Case report
A 46-year-old female patient (weight 64.9 kg, height 165.2 cm) was scheduled to undergo subtotal gastrectomy for gastric cancer. She had no previous disease history and no history of drug use. Preoperative laboratory tests and chest x-ray revealed no abnormality, and electrocardiography (ECG) showed sinus rhythm with multiple atrial premature beats. Because she was not an elderly patient, had no previous history of cardiovascular disease, and her echocardiogram was normal, we decided to proceed with the operation without receiving further examination by cardiologist. The patient was taken to the operating room, and monitoring of non-invasive blood pressure, ECG, heart rate, and peripheral oxygen saturation (SpO2 ) was started. Anesthesia was induced with 300 mg thiopental sodium and 50 mg rocuronium. After endotracheal intubation, anesthesia was maintained with sevoflurane (end-tidal sevoflurane 1.7~2.1 vol%) in a 50% air/O2 mixture at a flow rate of 2 L/min. The body temperature of the patient was continuously measured by an esophageal temperature probe (Esophageal stethoscope with temperature sensor, Sewoon Medical Co., Ltd., Cheonan-si, Chungcheongnam-do, Korea), and the initial body temperature was 35.7℃. During the surgery, the patient’s hemodynamic status was stable except for several atrial premature beats on ECG. Because the surgeon requested deep neuromuscular blockade to facilitate surgery, rocuronium was administered continuously at the rate of 8 µg/kg/min and was stopped 40 min before the end of surgery. The total dose of administered rocuronium including the induction dose was 97 mg, during general anesthesia. Thirty minutes before the end of the surgery, 0.6 mg hydromorphone was administered for postoperative pain control. The total duration of the surgery was 130 min, and after its completion, 200 mg sugammadex was administered to reverse residual neuromuscular blockade (at 155 min after induction of anesthesia). Three minutes after the administration, the heart rate decreased from 55 beats/ min to 23 beats/min (Figure 1). At that time, other vital signs were as follows: blood pressure, 58/45 mmHg; SpO2, 100%; peak airway pressure, 17 cm H2O; end-tidal CO2, 42 mm Hg; and body temperature 35.3℃. No rash or urticaria was observed. A dose of 0.5 mg atropine was quickly administered intravenously, and 1 min later, the heart rate increased to 89 beats/min and blood pressure was 178/115 mm Hg. The neuromuscular block level of the adductor pollicis muscle was checked with a peripheral nerve stimulator (EZstimII, Life-Tech, Inc., Stafford, Texas, USA), and the train-of-four (TOF) count was nearly 0/4 and the post-tetanic count was 4. Ventilation was assisted with intermittent manual bagging, and after 5 min, when the TOF count reached 2/4, 15 mg pyridostigmine and 0.4 mg glycopyrrolate were administered to reverse residual neuromuscular blockade. Ten minutes after administration of pyridostigmine, the TOF count showed 4/4 and self-respiration recovered, but the patient still could not open her eyes or obey verbal commands. Twenty minutes later, she could follow verbal commands and her muscular strength recovered completely, extubation was therefore performed. The vital signs of the patient were stable for 1 hour in the postoperative care unit, and she was discharged to the general ward without any complications.
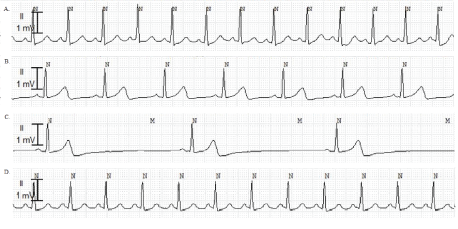
Figure 1: ECG changes during anesthesia.
A. Before anesthesia induction, ECG showed normal sinus rhythm (HR 93 beats/min, QTc 441 ms).
B. After surgery, ECG showed slight sinus bradycardia (HR 55 beats/min, QTc 491 ms).
C. Three minutes after 200 mg sugammadex administration, ECG showed severe bradycardia (HR 23 beats/min, QTc 490 ms).
D. One minute after 0.5 mg atropine injection, HR increased to 89 beats/min (QTc 490 ms).
Note: ECG-Electrocardiography; HR-Heart Rate; QTc-Corrected QT
Discussion and Conclusion
Cholinesterase inhibitors have been used to antagonize the effects of non-depolarizing neuromuscular blocking agents (NMBA) [9]. However, some adverse effects of cholinesterase inhibitors have been reported such as bradycardia, hypotension, bronchoconstriction, hypersalivation and recurarization [2,10]. Sugammadex is a selective NMBA-binding agent and has been used to antagonize the block caused by non-depolarizing aminosteroidal NMBA, primarily rocuronium [2,3]. Sugammadex is generally well tolerated, and common adverse effects are insufficient depth of anesthesia, nausea, vomiting, hypotension, and pain [3,4,11]. Recently, hypersensitivity reactions including rash, tachycardia, and even anaphylactic shock have been reported [6,8]. Godai et al. [6] reported 3 cases of hypersensitivity reactions after sugammadex injection such as facial or generalized erythema, hypotension, increased peak airway pressure, or wheezing, and the symptoms improved after administration of an antihistamine or methylprednisolone. Yoo et al. [8] also reported a case of a patient who developed edema, dyspnea, and shock 2 min after sugammadex injection, and the symptoms disappeared after administration of epinephrine. However, in our case, there was no other symptom indicative of hypersensitivity reaction or anaphylaxis such as a rash, urticaria, or increased airway pressure, and only severe bradycardia and hypotension were observed. Therefore, we concluded that our case was not one of hypersensitivity or an anaphylactic reaction.
There have been a few reports of ECG abnormalities such as QT prolongation or heart block induced by sugammadex [3,7,11]. According to the phase 1 study of sugammadex, 8 cases of corrected QT (QTc) prolongation were observed in six volunteers, but no abnormal heart rate was observed [3]. Sparr et al. [11] reported that patients who received sugammadex showed mild QTc prolongation compared to that in those who received placebo, but the changes were not significant. Osaka et al. [7] reported a case of second-degree atrioventricular block after sugammadex administration; however, in their case, there were no changes of heart rate and normal sinus rhythm was restored within a few minutes without treatment. However, in our case, the patient showed only severe sinus bradycardia and no other arrhythmia or QTc prolongation.
There have been a few cases of bradycardia after administration of sugammadex [3-5,12]. The incidence of bradycardia was approximately 1.9 % after sugammadex injection [4], and treatment with an anticholinergic agent was recommended in cases of clinically significant bradycardia [3,4]. Groudine et al. [12] reported mild bradycardia 2 min after administration of 8 mg/kg sugammadex. In their report, the heart rate of the patient decreased from 62 beats/min to 42 beats/min, and increased to 65 beats/min after glycopyrrolate injection. Bilgi et al. [5] reported a case of persistent bradycardia associated with sugammadex. In their case, after 200 mg sugammadex administration, the patient’s heart rate decreased to 35 beats/min, and the bradycardia recovered after repeated injections of atropine (total dose 2 mg). Similarly, in our case, the bradycardia recovered after atropine administration.
In addition, in our case, neuromuscular blockade was not reversed by sugammadex, so pyridostigmine was also administered to restore muscle relaxation. Although we did not check the level of neuromuscular block before sugammadex administration, 200 mg sugammadex (resulting in 3.08 mg/kg in the patient) was the dosage that could be expected to moderately reverse neuromuscular blockade [3,4]. However, in our case, the TOF count was still nearly 0/4 after administration of 200 mg sugammadex. Several hypothetical explanations of this observation are possible. First, it is possible that 200 mg sugammadex was not sufficient to reverse the total dose of rocuronium. We administered 50 mg rocuronium (resulting in 0.77 mg/kg in the patient) for induction of anesthesia (155 min before sugammadex injection) and we infused an additional 47 mg of rocuronium continuously during the operation for up to 40 min before the end of the operation. According to previous studies, the mean time to recovery of the TOF ratio to 0.9 was approximately 52 min after 0.6 mg/kg rocuronium administration [11], and approximately 122 min after 1.2 mg/kg rocuronium administration [9]. Therefore, in our case, rocuronium administered for induction of anesthesia seemed to cause negligible neuromuscular blockade at the time of sugammadex administration. In addition, because 200 mg sugammadex can encapsulate 55 mg of rocuronium [13], it is considered sufficient to reverse neuromuscular blockade caused by 47 mg rocuronium which was additionally administered during the surgery, making this explanation less likely. However, it is still our limitation that we did not monitor the level of neuromuscular block during the operation and we did not check the exact level of neuromuscular block before sugammadex administration. Second, it is possible that the administered sugammadex did not bind to the rocuronium molecules but bound to other molecules. For example, dexamethasone structurally resembles the aminosteroid neuromuscularblocking agent [14], and some antibiotics may have the potential for a displacement interaction with sugammadex [15]. However, such drugs were not administered in our case, so this hypothesis is also considered to be less likely. Third, the reversal from neuromuscular blockade might be prolonged by hypothermia. According to the previous studies, the duration of action and recovery of rocuronium were prolonged by moderate hypothermia [16,17]. However, Lee et al. [18] reported that sugammadex could completely reverse rocuronium-induced neuromuscular block in patients with mild hypothermia. In their study, the mean recovery time after sugammadex administration in deep neuromuscular blockade caused by rocuronium was 171.1 sec (range 61-305 sec) in patients with mild hypothermia (esophageal temperature 34.5-35℃) which was prolonged by only 46 sec compared with normal thermal condition [18]. In our case, the patient’s body temperature was maintained above 35℃ during the operation and there was a time interval of about 4 min from sugammadex administration to the neuromuscular block level check, but it is thought that the effect of hypothermia cannot be completely excluded.
In summary, we report a case of severe bradycardia and hypotension in a patient who received 200mg sugammadex after general anesthesia for reversal of neuromuscular blockade, which persisted despite the sugammadex administration. Although the exact mechanism is unknown, severe bradycardia may occur within a few minutes after sugammadex administration, and can be treated with anticholinergic agents such as atropine. Therefore, close monitoring including ECG and neuromuscular block level is recommended to the patient who received sugammadex injection.


