Abstract
IRIS is a paradoxical reaction occurring in HIV-infected patient after initiation of highly active antiretroviral therapy (HAART). Incidence is
estimated 12 to 19% of child starting HAART. IRIS with tuberculosis in non-HIV patients has not been reported in the literature. We report the case
of an HIV negative 8 years-old girl who did develop IRIS and dilated cardiomyopathy during her treatment for severe disseminated TB (lungs,
mediastinal lymph nodes, spleen, liver and kidneys). An appropriate antibiotic treatment was begun against the multiple sensitive mycobacterium
tuberculosis strain retrieved: RHZE for 2 months followed by RH 10 months. The child appeared stable the 2 first weeks on treatment but not
apyretic. She remained in the hospital for 6 months and experienced daily recurrent fever 39-40°C with rigors, diffuse bone pains, elevated
transaminases and deterioration of chest infiltrates. Sequential abdominal CT and Gallium scans demonstrated a progressive involvement
of liver, spleen, kidneys, medullar bones and abdomen lymph nodes for 2 weeks to 5 months. Then she gradually improved. Her complete
immunological work-up at that point was considered normal.
We concluded from an extensive review of the literature that this case highlights a paradoxical immune mediated reaction to Mycobacterium
tuberculosis during an appropriate anti-TB treatment. IRIS results in clinical deterioration but is not a failure to treatment. Early recognition of IRIS
could prevent unnecessary changes in anti-TB regimen.
Keywords
Immune Reconstitution Syndrome (IRIS); Tuberculosis (TB); HIV; Highly Active Antiretroviral Therapy (HAART)
Introduction
Immune reconstitution inflammatory syndrome (IRIS) is a paradoxical
reaction initially described in HIV-infected patient after the initiation of an
antiretroviral therapy (ART). IRIS is mostly seen in HIV-infected patients
[1]. The incidence is estimated between 12 to 19% in children starting ART
[2-4]. TB associated IRIS is characterized by a clinical worsening after the
start of ART in patients receiving Tb treatment. It can also be diagnosed
as a new presentation of TB that is «unmasked» in the weeks following the
initiation of ART and the exclusion of other causes that could explain a
deterioration (such as antimicrobial drug resistance, drug hypersensitivity
reaction, or another opportunistic infection) [5]. IRIS in association with
tuberculosis in non-HIV patients has not been reported in the literature.
We reported the case of IRIS in child-disseminated tuberculosis.
Case Report
An 8-old-year girl was referred to our hospital for fever and abnormal
CXR, no medical investigation upon her arrival in Canada 3 years ago,
unknown immunization status and no pets. Medical history noticed right
thigh pain, weight loss over 2 months with sleepiness, fatigue and lack
of energy. She coughed for 3 weeks presenting dyspnea on exertion. She
suffered neither from any sputum, hemoptysis, and thoracic pain nor
wheezed any gastrointestinal symptoms. When arrived, the temperature
was 39.1 degree centigrade, the weight below 3rd percentile and the height
on the 10th percentile. Normal blood pressure and SpO2
98% on air room.
On examination, she was cachectic, pale conjunctivae, no clubbing and
peripheral œdema. The respiratory system exam found normal thorax,
tachypnea (respiratory rate = 28/min), no in drawing, no grunting
or stridor diffusing crackles lung bases (right > left). Cardio-vascular
system objectived tachycardia (heart rate=172/min) and well perfused.
Her abdomen was soft without any hepato-splenomegaly. She hadn’t
any lymphadenopathy and neurological signs. The hemogram showed
hypochromic microcytic anemia (Hb= 59 g/L), leukocyte count of 5430/
μl with polymorphonuclear predominance and platelet count normal.
Serum electrolytes and creatinine were normal. C-reactive protein
(CRP) was 60 mg/l; Liver transaminases were mildely elevated (ALT:
42 and AST: 78 IU/L, respectively) ; normal capillary gas. Mantoux test
was non-reactive. The chest radiograph showed small foci of alveolar
consolidation distributed in lungs predominant in middle lobe, no pleural
effusion or cavitation (Figure 1), right femur and pelvic x ray normal. The
chest computed tomographic (CT) (Figure 2) showed diffuse bilateral
nodular lesions in the lung parenchyma, predominant in middle lobe
associated with multiple mediastinum lymphadenomegaly, right lower
lobe atelectasis, slight right and left pleural effusion. Abdomen and pelvis
CT scan findings were heterogeneous spleen with multiple hypodensities,
homogenous liver and pancreas, Homogenous kidneys, Ascites in mild
to moderate. She still complained both persistent right thigh pain and
diffuse bone one. So Technitium and Gallium bone scan (Figure 3)
performed showed mild diffuse enhancement of long bones, pelvis,
skull and increasing activity in lungs, kidneys, liver, evidence medullar
expansion. Gastric aspirate and sputum induction were positive for acid
fast bacilli (AFB). PCR for Mycoplasma pneumonia and Chlamydia one
were negative, blood culture isolator, Viral IF, Legionella antigen were also
negative (urine). Eye exam was normal.
So, our diagnosis was disseminated TB (Pulmonary, lymph nodes, liver,
spleen, kidney localizations) with possible bone marrow involvement.
Co morbidities were severe malnutrition (BMI=14 kg/cm2
) and severe
anemia. HIV serologies were negative twice. An anti-TB treatment
was initiated (RHZE 2 months; RH 10 months), adjuvant therapy (oral
Pyridoxine) and nocturnal gavage (a total of 74 days), psychosocial and
multidisciplinary fellow-up. She appeared stable the first week on anti-TB
treatment, but not apyretic (38°-39°C), tachycardia, dyspnea at rest, gallop
was noticed. Echo-cardiography showed left ventricular dilatation with
diffuse ventricular hypokinesia and severe diastolic dysfunction; EKG
found signs of left venticular dilatation. We conclude associated dilated
myocardioathy probably multifactorial etiologies (tuberculosis, anemia
and severe malnutrition). She benefited digoxin, diuretics, captopril,
Steroids (Prednisone (1 mg/Kg/jour) and blood transfusion. Despite
the start of the anti-TB treatment, she continued to have persistent
fever (peak fever 40°C), both diffuse bone pains persisted and liver
transaminases increased. Chest x ray (day 28 anti-TB) (Figure 4) showed
no improvement, and with increasing infiltration. Abdominal ultrasound
(repeated) Liver enlarged (9.2 cm) and scattered by multiple nodules,
Spleen stuffed micronodules, innumerable abdominal lymph nodes and
Kidney lesions. Differential diagnosis on day was: multiple resistant TB,
neoplasia (lymphoma), related anti-TB drug (drug toxicity), connective
tissue diseases or other immune diseases. Performed bronchoscopy
and bronchoalveolar lavage (BAL) found enlarged carina with a 20%
reduction in calibrum of the main right brochus and inflammatory
mucosae; Culture isolated Hemophilus, klebsiella, Strepotococcal species
in BAL specimen and no Mycobacterium, Legionnella or Pneumocystis.
We did further investigations including immunology and hematology
assessment (both normal exhaustive immunologic work-up and normal
bone marrow biopsy) and comprehensive assessment: infectious work-up
(viral, parasites and fungi) negative, spleen biopsy (auramine negative),
liver biopsy x 2 (auramine doubtful) and normal lumbar function. At the
end of the first month of the anti-TB treatment, we decided to modify the
anti-TB regimen by introducing second-line anti-TB drugs (IV Amikacin,
Levaquin and Ethionamide, and Ethambutol discontinued). And steroid
doses (Prednisone) increased up to 2 mg/Kg/day. Finally we got astrain
susceptibility and the strain Mycobacterium tuberculosis was very sensitive
to R, I, Z, and E and the treatment was continued by RH for 10 months.
Our final diagnosis was immune reconstitution inflammatory
syndrome (IRIS) in a negative HIV-patient. It took many weeks before
IRIS manifestations decreased despite a good gain weight, resolution
of CX ray abnormalities and recovery of cardiac function. She was
discharged after five months of hospitalization. Chest X ray on 6 monthanti-TB
(Figure 5).
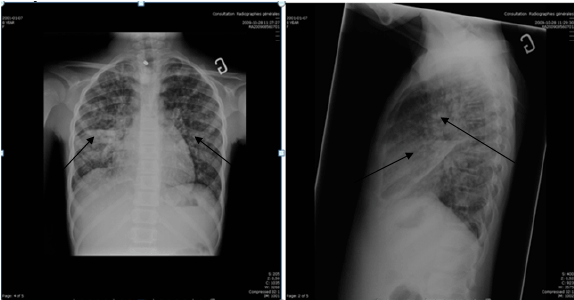
Figure 1 : Initial chest x Ray of the of an HIV negative 8 years-old girl who did develop immune reconstitution inflammatory syndrome during her treatment
for disseminated Tb, showed small bilateral foci of alveolar consolidation distributed in both lungs (black arrows), no pleural effusion and no cavitation.
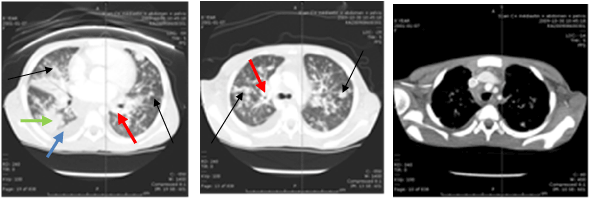
Figure 2 : CT scan of our 8-old-year patient showed diffuse bilateral nodular lesions in both lungs parenchyma (black arrows), associated with multiple
mediastinum lymphadenomegaly (blue arrows), right lower lobe atelectasis (green arrow), slight right pleural effusion (marroon arrows).
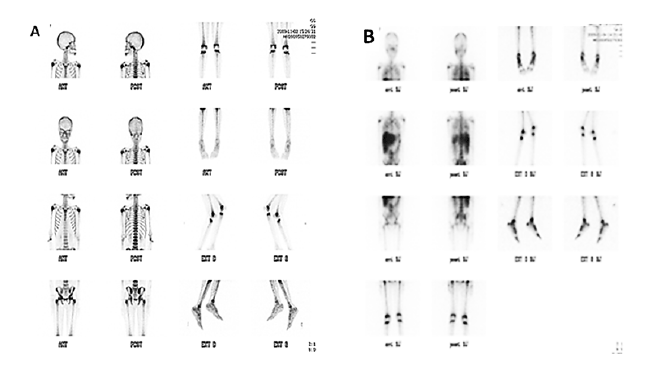
Figure 3 : Technicium bone scan (A) and Gallium bone scan (B) of an HIV negative 8 years-old girl who did develop immune reconstitution inflammatory
syndrome during her treatment for disseminated Tb, showed mild diffuse enhancement of bones and increasing activities in both lungs, kidneys, liver,
evidence of medullar expansion.
Discussion
IRIS is a phenomenon observed in patients recovering from
immunodeficiency. Generally, two clinical scenarios commonly occurred:
an unmasking IRIS underlying unrecognized infection flares after starting
ART; and paradoxical IRIS, resulting as the exacerbation of an often past
known treated infection [5]. Pathogenesis of IRIS involves a combination
of underlying antigenic burden, the degree of immune restoration
following HAART, and host genetic susceptibility. These pathogenic
mechanisms may interact and likely depend on the underlying burden of
infectious or noninfectious agent [5-7].
In children, IRIS incidence is estimated between 12 and 19% of all
children who initiate antiretroviral therapy [2-4] with a higher incidence
in patients who have been diagnosed with an active opportunistic infection
at the time of ART initiation [8,9]. Most data concerning IRIS originated
from limited resources settings. Risk factors for the development of IRIS
include a high baseline HIV viral load before the initiation of ART, rapid
decline in HIV viral load after initiation of ART, a lower baseline CD4
count before ART initiation, a high antigenic burden and disseminated
opportunistic infection, initiation of ART concomitant with or soon after
the initiation of the treatment of an acute infection, and lack of exposure
to ART before initiating the current regimen [7]. IRIS causes in HIVinfected
patients include infectious etiologies and non infectious causes
[6]. Recent report also documented IRIS due to Mycobacterium bovis
BCG in HIV-infected children in whom ART was initiated [10,11]. TB
associated IRIS is characterized by a clinical worsening after the start
of ART in patients receiving Tb treatment. It can also be diagnosed as
a new presentation of TB that is «unmasked» in the weeks following
the initiation of ART and the exclusion of other causes that could explain
deterioration [5].
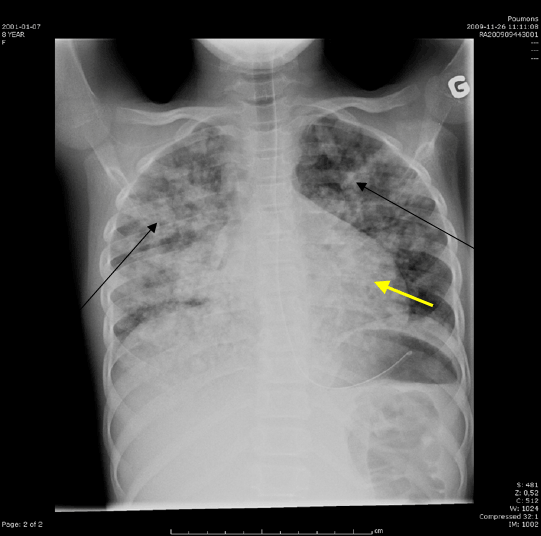
Figure 4 : Chest x ray (28 days on anti-Tb treatment) an HIV negative
8 years-old girl who did develop immune reconstitution inflammatory
syndrome during her treatment for disseminated Tb, showed no
improvement with bilateral diffuse lungs infiltrations (black arrows) and
cardiomegaly (yellow arrow).
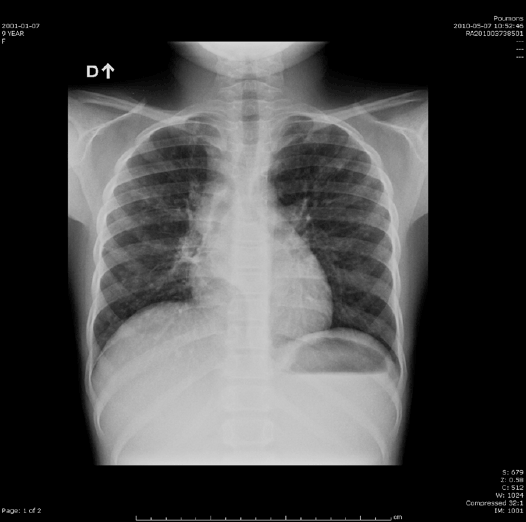
Figure 5 : Chest X ray (6 months on anti-TB) an HIV negative 8 yearsold
girl who did develop immune reconstitution inflammatory syndrome
during her treatment for disseminated Tb, showed improvement with
disappearance of radiological infiltrates.
TB is among the most frequent reported pathogen associated with IRIS.
TB-IRIS can have a variety of clinical manifestations: fever, pulmonary
disease, disseminated TB, systemic reaction inflammatory syndromes,
acute renal failure [12]. Transient worsening of patient has been reported
after the initiation of ART in severely immune compromised patients
[13,14] and paradoxical CNS TB reactions are well described in both
children and adult HIV-negative patients [15,16]. No treatment has been
prospectively studied in IRIS patients. Non-steroidal anti inflammatory
drugs (NSAIDS) or corticosteroids are commonly used to decrease
inflammation in patients with severe IRIS [7,17].
Indeed, IRIS is not previously described in literature in negative HIV
patients. We think that severe malnutrition is the main clinical risk causing
immune impairment by developing IRIS in negative HIV patients. The
mechanism is similar unlike in positive HIV by the restoration of the
immune system. Genetic host susceptibility may certainly play a role in
the development of this syndrome.
We concluded from an extensive review of the literature that this
case highlights a paradoxical immune mediated reaction similar to
the previously described IRIS in infected HIV patients but this time to
Mycobacterium tuberculosis during an appropriate antibiotic treatment.
Such an IRIS resulted in an apparent clinical deterioration but turned out
not being a failure to treatment. In the future, early recognition of an IRIS
in tuberculosis might prevent unnecessary changes in anti-TB regimen.
Acknowledgments
We thank the Ste-Justine University Hospital Center, particularly the
Pediatric Pulmonology staff and Infectious diseases staff, Dr. Guy Lapierre,
Dr. Jacques-Edouard Marcotte, Dr. Denis Bérubé, Dr. Chantal Buteau, Dr.
Sheila Jacob and Professor Mamadou Ba (Albert Royer National Hospital
Children’s Center, Dakar (Senegal), my mentor for all support during our
Pediatric training at Dakar and fellowship at Montreal.
Conflict of Interest
None
Article Information
Article Type: Case Report
Citation: Ba ID, Thiongane A, Ba A, Faye PM,
Sow A, et al. (2015) Immune Reconstitution
Inflammatory Syndrome in a Child with
Disseminated Tuberculosis. Autoimmun Infec Dis
2(1):doi http://dx.doi.org/10.16966/2470-1025.110
Copyright: © 2015 Ba ID, et al. This is an openaccess
article distributed under the terms of
the Creative Commons Attribution License,
which permits unrestricted use, distribution, and
reproduction in any medium, provided the original
author and source are credited.
Publication history:
Received date: 09 Sep 2015
Accepted date: 05
Dec 2015
Published date: 13 Dec 2015