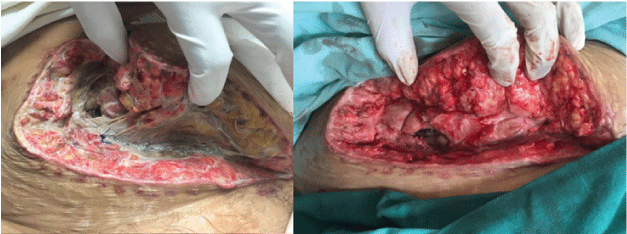
Figure 1: Infected surgical wound before (left) and after (right) vacuum assisted closure.


Francisco Reyna-Sepúlveda1 Aurora Ponce-Escobedo1 Asdruval GuevaraCharles2 Miguel Escobedo-Villarreal2 Edelmiro Perez-Rodríguez2 Gerardo Muñoz-Maldonado1 Marco Hernandez-Guedea2*
1Department of General Surgery, Universidad Autónoma de Nuevo León, University Hospital “Dr. José Eleuterio González”, Monterrey, México*Corresponding author: Dr. Marco Hernandez-Guedea, Department of General Surgery University Hospital “Dr. José Eleuterio González” Francisco I. Madero Pte. y Av. Gonzalitos s/n Monterrey, México, Tel: +52 (81) 834678 00; E-mail: publications.uanl@gmail.com
Background: Kidney transplantation is the most cost-effective therapy for end-stage renal disease. Postoperative complications account for 15 to 17%of all cases and are associated with significant morbidity. Currently 4.8% of post-transplant patients have returned to dialysis. Our center’s main transplant origin is cadaveric donation.
Objective: To review surgical complications of kidney transplant over the past 5 years.
Material and Methods: This was a retrospective, observational, descriptive study that included all patients from 2011 to 2015. The variables were analyzed with statistical software.
Results: A total of 55 cases were reviewed. Diabetic nephropathy was the etiology in 30.9%. Postsurgical complications occurred in 12.7% of patients with a postoperative mortality of 4%. Graft survival at 1 year was 82.4% with a 91% 1-year patient survival.
Discussion: Surgical complications in kidney transplantation are usually associated with reoperation and can significantly affect graft survival. To minimize the morbidity and mortality, diagnosis should be established promptly in order to provide appropriate treatment. Surgical complications can be minimized with standardization of the transplantation technique and organs must be used in their best condition.
Conclusion: Early identification and treatment of these complications is critical for patient and graft survival. Complications are low but significant.
Renal transplantation; Surgical complication; Graft survival
Kidney transplantation is considered the mostcost-effective therapy for end-stage renal disease (ESRD). Since the first human kidney transplant by Joseph Murray in 1954 [1], improvements in morbidity and mortality have been attributed to patient selection, advances in surgical technique, perioperative management, and immunosuppressive regimens.
Complications of renal transplantation can be classified as pathologic or surgical. Pathological complications include rejection, infection or cardiovascular events, while surgical complications involve vascular and urological complications, lymphocele, wound infection, or herniation.
Despite all advances, graft-endangering complications are primarily of a vascular etiology. Vascular complications account for 3%-15% of all cases [2]. These include thrombosis or stenosis of the renal artery or vein. Other rare complications are the formation of aneurysms, arteriovenous fistulas or hematomas, which can be diagnosed and treated by interventional radiology. Some risk factors include poor surgical technique, torsion or compression of vessels, the presence of multiple renal vessels ora renal artery atheroma. Other factors involved are the presence of anatomical variations, such as double ureters and multiple renal arteries or veins which represent a challenge for the transplant surgeon. The most common variant which is found in 8%-30% of all potential kidney donors is the presence of multiple renal arteries [3].
According to the United Network of Organ Sharing (UNOS), graft survival at 2 years is 90%; however, in 5-year surveillance reports, 30% of patients have lost the graft or died with a functioning kidney [4]. The main causes of early graft failure (first 6 months) are acute rejection, technical problems, and a nonviable kidney [5]. In the case of chronic failure, it is caused by death because a non kidney related problem with a functional graft and chronic kidney disease. Currently, 4.8% of post-transplant patients have returned to dialysis [6].
Knowledge of the incidence of clinical manifestations and management of surgical complications is necessary for all kidney transplant surgeons; therefore, our objective is to report our experience with the epidemiology, perioperative variables, and surgical complications of kidney transplants performed over the past 5 years.
A retrospective, observational and descriptive study was conducted between 2011 and 2015 at the “Dr. José Eleuterio González” University Hospital. All renal transplant recipients were included. Demographic variables, medical history, laboratory tests, details of the surgical procedure and post-transplant follow-up at 3 months and 1 year were reviewed.
Patients with a complete transplant protocol, including cardiovascular, infectious, psychosocial and urological evaluations were included. All patients underwent panel reactive antibodies (PRA), human leukocyte antigens (HLA), and cross-lymphocyte cytotoxicity tests. All patients signed informed consent and all organ donations were cadaveric.
Regarding the surgical technique, after washing the bladder with amikacin and placement of a Foley catheter, we proceeded to place the donated kidney in the right or left iliac fossa using side-to-end anastomosis of the renalartery and vein to the iliac vessels. To perform neoureterostomywe used the antireflux Laedbetter-Politano and the Lich-Gregoir techniques with double J catheteraccording to the decision of the operating surgeon. Postoperative immunosuppression regime includedprednisone, tacrolimus and mycophenolate mofetil. Statistical analysis was performed using SPSS v20 (SPSS, Inc., Chicago, Ill).
A total of 55 heterotrophic cadaveric kidney transplants were performed in 31 (56%) men and 24 (43%) women. Mean age was 49.5 years and mean body mass index was overweight. The origin of the nephropathy was type 2 diabetes mellitus in 17 (30.9%), hypertension in 10 (18%), and other pathologies, such as nephrolithiasis, glomerular diseases, or idiopathic in the remaining 28 patients. Of the total, 48 patients were on renal replacement therapy for a mean time of 2.18 years with 21 (43%) patients in hemodialysis and 27 (57%) in peritoneal dialysis (Table 1).
Total |
55 (100) |
Sex |
|
Male |
31 (56) |
Female |
24(43) |
Body Mass index (average) |
27 |
Nephropathy |
|
Diabetes Mellitus type 2 |
17(30.9) |
Arterial Hypertension |
10(18) |
Other |
28(51.1) |
Kidney replacement therapy |
|
Time (years) |
2.18 |
Hemodialysis |
21(43) |
Peritoneal Dialysis |
27 (57) |
Surgical Procedure |
|
Cold Ischemia (hr.) |
8.4 |
Surgical time (hr.) |
4.15 |
Transoperative bleeding (ml) |
450 |
Multiple renal arteries |
9(16) |
Table 1: Sociodemographic factors in kidney transplant n(%)
From the transplanted kidneys a total of 49 (89%) were allocated in the right iliac fossa and 6 (11%) in the left. Out of the donated specimens, 33 (60%) were right kidneys and 22 left (40%).
The mean times of cold ischemia were 8.44 hours and 4.15 hours for the surgical procedure with a mean approximate total blood loss of 450mL. Nine (16%) patients had multiple renal arteries (MRA). In the immediate postoperative period, only 1 patient received more than 3 units of packed red blood cells and 4 patients remained in critical care for more than 48 hours. The mean hospital stay was 12 days. For ureterovesical anastomosis, we performed 47 (85%) Laedbetter-Politano and 7 (12%) Lich-Gregoir pr ocedures. Vascular anastomoses were performed side-to-end and in one case with three renal arteries they were anastomosed together with a cadaveric graft iliac vein.
There were 7 (12.9%) postsurgical and 5 (9%) chronic complications, stratified by etiology (Table 2). Two (4%) deaths were reported from nosocomial pneumonia and pulmonary embolism, and two (4%) deaths in the subsequent three months because of abdominal sepsis. There was only one patient which graft loss can be related to a surgical complication which related to a ureteral injury with ferula placement and posterior urinoma formation.
Postoperative |
7(12.9) |
Hematoma |
2(3.6) |
Wound Infection |
2(3.6) |
Ureteral injury |
1(1.8) |
Arterial injury |
1(1.8) |
Venous thrombosis |
1(1.8) |
Chronic |
5(9) |
Incisional hernia |
2(3.6) |
Arterial stenosis |
1(1.8) |
Vesicoureteral occlusion |
1(1.8) |
Linfocele |
1(1.8) |
Table 2: Surgical complications of kidney transplants
Preoperative, postoperative and 3-month follow-up laboratories were reviewed (Table 3). For statistical analysis, we compared the groups with and without complications, obtaining only preoperative creatinine and albumin, in addition to the delay in organ function, as statistically significant (p=<0.05).
Value |
Preoperative |
Postoperative |
3-month follow-up |
Blood Urea Nitrogen |
59.9 |
34.1 |
22.5 |
Creatinine |
9.6 |
1.7 |
1.3 |
Hemoglobin |
11.1 |
9.4 |
11.6 |
Albumin |
3.7 |
2.8 |
4.2 |
Tacrolimus blood level |
- |
12.8 |
10.6 |
Table 3: Preoperative, postoperative and follow-up laboratories of uncomplicated patients.
Most of our patients were overweight with an average BMI of 27, which represented an extra challenge in surgical procedures. In our surgical technique we commonly transplant on the right side of the patient, transplant was done in the left side in the case of a previous transplant or surgical history.
Our prevalence of 16% of multiple renal arteries represented a surgical challenge; we performed multiple anastomosis or used iliac vein graft to gain additional working space. In the case of the different techniques used for the vesicoureteral anastomosis this was chosen by the surgeon according to their experience.
In regards of our surgical complications, the hematoma formation was related to a biopsy and in both cases open surgery was the preferred approach. Wound infection was treated with a vacuum device without complications (Figure 1). The ureteral injury was a small puncture during dissection which as primarily repaired and a ferula placed. The arterial injury was related to an injury of a polar artery which was ligated. In the case of venous thrombosis surgical management was preferred.

Figure 1: Infected surgical wound before (left) and after (right) vacuum assisted closure.
Chronic complications include incisional hernia which was repaired with mesh at 6 months. The arterial stenosis was managed with endoluminal stent placement. The case of vesicouretheral occlusion with ferulization of the anastomosis. Conservative treatment was performed in our case of linfocelelymphocele.
Laboratory results include the expected decrease in blood urea nitrogen and creatinine. With an important increase in albumin in the following 3-month period. Tacrolimus blood levels remained stable in the followup. The statistical analysis performed the only significant variables by comparing complicated uncomplicated patients were creatinine, albumin and delayed the start of the graft function. Low albumin and elevated creatinine in these groups is associated with a delay and an inability of tissues to carry out a proper repair process.
Surgical complications in kidney transplantation are usually associated with reoperation and can rapidly and severely affect graft survival. To minimize the morbidity and mortality, we must quickly diagnose and treat appropriately. Surgical complications rarely lead to graft loss, with the exception of vascular pathology [7]. In our study we report an incidence of 12.7% of surgical complications similar to the 15.9% reported in the literature [8].
Renal arterial stenosis is the most common vascular complication; it occurs in 3 to 23% of all transplantations [9] in the first 12 months [10]. This rate has been associated with the end-to-end anastomosis and cadaveric donor grafts. If left untreated this pathology leads to kidney dysfunction, resistant hypertension and subsequent deterioration of the graft [11]. The main management is the endoluminal percutaneous angioplasty with or without stenting and the main marker of recovery are improved renal function and blood pressure.
The incidence of arterial thrombosis is 0.3 to 6.1% [12]. It is most common in the first 2 weeks after transplantation, 80% in the first month and 93% in the first year [13]. After the first month, thrombosis of the renal artery occurs mainly because of rejection or a high degree of stenosis.
In the case of the renal vein thrombosis, it is due to the spread of deep vein thrombosis from the lower extremity or extrinsic compression by a collection. It is characterized by the presence of oliguria, hematuria, elevated creatinine and pain at the surgical site [14]. Thrombosis is diagnosed with Doppler ultrasound and treated surgically by laparotomy, thrombectomy or even graft nephrectomy. There are multiple reports for endoluminal management of thrombosis, however the role of interventional radiology is not well defined [15].
The renal vein thrombosis is a dramatic early vascular complication of renal transplantation, with a reported prevalence between 0.5 and 4%. Despite its low incidence, it is one of the most important causes of graft loss in the first month after transplantation [16].
Hematoma formation is a frequent minor vascular complication that occurs in the postoperative period. The most common source is a small leakage of the vascular anastomosis or minor bleeding from the renal surface or the surrounding tissues and when they grow and produce clinical signs or symptoms by external pressure, it may lead to graft dysfunction and later to thrombotic complications. Ultrasound or computerized tomography (CT) are used for diagnosis [17]. Hematoma may cause complication with the presence of infection and can be treated with ultrasound-guided percutaneous drainage. Large hematomas in the immediate postoperative associated to hypovolemic shock should be treated as a surgical emergency.
Urological complications are the most common complications in the late period after kidney transplantation, presenting an incidence ranging from 2.5 to 12.5% [18], lower compared to the beginning of renal transplantation era of 25% [19]. These complications are a major cause of morbidity, delayed graft function and increased hospitalization costs. The decrease in the vascular irrigation of the donor ureter and failure in surgical technique are the leading causes of urological complications [20].
High immunosuppressing regimens, acute rejection, BK virus and infection also lead to the obstruction. A low steroid protocol and meticulous surgical technique decreases the incidence [21] and the use of older graft donors increases it. Different techniques for the ureterovesical anastomosis have been described,however they do not affect the incidence of complications [21]. Stenosis and fistula are the main causes and managed with open surgery.
In the case of multiple renal arteries (MRA), many centers prefer not to use these kidneys when they coexist other suboptimal conditions such as advanced donor age [22]. The main concerns are the technical complications associated with anastomosis and graft loss. More than 70% of the kidneys have a single renal artery [23] which normally divides in the hilum. Sometimes it can bifurcate early or near the origin of the aorta, and the only option to take the kidney and divide the segmental arteries and performing a challenging anastomosis when they are short. The increased difficulty and more time needed for these anastomosis also increased the ischemia time causing a negative impact on the rate of acute tubular necrosis (NTA) and graft survival [24].
Sometimes a small low polar renal artery may be the only irrigation the ureter has so these vessels cannot be sacrificed during implantation [25] if this vessel results in thrombosis the ureter will become ischemic followed by necrosis [26]. In these cases, when MRA are present complications such as stenosis and thrombosis are more likely to occur [27], however it does not adversely affect patient or graft survival [28].
In the case of a lymphoceles they occur during the dissection process by opening the lymphatics. In most patients these fluid collections are asymptomatic and are finding in the ultrasound not requiring invasive treatment. Larger collections may be associated with dilation of the collector system, pain, fever or declined renal function. In these cases, ultrasound-guided aspiration should be performed. Some cases may require the use of a nephrostomy tube although another option is to open non infected lymphocele to peritoneal cavity. Wound infection rate in these patients is high due to the immunosuppression, but may be managed medically with antibiotics. Hernia formation requires surgical repair.
The 4 deaths we report were in patients older than 60 years, with type 2 diabetes mellitus, hypertension and in renal replacement therapy. One mortality was directly associated with graft failure at 3 months because of a ureteral injury with posterior urinoma formation. We were not able to establish statistical significant prognostic factors for surgical complications based on complications in the statistical analysis.
Kidney transplantation is the treatment of choice for ESRD, surgical complications which despite few occur; when present they may have a significant impact in the graft. It is recommended that routine and emergency examinations with Doppler ultrasound be done for graft preservation. Surgical complications of kidney transplantation can be minimized with standardization of surgical techniques; additionally each organ should be used in its best condition. Early identification and treatment of these complications is critical to patient and graft survival. Thanks to the advances in different surgical complication therapies kidney transplantation continues to be a safe treatment option.
We thank Sergio Lozano M.D. and Alejandro Quiroga-Garza, M.D. in their editing of this manuscript.
Download Provisional PDF Here
Article Type: Research Article
Citation: Reyna-Sepúlveda F, Ponce-Escobedo A, Guevara CA, Escobedo-Villarreal M, Perez-Rodríguez E, et al. (2016) Outcomes and Surgical Complications in Kidney Transplantation. Transplant Res J 1(2): doi http://dx.doi.org/10.16966/2473-1730.107
Copyright: © 2016 Reyna-Sepúlveda F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access