Article Information
Article Type: Case Report
Citation: Merced DP, Ellis T, Applefield D, Krishnan S (2017) Management of Mitral Clip Detachment in an Elective Mitral Valve Clipping. J Clin Anesth Manag 2(1): doi http://dx.doi.org/10.16966/2470-9956.123
Copyright: © 2017 Merced DP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
Received date: 21 Oct 2016
Accepted date: 10 Feb 2017
Published date: 14 Feb 2017
Abstract
According to the American College of Cardiology, more than 42,000 patients undergo mitral valve surgery in the United States per year. The treatment of mitral valve regurgitation can be extremely complex and has been delegated almost exclusively to cardiothoracic surgeons. In recent years, the creation of a commercial available percutaneous mitral clipping device called the MitraClipTM (Abbott Global Healthcare), that anchors one edge of the mitral valve leaflet to its corresponding opposite leaflet, has gained favor among cardiothoracic surgeons and cardiologists for patients who would be at high risk for morbidity and mortality with open cardiac surgery. In the recent EVEREST II 5-year follow up study, Feldman et al. [1] reported that both percutaneous mitral clipping and surgical intervention have similar long term results in MR reduction. We report a case of a deployed mitral clip detaching from the posterior leaflet, while remaining attached to a flail segment of the anterior leaflet. Another percutaneous mitral clip was placed alongside the failed mitral clip, resulting in a clinically significant reduction in MR.
Case Report
A 75-year-old male was admitted to the hospital for elective percutaneous mitral valve clipping. He reported a past medical history of coronary artery disease, status post myocardial infarction and coronary artery bypass grafting 10 years prior, hypertension, benign prostatic hypertrophy, obesity, diet-controlled diabetes mellitus, and severe MR. The patient complained of dyspnea with minimal exertion, orthopnea, and New York Heart Association (NYHA) class III symptoms (Table 1). His pre-operative transesophageal echocardiogram (TEE) demonstrated a left ventricular (LV) ejection fraction of 40%, a dilated left atrium (LA), and severe MR due to a flail A2 segment on the anterior leaflet (Figures 1-3). The mitral regurgitant fraction was estimated to be 53%, and the effective regurgitant orifice area was measured to be 0.46 cm2 , consistent with the diagnosis of severe MR. The patient was taken to the operating room (OR) and prepared for surgery. A pre-induction 20-gauge arterial line was placed in the right radial artery, and the patient was induced with anesthetic agents and placed under general endotracheal anesthesia. The patient remained hemodynamically stable throughout the induction, and a rightsided 9.0 french Swan-Ganz Introducer was placed into the right internal jugular vein under sterile conditions without complications. Stomach contents were suctioned, and a TEE probe was placed into the patient’s esophagus without difficulty. The right femoral vein was cannulated by the cardiology team, and a trans-septal puncture kit was used to facilitate introduction of the mitral clip sheath into the left atrium. The mitral clip deployment sheath was placed into the right femoral vein, advanced into the right atrium, and finally across the interatrial septum into the LA. The mitral clip deployment device was then placed through the sheath and into the LA (Figure 4). Using TEE and radiographic guidance, the mitral clip was positioned between the anterior and posterior leaflets at the site of the flail segment. Multiple attempts were required in order to successfully grasp both leaflets simultaneously, after which the clip was deployed and tightened. Insertion of both mitral leaflets into the mitral clip was confirmed with TEE prior to deployment. Evaluation of positioning of the mitral clip showed that although there was a small reduction in MR, the mitral clip had not succeeded in reducing the MR to the degree anticipated. After a thorough review of the status of the valve on TEE, as well as discussion between the cardiology team, the cardiac surgeon, and the cardiac anesthesiologist, the decision was made to attempt placement of a second clip. A second mitral clip was subsequently prepared by the OR team for deployment. During the preparation of the second mitral clip by the OR team, the first mitral clip was noted to be dislodged from the posterior leaflet of the mitral valve on TEE. This initial clip was still attached to the flail segment of the anterior leaflet of the mitral valve, but the degree of MR had regressed to pre-mitral clip conditions (Figure 5). The patient remained hemodynamically stable after the detachment of the initial clip from the posterior mitral leaflet. The second clip was positioned laterally to the first clip between the A2 and P2 segments of the mitral valve (Figure 6). After the simultaneous grasp of both leaflets with the mitral clip and a significant reduction in MR were noted on TEE, the second clip was deployed successfully without incident. A comprehensive TEE was performed by the cardiac anesthesiologist after the deployment of the second mitral clip to evaluate the patient’s cardiac status. The MR was noted to be reduced from severe to moderate, and the patient had a small atrial septal defect from the trans-septal puncture (Figures 7-9). All other TEE findings, including LV function, remained unchanged from prior to the procedure.
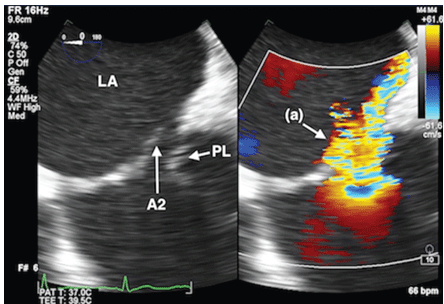
Figure 1: Mid-esophageal 4 chamber view (zoomed) showing the flail A2 segment and severe, posteriorly-directed MR jet (a). LA = left atrium; PL = posterior leaflet of mitral valve; A2 = A2 segment of anterior leaflet of mitral valve; MR = severe mitral regurgitation.
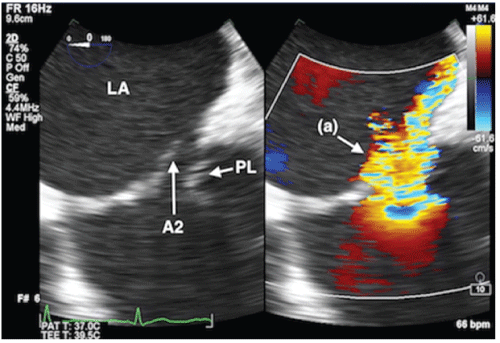
Figure 2: Mid-esophageal commissural view showing severe MR (a) between A2 and P2 segments. P1 = P1 segment of posterior leaflet of mitral valve; P3 = P3 segment of posterior leaflet of mitral valve; A2/P2 = A2/P2 segments of mitral valve.
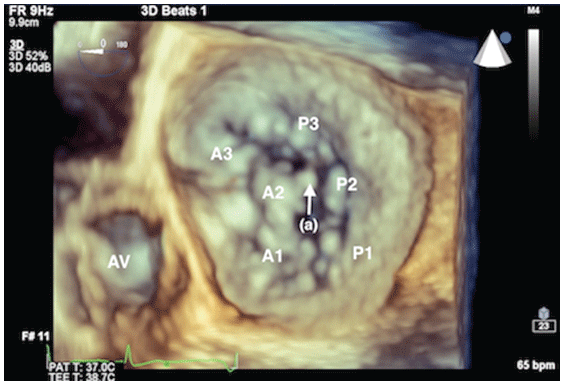
Figure 3: En face 3-dimensional view of mitral valve apparatus and its corresponding segments, with flail segment at A2 shown (a). AV = aortic valve.
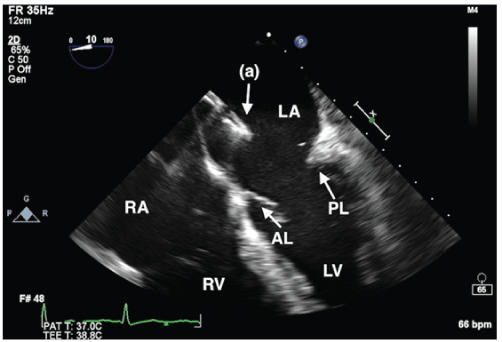
Figure 4: Mid-esophageal 4-chamber view showing the advancement of the mitral clip deployment device (a) towards the mitral valve through a transseptal approach. The closed mitral clip can be seen at the tip of the device. RA = right atrium; RV = right ventricle; LA = left atrium; LV = left ventricle; AL = anterior leaflet of mitral valve; PL = posterior leaflet of mitral valve.
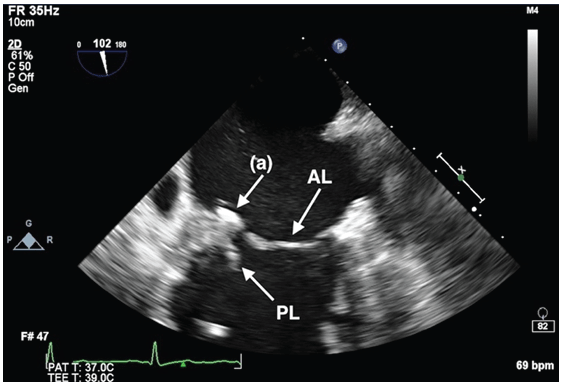
Figure 5: Mid-esophageal 2-chamber view, showing the first mitral clip after deployment (a). It has detached from the posterior leaflet and is seen attached to the flail A2 segment, floating above the level of the mitral annulus. AL = anterior leaflet; PL = posterior leaflet.
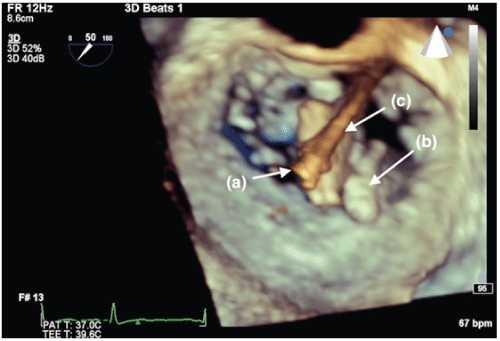
Figure 6: En face 3-dimensional view of the new, second clip (a) being advanced into position lateral to the first, detached clip (b), which can also be seen. The second clip is still attached to the deployment device (c) in this image.
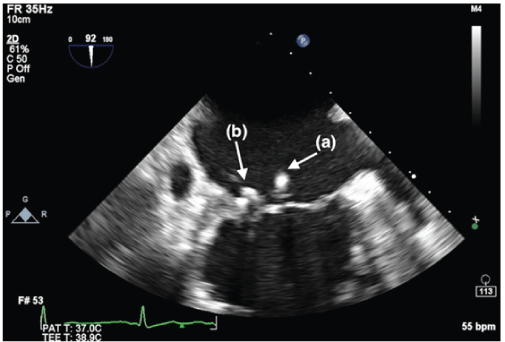
Figure 7: Mid-esophageal 2-chamber image, showing the second clip in place (a). The first clip (b) can be seen detached from the posterior leaflet.
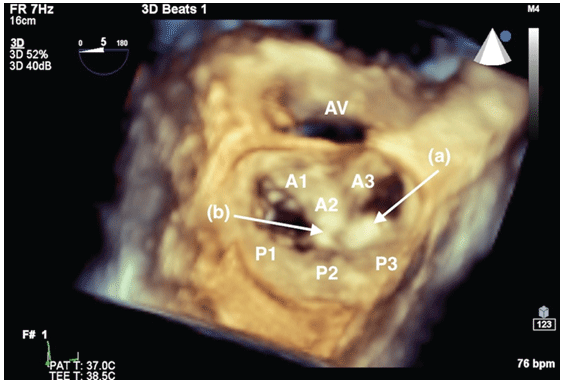
Figure 8: En face 3-dimensional image of first (a) and second (b) clips in place. The first clip can be seen detached from the posterior leaflet.
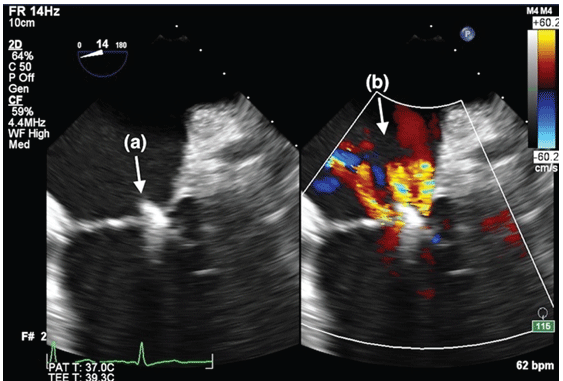
Figure 9: Mid-esophageal 4-chamber view (zoomed), showing the second mitral clip (a) after deployment. There is a significant reduction of the MR (b).
NYHA
Class |
Symptoms |
| I |
No limitation of physical activity. Ordinary physical activity does not cause undue fatigue, palpitation, dyspnea. |
| II |
Slight limitation of physical activity. Comfortable at rest.
Ordinary physical activity results in fatigue, palpitation, dyspnea. |
| III |
Marked limitation of physical activity. Comfortable at rest. Less than ordinary activity causes fatigue, palpitation, or dyspnea. |
|
IV |
Unable to carry on any physical activity without discomfort. Symptoms of heart failure at rest. If any physical activity is undertaken, discomfort increases. |
Table 1: NYHA Functional Classification. It places patients into 1 of 4
categories based on their degree of functional limitation during physical activity.
Outcome and Follow-Up
The patient remained hemodynamically stable and was subsequently transported with full monitoring, while intubated, to the cardiovascular intensive care unit (CVICU). The patient was extubated 3 hours after arriving in the CVICU. He related that his dyspnea had improved and reported no ill-effects from the procedure. He remained stable and was discharged home on post-operative day 3.
Discussion
The average life expectancy of the general population continues to increase, and thus the prevalence of MR continues to rise [2]. Surgical intervention has consistently been the mainstay of MR management, as even asymptomatic MR often progresses to LV failure. For many years, surgical intervention for MR meant mitral valve replacement. However, in the mid-1990s, Enriquez-Sarano et al. [3] conducted a study comparing mitral valve repair to valve replacement. The study demonstrated that mitral valve repair was superior to mitral valve replacement due to its significant improvement in postoperative outcomes. In the years to follow, cardiac surgeons began focusing on mitral valve repair versus replacement. With increasing emphasis on minimally invasive surgical techniques, surgeons began developing minimally invasive mitral valve repair techniques, ranging from a right lateral mini-thoracotomy approach to a robotic approach with the da Vinci Robotic System (Intuitive Surgical, Sunnyvale, CA, USA). Minimally invasive mitral valve surgery has been heavily studied to determine outcomes in comparison to open surgery. A study by Ramlawi et al. [4] evaluated minimally invasive mitral valve surgery techniques, and they found similar rates of renal failure, stroke, and survival outcomes with either minimally invasive or conventional mitral valve surgery; however, they also reported a decreased rate of red blood cell transfusions, frequency of post-operative atrial fibrillation, and time to recovery.
A strong interest in developing safer minimally invasive techniques has resulted in the evolution of percutaneous approaches to mitral valve repair. These percutaneous techniques have gained popularity in recent years. After completing the initial EVEREST trial in 2009 evaluating a percutaneous mitral clip device, Feldman et al. [5] released the results of their investigation. They concluded that ‘percutaneous repair with the mitral clip system can be accomplished with low rates of morbidity and mortality and with acute MR reduction to <2+ in the majority of patients, and with sustained freedom from death, surgery, or recurrent MR in substantial proportion.Glower et al. [6] reinforced those findings in the EVEREST II trial, concluding that the mitral clip procedure had a similar repair rate to surgery during the first year. The EVEREST II trial included only patients who were acceptable surgical candidates with preserved LV function. Investigators then attempted to study the 1-year results of patients deemed to be at high surgical risk. They classified high surgical risk as having an operative mortality rate of ≥ 12%, based on STS risk calculations or surgeon-estimated surgical risk. Of the 78 patients they included in their study, 75% of the patients with mitral clip implantation showed a reduction of MR from severe to moderate. 70 of the 78 patients selected were NYHA class III/IV pre-intervention; after the mitral clip intervention, 74% of the surviving patients were graded NYHA class I/II. Additionally, 75.4% of the high-risk mitral clip patients were alive at 1 year compared to 55.3% of patients in a retrospective comparator group managed medically [7]. This data demonstrated that the mitral clip placement improved both MR and clinical symptoms in the majority of high risk patients. Feldman et al. [1] went on to evaluate 5-year outcomes of mitral clip repair versus conventional mitral valve surgery. They concluded that the mitral clip device showed improved safety when compared with mitral valve surgery, but with more need for surgical repair of MR during the first year after surgery. Between years 1-5, there were comparable rates of surgery for mitral valve dysfunction with either mitral clip placement or surgical intervention. These EVEREST trials helped to illustrate the efficacy and safety of mitral clip placement, and solidified its role as an alternative to conventional surgical approaches in high-risk surgical patients. Although mitral clipping has been shown in studies to have superior safety when compared to surgery, it is not without complications. Eggebrecht et al. [8] evaluated complications in 828 patients during and after mitral clipping, with major complications in occurring in 12.8% of those patients. These complications included bleeding (7.4%), in-hospital death (2.2%), pericardial tamponade (1.9%), partial clip detachment (1.9%), and stroke (0.9%). However, Magruder et al concluded in a more recent review of the mitral clip device, that clip placement has been shown to be a safe alternative to surgery in high-risk surgical patients, and can dramatically improve the symptoms and degree of MR in patients [9]. In conclusion, this case study intends to expose clinicians to an innovative and effective procedure for improvement of MR. The first clip detaching from the posterior leaflet was an interesting finding that was readily seen on TEE. Successful placement of a second clip placed laterally to the first clip proved an efficacious method of salvage of the percutaneous procedure in a high-risk patient resulting in significant improvement of the MR on TEE.
Download Provisional pdf here










