Introduction
Local delivery of an antimicrobial to the site of infection has been established as an effective way of achieving therapeutic levels of the drug while avoiding the possible adverse effects of systemic administration [1]. This approach to the use of antimicrobials has been a major focus for the prevention as well as treatment of osteomyelitis and infections resulting from orthopedic implant surgery as well as per-implantitis associated with implants in the oral cavity [2]. Placement of an antimicrobial loaded osteoconductive scaffold within a bone site has been used to create a reservoir of the drug that can be maintained at an optimal concentration for a prolonged period of time, reducing the potential for infection while simultaneously providing a regenerative scaffold for new bone formation [3].
Calcium sulfate hemihydrate has been demonstrated to be a successful regenerative bone material in various in-vitro, animal, and human clinical studies [4-7]. This material has also has been used for many years as a carrier of antibiotics with reports of its use with the antimicrobial, Rivanol, in the treatment of osseous infections in dogs dating back to 1928 [8]. More recently calcium sulfate has been used as a delivery of antibiotics as documented in a number of clinical and experimental reports [5,9- 12]. These reports highlight the general effectiveness of placement of antimicrobial loaded calcium sulfate at osseous sites to serve as both bone filler and a source of agents to prevent and/or treat infections in skeletal tissues. In an attempt to overcome some of the drawbacks such as undesirable fast resorption rates [13-15] associated with conventional sized, medical grade calcium sulfate products, a nano-crystalline form of calcium sulfate (nCS) hemihydrate has been developed in our laboratory. This nCS has been shown to undergo sustained degradation when compared to conventional calcium sulfate (CS) and to be capable of supporting bone regeneration in a critical sized-mandibular bone defect in a canine preclinical model [16,17]. In vitro studies have shown that the material is virtually non-toxic and can be used as an effective vehicle for the delivery of preloaded growth factors such as platelet derived growth factor (PDGF-BB) and bone morphogenetic protein (BMP-2) [16,18]. The potential effectiveness of nCS as a vehicle for antibiotic release has not been previously studied.
Tetracycline derivative antimicrobials represent a group of drugs commonly known for their broad-spectrum nature and bacteriostatic properties [19]. Furthermore, they have been shown to significantly increase the proliferation of human osteoblastic cells without negatively altering their functional activity [20]. The long-term exposure to these drugs has been reported to increase the number of active osteoblastic cells and to increase mineralized matrix formation suggesting a potential therapeutic approach for enhanced bone formation [20]. In this present study, the release of two tetracycline derivative antibiotics, minocycline and doxycycline, commonly used in minimize infections associated with periodontal bone regenerative procedures and implant placements, as well as the effects of these released antibiotics on osteoblastic cell activity and differentiation were assessed in vitro. The goal of this study was to provide a rational approach for the use of nCS preloaded with minocycline or doxycycline in clinical procedures such as peri-implantitis that require both the control of bacterial infections as well as support of bone regeneration at specific local sites.
Materials and Methods
Preparation of Nano-calcium sulfate powder
Nanocalcium sulfate (nCS) was prepared from medical grade dehydrate calcium sulfate (CS) using techniques that have been previously described [16]. Calcium sulfate dihydrate solution was freeze-dried under cryogenic conditions under vacuum conditions to produce a cotton-like calcium sulfate powder. Using an oven drying process, the (nCS) powder was heated to temperatures between 135∼150°C for 30 minutes. At approximately 45°C, the dihydrate nCS was converted first to the hemihydrate form, and then at approximately 133°C, to the completely anhydrous form. The nCS was subjected to glow discharge treatment (GDT) for sterilization immediately before its use in the experiments [16].
Antibiotics and other materials
Doxycycline hyclate (DOXY) and Minocycline hydrochloride (MINO) were purchased from Sigma-Aldrich, St. Louis, MO, USA. Tissue culture supplements, fetal calf serum (FCS), α-minimal essential medium (α-MEM), and phosphate buffer saline (PBS) were obtained from Life Technologies, Inc (Grand Island, NY, USA). Culture flasks and disposable plates were obtained from Corning, Inc (Corning, NY, USA) and tryptic soy agar and brain heart infusion media were obtained from Becton, Dickinson, Inc (Franklin Lakes, NJ, USA).
Preparation of drug loaded nCS samples for antibiotic release
Each nCS sample either containing DOXY or MINO was prepared by mixing the nCS powder with a liquid phase in a 1:1 powder to liquid ratio of (100 mg/100 ul). The composition of the liquid phases consisted of 1 mg/ml of either DOXY or MINO that was prepared as a stock solution with nanopure distilled water. Each sample was made by loading a DOXY or MINO containing solution to obtain the drug at an amount of 100 ug/ sample. Powder and liquid phases were mixed manually for 1 minute until a pliable paste was developed. The final composite material was packed into a polyvinyl siloxine (PVS) mold to form disk-shaped pellets with dimensions of 3 mm of height and 1 mm in thickness; the material was then left to set for one hour so that solid sample pellets were formed. Control samples were prepared in a similar manner however; nanopure distilled water without an antibiotic preparation was used as the liquid phase.
Setting time of nCS
Gillmore apparatus needles (Humboldt Manufacturing Co, Schiller Park, IL, USA) were used to assess the setting times of nCS mixed with water or with 0.05 mM DOXY or MINO in a 1:1; liquid: powder solution. After nCS was mixed with its respective solution, the mixture was then placed in the polyvinyl siloxane molds, described above. The common practice for obtaining initial and final setting times with the Gillmore apparatus was used [21]. The initial setting time was recorded 5 minutes after the solution was first added to nCS. The initial weight (smaller and lighter weight) was rested every 2.5 minutes for 5 seconds when the weight no longer left a print on the nCS material; the final setting time weight was then used every 2.5 minutes for 5 seconds.
Vickers hardness
The Vickers hardness test was conducted using a Buehler Micromet 5104 Micro Hardness Testing Machine according to the manufacturer’s instructions. Disks of nCS with or without DOXY or MINO were fabricated as described above, waiting until each disk had reached maximum setting and could be lifted intact out of its PVS mold. Each group consisted of four disks.
Antibiotic release detection from nCS sample
DOXY and MINO release from nCS samples was conducted at 37°C in vitro with each test sample placed in a sealed brown glass bottle to protect the contained antibiotic from light. Five parallel test samples for each antibiotic and five control samples (without antibiotic) were assessed simultaneously to obtain an average concentration release pattern. Samples were placed in 1 ml of PBS that was then withdrawn completely and replaced with fresh buffer at predetermined time intervals of (2, 4, 6 hours) and (1-10 days). Aliquots of the test samples were immediately analyzed by UV-Visible spectrophotometer (Beckman Coulter DU-800 spectrophotometer) to determine the released amount from the sample pellet using standard calibration curves made for each antibiotic. The test samples were scanned at maximum absorbance values at 351 and 343 nm, for either DOXY or MINO respectively. These values had been determined using a solution of 100 ug/ml, prepared PBS for both DOXY and MINO, to be the maximum absorption wavelength of each antimicrobial agent.
The antibiotic concentrations of the test samples were plotted as log cumulative percentage drug release versus time to further study the release characteristics of the antibiotics from the nCS scaffold.
Preparation of drug loaded nCS samples for cell viability and differentiation
nCS control and experimental samples were processed into disk-shaped cylindrical pellets using the PVS mold described above. The control group (n=6) consisted of pure nCS powder that was mixed manually with double distilled water. Each sample in the control group was prepared by mixing 100 mg of nCS powder with 100 ul of double distilled water at a ratio of 1:1 for one minute until a pliable paste was developed. The composite material was then left to set for one hour so that solid sample pellets were formed. For the test samples, six experimental groups for each antibiotic (DOXY and MINO) were prepared by substituting the liquid part (distilled water) with a known antibiotic stock solution (5, 1, 0.5, 0.25, 0.05 mg/ml) and then mixed at the same ratio and time as the control group. Subsequently, there were groups containing 500, 100, 50, 25, 10 and 5 ug DOXY or MINO /pellet. Each experimental group had (n=6) samples.
Osteoblastic cell culture
Human osteoblastic cell cultures were obtained from specimens of alveolar bone from healthy subjects that underwent oral surgery. All specimens were obtained in accordance with the Human Subjects Institutional Review Board of the University of Buffalo, State University of New York. The details of the cell isolation and culture conditions have been previously described in detail [22].
MTT cell activity assay
Osteoblastic cells at a concentration of 20,000 cells per ml of media were incubated in 96 tissue culture well plates with either nCS (alone) samples or nCS containing antibiotics at various concentrations for 48 hrs at 37C in a CO2 incubator. At the end of the incubation period, MTT reagent (Sigma-Aldrich, St. Louis, MO, USA) was added to all the wells and the assay was conducted as previously described in detail [23].
Alkaline Phosphatase activity
Osteoblastic cells were incubated with nCS (alone) samples or with nCS with various concentrations of antibiotics as described above. After a 48 hr incubation period at 37°C, the media was removed and 200 ul of 1% Triton-X-100 was placed in each well for 1 hour at 4°C to lyse the cells. This was then followed by the addition of 2-amino-2-methyl-1-propanol buffer p-nitrophenol phosphate (Sigma-Aldrich, St. Louis, MO) incubation for another 1h at 37°C and an addition of 50 ul of 0.5 M NaOH to each well to stop the alkaline phosphatase reaction as previously described in detail [23]. Aliquots of each sample were transferred to fresh 96 well culture plates and absorbances were then read by a spectrophotometer (Bio-Rad laboratories, NY, USA) at 405 nM.
Antimicrobial activity of drug loaded nCS samples
To assess the antimicrobial activity of released antibiotics from the nCS samples, a strain of Porphyromonas gingivalis (ATCC 53977), kindly donated by Dr. J. Zambon of the University at Buffalo, was cultured anaerobically on tryptic soy agar supplemented with 5% sheep blood (Becton Dickinson Inc, Franklin Lakes, NJ, USA) for over 48 hours. A stab of the colony was suspended in prepared brain heart infusion broth (BHI) (Becton Dickinson Inc, Franklin Lakes, NJ, USA) to achieve a standard bacterial cell concentration of 1 × 105 CFU/mL per six 10 mL test tubes. nCS pellets containing a dose of 25 or 50 ug of either DOXY and MINO were then gently placed in 10 mL test tubes. A nCS pellet without any antibiotic was used as positive control and a test tube with P. gingivalis alone was used as negative control. All samples were then incubated for 24 hrs in an anaerobic environment composed of 80% nitrogen, 10% carbon dioxide and 10% hydrogen atmosphere. After incubation, turbidity of the tubes was compared to both positive and negative control and optical density was measured at 600nm. The presence or absence of growth turbidity within the tubes is an indirect measure of the ability of the released compound to inhibit that organism. Porphyromonas gingivalis was used in the experiment since it has been strongly implicated in ligature induced peri-implantitis, and has the ability to invade surrounding tissues [24].
Statistical analysis
Standard calibration curves were analyzed by linear regression analysis using Statplus™ software for Microsoft Excel at p<0.05. To analyze the difference between the experimental groups, one-way ANOVA test was followed by Tukey’s honestly significant difference (HSD) test for multiple comparisons with a significant level of 5%.
Results and Discussion
Setting time
The presence of the antibiotics appeared to have some, relatively slight, effects on the setting time of nCS under the conditions tested (Table 1). In particular, the presence of DOXY had significant increases in both the initial and final setting times, but MINO under the same conditions produced small, but not significant increases in these parameters.
| Material |
Initial setting time (min) |
Final setting time (min) |
| Ncs |
20.8 ± 2.3 |
8.3 ± 1.2 |
| nCS+DOXY |
36.6 ± 3.2 a) |
15.8 ± 1.6!a) |
| nCS+MINO |
25.8 ± 1.2 |
12.5 ± 2.0 |
Table 1: Setting time of nCS with/without antibiotics
Nanocalcium sulfate (nCS) samples were mixed for 1 min at room temperature, with either water only or an aqueous solution of either antibiotic (Doxycycline hyclate (DOXY) or Minocycline hydrochloride (MINO) (0.5 mg/ml), at a ratio of 1:1 (nCS: liquid) then let to sit for 5 min before the testing with a Gillmore Apparatus was conducted as described in the methods section. The weight was allowed to rest on the samples for 2.5 min intervals. The initial and final setting times were determined when there was no longer an obviously noticeable circular imprint left on the material. The data are the mean ± SD of three different spots on the surfaces of each material tested. a) Statistically significant difference from nCS (P<0.05).
Vickers hardness
Standard hardness testing revealed that the addition of either MINO or DOXY under the conditions used throughout this study, resulted in significant increases in this parameter compared to control nCS material without antibiotics added to the water used to make the disks shaped samples (Table 2).
| Material |
HV units |
| nCS |
11.78 ± 0.07 |
| nCS+DOXY |
16.28 ± 0.15a) |
| nCS+MINO |
17.85 ± 0.07a) |
Table 2: Vickers Hardness of nCS with/without antibiotics
Nanocalcium sulfate (nCS) samples were mixed for 1min at room temperature, with either water only or an aqueous solution of either antibiotic (Doxycycline hyclate (DOXY) or Minocycline hydrochloride (MINO) at 0.5 mg/ml, then let to sit in the molds until the disks would be removed intact and evaluated using a Micro Hardness Testing Machine according to the manufacturer’s instructions. HV=Hardness Vickers units. The data are the mean ± SD of 4 samples from each group. a) Statistically significant difference from nCS (P<0.05).
Antibiotic loaded nCS release profile
The in-vitro cumulative release percentage profiles for DOXY and MINO from the nCS scaffold are presented (Figures 1 and 2). Both antibiotics are characterized by two similar release stages. The first stage, involved a high initial burst release of MINO from the nCS sample pellet (59.6%) within the first 24 hours; this burst release was comparable and slightly larger than what was observed for DOXY in the same time period (55.8%). Measured release concentration for MINO was (38.1, 12.9, 8.7, 5.7 ug/ml) at (2, 4, 6, 24 hours). DOXY release concentration was 29.6 ug/ ml at 2 hours, 10.4 ug/ml at 4 hours, 6.9 ug/ml at 6 hours and 4.8 ug/ml at 24 hours. The second stage of release was characterized by a slow and continuous release for both antibiotics; the concentration range of DOXY release from day 2-7 was between (1-4 ug/ml) while MINO concentration release ranged between (1-5 ug/ml) in the same time period. Neither of the antibiotics was detected in the sample aliquots after day 7. Of the loaded DOXY and MINO samples, 19.9 % and 16.6 % was released respectively during the time period of 2-7 days. The total cumulative release percentage was 75.7% and 86.9% for DOXY and MINO, respectively.
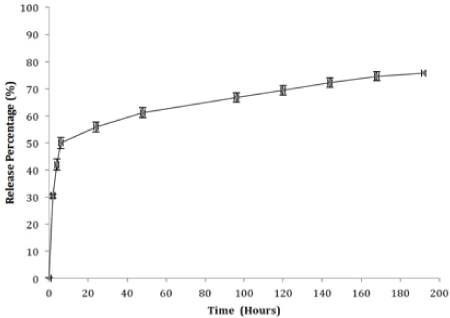
Figure 1: In vitro cumulative release percentage of doxycycline hyclate from preloaded nanocalcium sulfate. Release of doxycycline (DOXY) from preloaded nanocalcium (nCS) samples was measured in phosphate buffered saline (pH 7.4) at 37°C as described in Methods and Materials. Data shown are the mean of 5 control and 5 test samples.
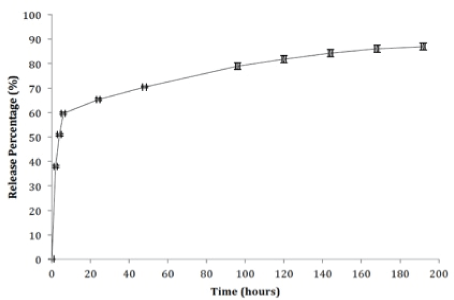
Figure 2: In vitro cumulative minocycline hydrochloride (MINO) from preloaded nanocalcium sulfate. Release of minocycline (MINO) from preloaded nanocalcium (nCS) samples was measured in phosphate buffered saline (pH 7.4) at 37°C as described in Methods and Materials. Data shown are the mean of 5 control and 5 test samples.
MTT Test for Doxycycline loaded nCS samples
Samples of nCS loaded with low concentrations of DOXY (10 or 5 ug) showed an increase in cellular activity statistically significantly different (p<0.05) from control, nonloaded nCS (Fig 3). Incubation of these cells with nCS loaded with DOXY at concentrations of 50 and 25 ug produced no significant differences (p>0.05) in cellular activity of these osteoblasts compared to nonleaded nCS controls. However, when nCS was loaded with DOXY at higher concentration of 100 and 500 ug osteoblast cell activity was significantly reduced compared to controls (p< 0.05).
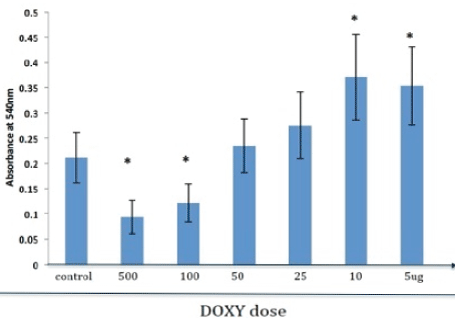
Figure 3: Cell activity of primary osteoblasts with doxycycline (DOXY) loaded nanocalcium (nCS). Osteoblastic cells were incubated for 48 hr with nCS samples preloaded with DOXY at various doses followed by the MTT assay with absorbance at 540 nm reflective of cell activity. Data are the mean +/- SD; * = difference from controls is significant at the P<0.05 level (n=6).
MTT Test for Minocycline loaded nCS samples
There was no significant difference (p<0.05) between control samples and test samples loaded with MINO at 5, 10, 25 and 50 ug. Higher loads of 100 and 500ug MINO produced statistically significant lower values from the control group and lower loaded samples indicating a reduction in the activity of primary osteoblasts (Figure 4).
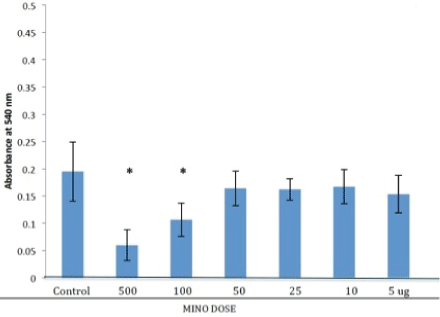
Figure 4: Cell activity of primary osteoblasts with minocycline (MINO) loaded nanocalcium sulfate (nCS). Osteoblastic cells were incubated for 48 hr with the nCS samples preloaded with MINO at various doses followed by the MTT assay with absorbance at 540 nm reflective of cell activity. Data are the mean +/= SD; * = difference from controls is significant at the P<0.05 level (n=6).
Alkaline Phosphatase) Activity and Antibiotic loaded nCS
Cells treated with nCS loaded with DOXY at 25, 50, 100 and 500 ug showed a gradual significant (p<0.05) increase in alkaline phosphatase activity compared to control, not loaded nCS samples while samples with lower amounts of DOXY (5 and 1 ug) showed no statistically significant difference (Figure 5). In contrast, MINO loaded nCS showed no statistically significant difference in any of the test groups when compared to control (nonloaded) nCS (Figure 6).
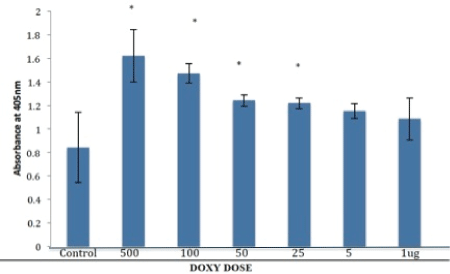
Figure 5: Alkaline phosphatase activity of primary osteoblasts with doxycycline (DOXY) loaded nanocalcium sulfate (nCS) samples. Osteoblastic cells were incubated for 48 hr with the nCS samples preloaded with DOXY at various doses followed by the ALP assay with absorbance at 405 nm reflective of alkaline phosphatase activity. Data are the mean+/- SD; * = difference from controls is significant at the P<0.05 (n=6).
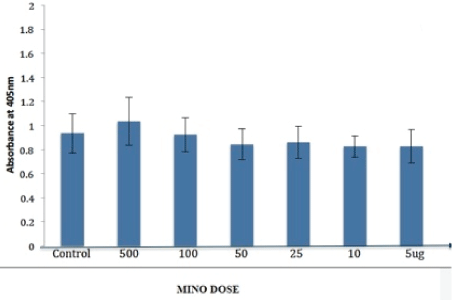
Figure 6: Alkaline phosphatase activity of primary osteoblasts with minocycline (MINO) loaded nanocalcium sulfate (nCS) samples. Osteoblastic cells were incubated for 48 hr with nCS samples preloaded with MINO at various doses followed by the ALP assay with absorbance at 405 nm reflective of alkaline phosphatase activity. No significant differences compared to control, (nonloaded) samples were observed; (P>0.05 (n=6) for all groups.
Antimicrobial activity drug loaded nCS samples
The antimicrobial effect of nCS samples loaded with DOXY and MINO in concentrations of 25 and 50 ug per pellet showed that these composite materials are capable of retarding the growth of P.gingivalis and decreasing turbidity at 24 hours when compared to control nCS without antibiotics. These results are illustrated in figure 7.

Figure 7: Antimicrobial activity against Porphyromonas gingivalis for doxycycline (DOXY) and minocycline (MINO) loaded nanocalcium sulfate (nCS) samples. Porphyromonas gingivalis at a concentration of 1 x 105 CFU/ml brain heart infusion broth was incubated with a nCS pellet containing a dose of 25 or 50 ug of either DOXY or MINO in 10 mL test tubes. An nCS pellet without any antibiotic was used for the positive control and a test tube with P. gingivalis alone served as negative control. All samples were incubated for 24 hrs in an anaerobic environment composed of 80% nitrogen, 10% carbon dioxide and 10% hydrogen atmosphere. After incubation, turbidity of the tubes was used as an indication of bacterial growth. The decreased turbidity in the tubes (observable here without magnification) containing nCS loaded with DOXY and MINO (both doses tested) compared to control and nCS alone is an indirect measure of the effectiveness of the released antibiotic to inhibit the bacterial growth.
Previous studies with the nanocalcium sulfate (nCS) product used here suggest that this material can be an effect scaffold for osseous regeneration in critical size defects [16,17]. Release profile results of loading nCS with human platelet lysates derived growth factor and bone morphogenetic protein suggest that the scaffold can be used for slow release delivery of biologic agents, therefore, providing a rationale to the incorporation of antimicrobials to induce healing in bone infection while simultaneously promoting bone regeneration [18]. To our knowledge, this is the first study that characterizes the ability of the nCS scaffold to affect the cell viability and differentiation of osteoblastic cells when loaded with tetracycline derivative antimicrobials.
Ultraviolet-Visible spectrophotometry analysis of DOXY solution in distilled water revealed two peaks in the wavelength at 267 and 351 nm. This result confirms other studies that have also reported similar findings at these wavelengths [25,26]. Our wavelength scan of MINO revealed only one peak at 353nm when distilled water was used as a solvent. A study by Mohite et al, reports; maximum wavelength absorption at 289 mm [27]. The differences in these wavelengths are most likely explained by the use of different solvent characteristics or different antibiotic manufacturing formulas [28].
Both DOXY and MINO showed release profiles that were characterized by an initial burst period that was then followed by sustained delivery in the release media for one week. According to Hezaraki et al, initial burst release of preloaded antibiotics is a unique characteristic of medical grade calcium sulfate [29]. Our findings agree with this type of release behavior, however, nCS showed less initial release burst (50-60%) and a more prolonged controlled release that continued for 7 days. Contrary to what was observed in our study here, the previous study with medical grade calcium sulfate showed that 80-85% of the loaded antibiotic was liberated within the first 10 hours and almost the whole amount of antibiotic was released in 48 hours [29].
These differences in release timing can most likely be due to the unique nano-structure of our calcium sulfate scaffold. Previous studies with nCS provide evidence that the diameter of nCS particles are ten times smaller than conventional medical grade calcium sulfate, hence increasing the scaffold pore diameter, surface area for more drug loading and improving the release potential of the absorbed material [16].
In terms of antimicrobial activity, the susceptibility and resistance of DOXY and MINO have been extensively studied against a large number of oral anaerobes [30-32]. O’Conner et al. [30] reported that, a concentration of 1ug/ml MINO was found to inhibit 55 strains of subgingival plaque bacteria while the minimal inhibitory concentration (MIC) ranged from 0.03 to 32 ug/ml. Similarly in another study by Sutter et al. [32], the MIC for MINO for a large number of oral anaerobes was found to be within the same range. Based on our results, both DOXY and MINO concentrations released from the nCS pellets were above the MIC for most common periodontal pathogens. Furthermore, these composite materials were capable of retarding the growth of P.gingivalis and decreasing turbidity at 24 hours when compared to control nCS without antibiotics (Figure 7).
The MTT cell activity assay was used as sensitive method to assess the metabolic activity/viability of human primary osteoblasts. Our results demonstrated that when nCS was preloaded with relatively low concentrations (5 and 10 ug) of Doxycycline hyclate, a significant increase in cell activity was seen when compared to the control group (p<0.05). Higher doses of this antibiotic produced either no effect or inhibitory effects on the primary alveolar osteoblastic cells tested here. These findings are in agreement with previous in-vitro studies, which show that low dose doxycycline is able to promote the proliferation and differentiation of primary osteoblasts [20,33], while higher doses can have inhibitory effects [23,32]. Although MINO is generally thought to be more lipophilic than DOXY, allowing it to more easily penetrate cell walls and increase cytotoxicity [33], we saw evidence of inhibitory effects of this antibiotic only at the relatively high doses at which DOXY produced similar decreases in osteoblastic cell activity with no evidence of stimulatory effects at the lower levels of MINO.
The results obtained here on the stimulatory effects of various doses of DOXY on alkaline phosphatase activity levels, well characterized as a marker of osteoblastic cell differentiation, in the primary human osteoblastic cells support previous studies with this antibiotic that suggest that it can be a potent inducer of osteoblastic cell differentiation [20,34]. Although MINO has been shown in other osteoblastic cell systems to decrease differentiation even at low concentrations [20,35], we did not observe any effect of this antibiotic at the range of doses that we used to preload the nCS material. The in-vitro microbiological assessment of antibiotic-loaded nCS presented here clearly demonstrates the ability of the scaffold to retard growth turbidity of P.gingivalis in BHI broth after 24 hours and suggests that the antimicrobial activity of DOXY or MINO is not affected by the presence of the calcium or sulfate present in the scaffold or the reaction conditions as the material solidifies.
From the present study we conclude that nanocalcium sulfate can be used as a controlled delivery system for antibiotics in-vitro. The results of both the MTT activity and alkaline phosphatase activity, indicative of proliferation and differentiation respectively show that the sustained release of DOXY from nCS has the ability to induce cell activity at low concentrations and enhance the metabolic activity and differentiation of osteoblasts as the concentration of the drug increases within the sample. On the other hand, MINO does not seem to affect osteoblast differentiation when used at various concentrations. Based on our results, DOXY and MINO concentrations that range between 0.25-0.5 mg/mL appear to retain effective antimicrobial activity and fall within a recommended safety margin of use on primary osteoblasts when mixed with an nCS scaffold. At least one previous study has reported that the addition of various antimicrobial agents can affect the setting time of calcium sulfate plasters used clinically in orthopedic surgeries [21]. Although DOXY or MINO were not studied in this previous study, our results in this present study suggest that under the conditions tested here, both of these antibiotics can slightly alter both the initial and final setting times of nCS, however, only DOXY produced statistically significant increases. Vickers hardness testing also revealed differences with the addition of DOXY or MINO. Although the clinician might wish to make adjustments in the ratio of nCS to solvent to attain the desired setting and hardness properties for a particular procedure, it is anticipated that this can be easily done without significantly compromising the release and scaffold properties of the material. In this present study the dissolution /resorption rates of nCS with or without the addition of antibiotics were not directly studied. However, previous studies showed that under similar in vitro conditions as those used here, only 28% of the nCS samples underwent dissolution over a 4 week period [36] in agreement with the general observations made in this present study that the nCS samples remained intact during the 7 day period that was used in some of the release experiments. It should be noted, however, that these measurements are dependent upon the particular in vitro conditions of experimentation and not necessarily representative of in vivo conditions at the site of implantation.
Conclusion
In summary, the studies presented here provide supportive data that nano-sized calcium sulfate has the potential to be an effective antibiotic releasing bone scaffold suitable for clinical procedures where calcium sulfate has been successfully utilized.
Conflict of Interest
Dr. Rosemary Dziak is an inventor on the patent: “Calcium sulfate based nanoparticles.” U.S. Patent 7, 767,226, issued August 3, 2010 and a partner in ProOsseous LLC.








