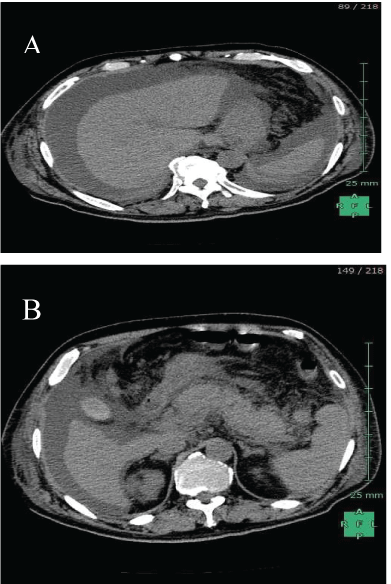
Figure 1: (A) CT revealed hepatatrophy and ascites accumulation. (B) CT also revealed swelling of the pancreas and disproportionate fat stranding around the pancreas.


Akira Umemura* Hiroyuki Nitta Koki Otsuka Takeshi Takahara Yasushi Hasegawa Akira Sasaki
Department of Surgery, Iwate Medical University, Japan*Corresponding author: Akira Umemura, Department of Surgery, Iwate Medical University, 19-1 Uchimaru, Morioka, 020-8505, Japan; E-mail: aumemura@iwate-med.ac.jp
This paper reports a rare case of acute pancreatitis (AP) caused by hepatitis B virus (HBV) in a patient who underwent deceased donor liver transplantation (DDLT). A 63-year-old Japanese man had jaundiced skin and had been suffering from anorexia for the past two weeks. Routine laboratory investigations revealed severe liver dysfunction with an active HBV infection and severe hepatic encephalopathy; hence, the patient was diagnosed with acute liver failure (ALF). A computed tomography (CT) examination also revealed swelling of the pancreas. Although this was suggestive of AP, it was asymptomatic at that point. Five days after admission, the patient underwent a DDLT. Liver dysfunction was observed at approximately two weeks after the DDLT. A CT examination revealed a walled-off necrosis (WON) between the stomach and pancreas; thus, we performed drainage of the WON via a laparotomy. The patient’s condition gradually improved and he left the intensive care unit on the 35th day after the DDLT. In this case, WON formation by AP caused severe liver dysfunction after DDLT. Therefore, we have to consider WON formation as a cause of postoperative liver dysfunction in patients with AP rather than acute rejection or hepatic artery thrombosis.
Acute pancreatitis (AP) can complicate the course of viral hepatitis, especially in cases of acute liver failure (ALF) [1,2]. Lisney [3] was the first to state that pancreatitis could be a complication of infectious hepatitis. In an autopsy series, Joshi et al. [4] reported that 3.7% of patients with AP also had viral hepatitis, whereas the overall autopsy incidence of viral hepatitis was 0.9%. As shown in some case series, AP occurring with ALF is relatively common and has been well documented [5,6].
Generally, AP invariably complicates persistent organ failures caused by cytokine cascades. These cascades result in a systemic inflammatory response syndrome during the early phase. Patients who develop persistent organ failure within the first few days of the disease are at an increased risk of death, with mortality reported to be as great as 36-50% [7,8]. Furthermore, AP often causes sepsis due to the bacterial translocation of enteric bacteria from the beginning to the end of the disease; therefore, sepsis can also be a fatal factor for AP patients.
In this paper, we will describe the first case report of a DDLT for a patient with ALF caused by the hepatitis B virus (HBV) and complicated by the presence of AP.
A 63-year-old Japanese man had jaundiced skin and had been suffering from anorexia for the past two weeks; he was referred to our clinic for further treatment. Routine laboratory investigations revealed severe liver dysfunction with a serum total bilirubin level of 23.6 mg/dL; an aspartate aminotransferase (AST) level of 781 IU/L; an alanine aminotransferase (ALT) level of 1473 IU/L; a prothrombin time-international normalized ratio of 4.98; a hepatitis B surface antigen (HBs-Ag) level of 2180.0 IU/ mL; and HBV-DNA level of 6.6 log copies/mL. Additionally, a computed tomography (CT) examination revealed ascites accumulation, and swelling of the pancreas (Figures 1A and 1B). As a result, the patient was diagnosed as having acute hepatitis due to the HBV. In addition, severe hepatic encephalopathy appeared before the day was over; therefore patient was diagnosed as having ALF and was registered as the most urgent candidate for a DDLT. Five days after admission, the patient underwent a DDLT from a brain-dead donor who had experienced a subarachnoid hemorrhage. Histo-pathological findings revealed that necrosis of hepatocytes widely occurred and only a few normal hepatocytes survived in nodular formation. Moreover, even survived hepatocytes were affected with degeneration and severe cholestasis (Figures 2A and 2B).

Figure 1: (A) CT revealed hepatatrophy and ascites accumulation. (B) CT also revealed swelling of the pancreas and disproportionate fat stranding around the pancreas.
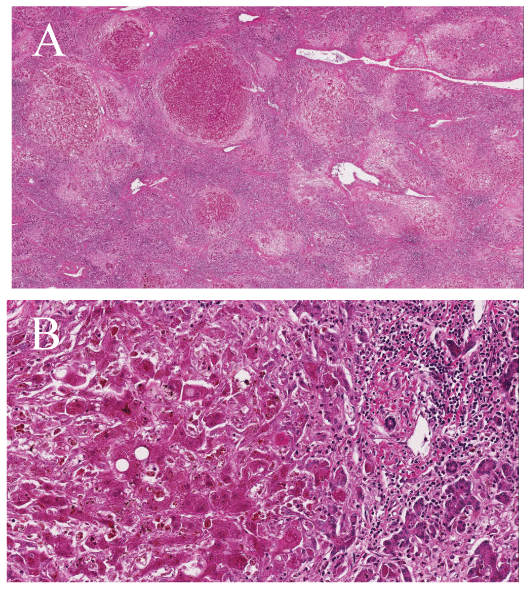
Figure 2: (A) Necrosis of hepatocytes widely occurred and only a few normal hepatocytes survived in nodular formation ( × 20). (B) Survived hepatocytes were affected with degeneration and severe cholestasis ( × 200).
Two weeks following the DDLT, the patient’s AST and ALT levels suddenly elevated and a CT examination revealed a walled-off necrosis (WON) caused by AP between the stomach and pancreas (Figures 3A and 3B). As a result, we performed drainage of the WON via a laparotomy. The patient’s liver function gradually improved after the drainage. He left the intensive care unit on the 35th day after the DDLT. He is now in rehabilitation to improve his physical strength.

Figure 3 A and B: CT revealed WON between the stomach and pancreas with a well-enhanced capsule.
Although the relationship between AP and viral hepatitis has been known for 50 years, the exact mechanism by which these viruses behave is still unknown [6]. Most cases of AP that are complicated by acute viral hepatitis occur in conjunction with ALF, including the HBV, the hepatitis A virus, and the hepatitis E virus [1,5,6]. Several mechanisms have been proposed, including the direct cytopathic effect that the virus has on pancreatic acinar cells. Documentation of HBs-Ag and hepatitis B core antigens in the pancreatic acinar cells and pancreatic juice supports this hypothesis [9,10]. Furthermore, Cavallari et al. have also detected HBVDNA in the cytoplasm and nuclei of scattered pancreatic acinar cells [1]. One possible mechanism for this is the development of ampullary edema with obstructions to the outflow of pancreatic secretions. Another potential mechanism is the release of lysosomal enzymes from the inflamed liver into the circulation with the activation of trypsinogen to trypsin from virus-damaged acinar cell membranes [6]. Although AP in ALF seems like a relatively common phenomenon, it is often asymptomatic. It is of interest that these patients’ mortality is not found to be significantly different from patients without this complication. This is likely because AP is usually not severe, and survival is more dependent on the severity of the ALF.
In this case, WON occurred at an early stage after DDLT and caused severe liver dysfuction. Typically, WON consists of necrotic tissue being contained within an encasing wall of reactive tissue; this is a mature, encapsulated collection of pancreatic and peri-pancreatic necrosis that has an inflammatory wall. This maturation usually occurs at least four weeks after the onset of AP [7]. In this case, the WON maturation was formed over a shorter period. The following points can be given as reasons. The first reason is that immunosuppression due to surgical stress and immunosuppressants exacerbated the WON maturation. A second reason is that there was a physiologic space between the stomach and the pancreas. The patient required immediate improvement for liver dysfunction; therefore, we performed an open drainage.
This was the first case report of a DDLT for a patient with ALF caused by HBV and complicated with AP. As AP occurring with ALF may often be asymptomatic and is not usually severe, survival is more dependent on the severity of the ALF. In this case, the surgical stress of the DDLT and the immune depression caused by ALF might have affected the progression of WON formation. Hence, it is important to pay attention to the possible concurrence of AP when we treat a patient with ALF caused by hepatitis viruses. Furthermore, if the patient undergoes a liver transplantation, we have to consider WON formation as a cause of postoperative liver dysfunction in patients with AP rather than acute rejection or hepatic artery thrombosis.
Akira Umemura and co-authors have no conflict of interest.
The authors would like to thank Dr. Naoko Ito, Dr. Hidenori Itabashi, Dr. Hirokatsu Katagiri and Dr. Shoji Kanno for helpful discussions.
Download Provisional PDF Here
Article Type: Case Report
Citation: Umemura A, Nitta H, Otsuka K, Takahara T, Hasegawa Y, et al. A Rare Case of Acute Pancreatitis Caused by Hepatitis B Virus Underwent Deceased Donor Liver Transplantation. Transplant Res J 1(1): doi http://dx.doi.org/10.16966/2473-1730.103
Copyright: © 2016 Umemura A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Publication history:
All Sci Forschen Journals are Open Access